
Catalysis Science and Technology p. 3281 - 3287 (2015)
Update date:2022-08-11
Topics:
 Wen, Xin
Wen, Xin
 Cao, Yingying
Cao, Yingying
 Qiao, Xianliang
Qiao, Xianliang
 Niu, Libo
Niu, Libo
 Huo, Li
Huo, Li
 Bai, Guoyi
Bai, Guoyi
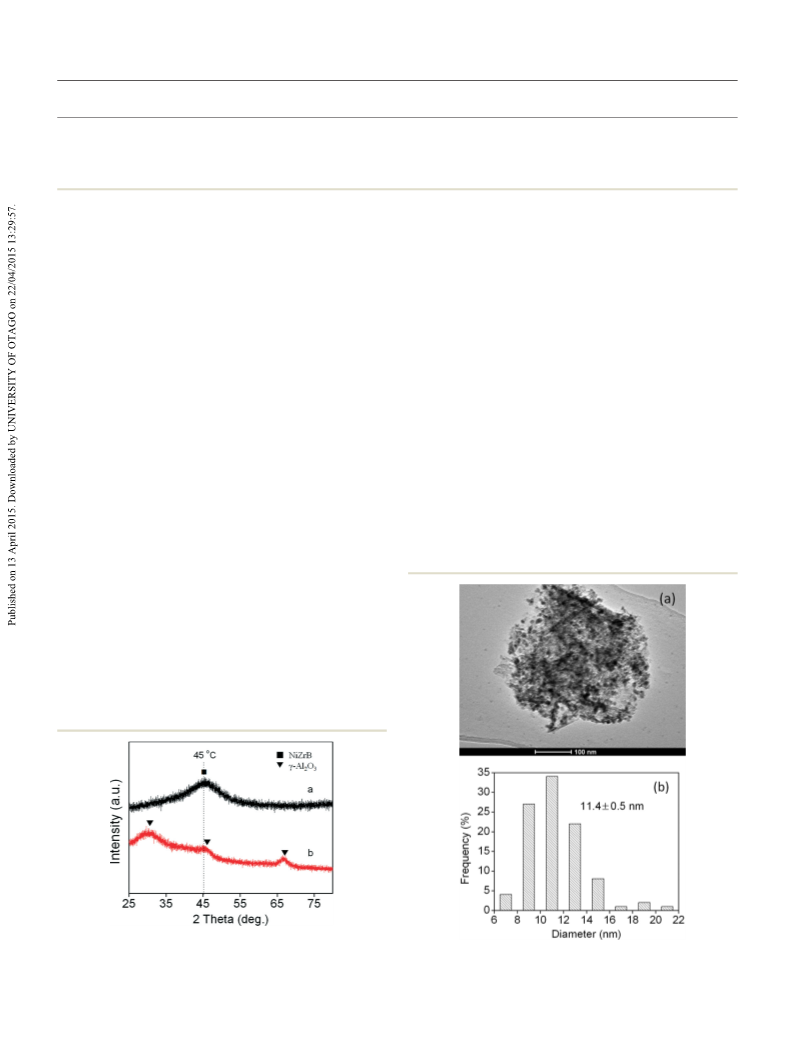
This study presents a facile way to improve the selectivity for cyclohexanecarboxylic acid by adding a base in the hydrogenation of benzoic acid over a non-noble metal NiZrB amorphous alloy supported on γ-Al2O3. It is found that alkali metal carbonates exhibit an excellent selectivity improvement from 50.3% to a range of 93.5-95.7%, with the conversion of benzoic acid higher than 92.3%. Even a very small amount of K2CO3 (1 mol% benzoic acid) was efficient for improving the selectivity for cyclohexanecarboxylic acid. In addition, a lower reaction temperature was beneficial to the improvement of selectivity. Based on the results of temperature programmed desorption of NH3 and inductively coupled plasma analysis, the improvement of selectivity in the presence of a base was attributed to the neutralization of the acidic sites on the surface of the catalyst by the in situ generated potassium benzoate, inhibiting the hydrodeoxygenation of carbonyl and resulting in a high selectivity for cyclohexanecarboxylic acid.
View More





Wuhan Kemi-Works Chemical Co., Ltd
website:http://www.kemiworks.com
Contact:86-27-85736489
Address:Rm. 1503, No. 164, Jianghan North Rd., Wuhan, 430022 China
website:http://www.apeptides.com/en/
Contact:+86-21-60871011
Address:No. 80 Chuanshan Shuyuan Steet,Pudong,Shanghai
Contact:
Address:308# dongwu avenue dongxihu district wuhan city
ZHIJIANG ZENVA SINO COMMERCE AND TRADE CO., LTD.
website:http://www.zenvasino.com
Contact:+86-138-72658998
Address:Shibeishan Road,Zhijiang, Hubei, China
Tianjin Dongchang Fine Chemical Industry Co., Ltd.
Contact:+86-22-29894595
Address:Economic Developing Zone, Ji County, Tianjin, China
Doi:10.1016/0040-4039(88)80009-1
(1988)Doi:10.1002/jhet.2747
(2017)Doi:10.1039/c9cc01937e
(2019)Doi:10.1021/ja00176a041
(1990)Doi:10.1016/0040-4039(84)80062-3
(1984)Doi:10.1021/ja01151a507
(1951)