NEW CHEMICAL MARKERS BASED ON PHTHALEINS
713
get on the object being marked. The phthaleins are used
separately as purgative, antiseptic, and antihelminth
agents in medicine; they are also used in perfumery and
as indicators in laboratories and in industry [4].
of nitrobenzene in a metal crucible, after which 5.6 g
(0.052 mol) of o-cresol was added, and the mixture was
heated to 50–60°С and stirred until the phenols dissolved
completely. Then, 16.5 g (0.11 mol) of phthalic anhydride
was added, the mixture was stirred, and 14.2 g (0.104 mol)
The chemical marker was prepared as follows.A100-
g portion of the marker was prepared from the base
mixture of phthaleins (57.0 g) and functional components
of anhydrous powdered ZnCl was added. In the second
2
step, the temperature was raised to 120–130°С, and the
mixture was heated for 4 h with intermittent stirring with
a putty knife. The following reactant ratio was used in the
(
43.0 g). The base mixture of phthaleins, containing
4
5
5.0 g of phenolphthalein, 5.9 g of o-cresolphthalein,
.6 g of thymolphthalein, and 0.5 g of fluorescein, was
synthesis: phthalic anhydride : phenol : ZnCl : o-cresol =
2
2.15 : 1.0 : 2.0 : 1.0 ; phenol : nitrobenzene = 1.0 : 3.4. The
mixed in the dry form, loaded into a laboratory mill, and
milled for 5 min. To prepare a 12% OAPP-N solution,
product yield after purification was 34.0% (5.9 g). The
product contained (wt %) 15.0 PP, 34.7 CP, and 50.3 CPP.
1
3.5 g of OAPP-N was loaded into a heated glass reactor
with a stirrer, 151.0 mL (99.7 g) of n-hexane was added,
and the mixture was stirred for 30 min at 50–60°С until
the polypropylene dissolved completely. The powdered
base mixture of phthaleins (57.0 g) was gradually added
to the solution, the suspension temperature was elevated
to 68.7°С, and the major fraction of hexane (100 mL)
was distilled off. The wet powder was unloaded from
the reactor onto a sheet of thick white paper, distributed
as a 5-mm-thick layer, and allowed to completely dry at
room temperature (25°С). The powder was intermittently
mixed with a putty knife to break down phthalein clots.
The dry powder of phthaleins does not adhere to the
putty knife and does not have the hexane odor. The base
mixture of phthaleins with the polymer deposited onto the
particle surface was loaded into a homogenizer, 26.0 g of
KSMG grade silica gel preliminarily ground in a mortar
and 3.5 g of citric acid were added, and the dry mixture
was homogenized for 5 min with vigorous stirring. The
prepared chemical marker was charged in 10-g portions
into small polyethylene bags. The finely dispersed powder
of the marker has gray color, is friable, and can be readily
distributed over the surface without particle aggregation.
The phthaleins were isolated from the reaction
mixture after distilling off nitrobenzene in a vacuum.
The solid residue was ground and boiled in a porcelain
cup with 100 mL of distilled water acidified with 5 mL
of concentrated HCl. The solution was filtered, and
the precipitate was washed with water. The washed
precipitate was dissolved in a warm 5% NaOH solution.
The resulting dark red solution was filtered to remove the
insoluble precipitate. The red filtrate was acidified with
glacial acetic acid added in 1-mL portions and then with
six drops of hydrochloric acid to obtain an acidic solution.
In the process, the solution color changed from dark
red to light brown. The solution was left overnight for
precipitation. The precipitate was filtered off and dried.
The reaction product was purified as follows. The
product was dissolved in approximately 50 mLof alcohol,
and the solution was refluxed with activated charcoal. The
hot solution was filtered, and the charcoal was washed
with hot alcohol. After cooling, the solution was diluted
with an eightfold amount of distilled water, filtered,
partially evaporated in a porcelain cup, and cooled. The
precipitated product was filtered off, dried, and weighed.
The reaction product is light brown. The synthesized
mixture of three homologs was used for preparing 9.1 g of
a chemical marker of the following composition (wt %):
PP 9.7 (0.9 g), CP 22.6 (2.0 g), silica gel 20.0 (1.8 g),
citric acid 3.0 (0.3 g), OAPP-N 12.0 (1.1 g), and CPP
32.7 (3.0 g). The marker obtained, consisting of a mixture
of three phthalein homologs including CPP, allows more
reliable marking, and its falsification (copying) becomes
less probable; also, the marker formula is simpler.
