
Journal of Molecular Structure p. 191 - 199 (2006)
Update date:2022-08-30
Topics:
 Tayyari
Tayyari
 Moosavi-Tekyeh
Moosavi-Tekyeh
 Zahedi-Tabrizi
Zahedi-Tabrizi
 Eshghi
Eshghi
 Emampour
Emampour
 Rahemi
Rahemi
 Hassanpour
Hassanpour