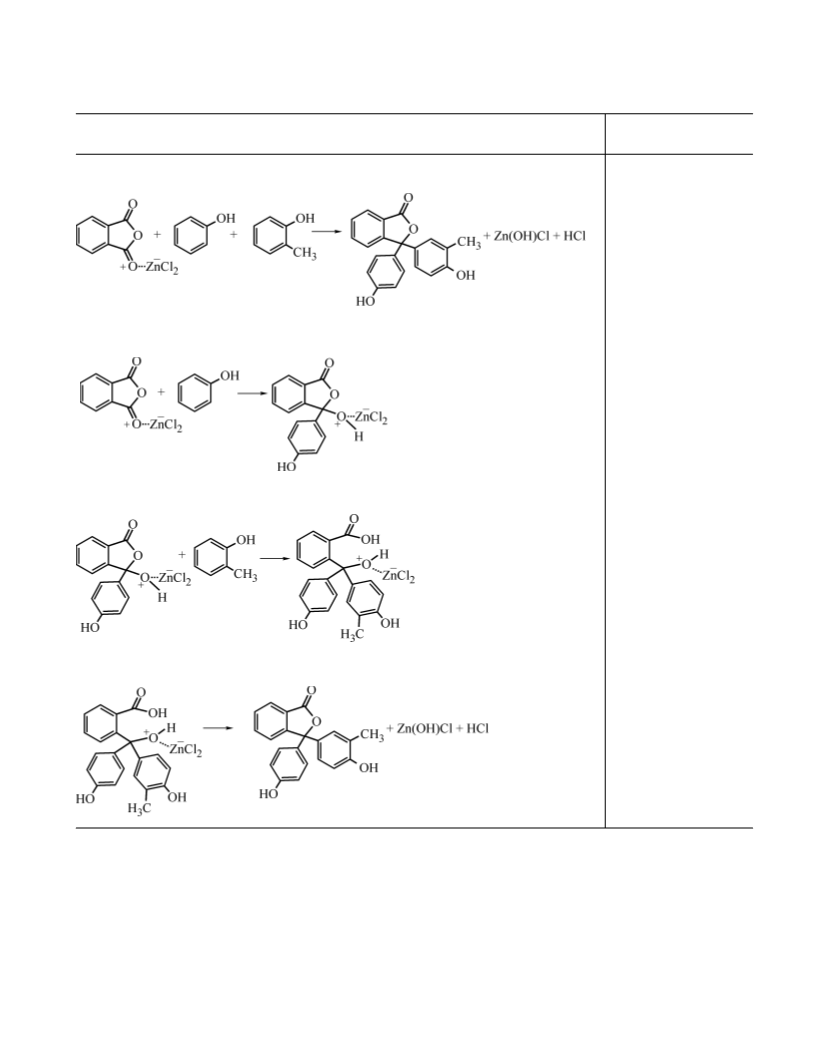
The second marker was prepared by synthesis
of three phthalein homologs including PP, CP, and
previously unknown phthalein with unsymmetrical
phenolic substituents, o-cresolphenolphthalein (CPP)
[
3-(3'-methyl-4'-hydroxyphenyl)-3-(4''-hydroxyphenyl)
phthalide]. The reaction was performed as follows [5].
First, we prepared the catalyst based on anhydrous
ZnCl . ZnCl was melted on a metal spoon over an alcohol
2
2
burner flame. The melt was heated until bubbles ceased to
evolve (≈10 min), after which it was cooled, and the cake
was ground in a mortar. The condensation was performed
in two steps using nitrobenzene as solvent. First, 4.9 g
Quantum-chemical calculations were performed using
standard GAUSSIAN’09 package [6]. Theoretical studies
were made using the density functional theory (DFT).
The calculations were performed by the B3LYP hybrid
(0.052 mol) of phenol was added to 18.6 mL (0.18 mol)
RUSSIAN JOURNAL OF APPLIED CHEMISTRY Vol. 88 No. 4 2015

 Nekhoroshev
Nekhoroshev
 Nekhoroshev
Nekhoroshev
 Poleshchuk
Poleshchuk
 Yarkova
Yarkova
 Nekhorosheva
Nekhorosheva
 Gasparyan
Gasparyan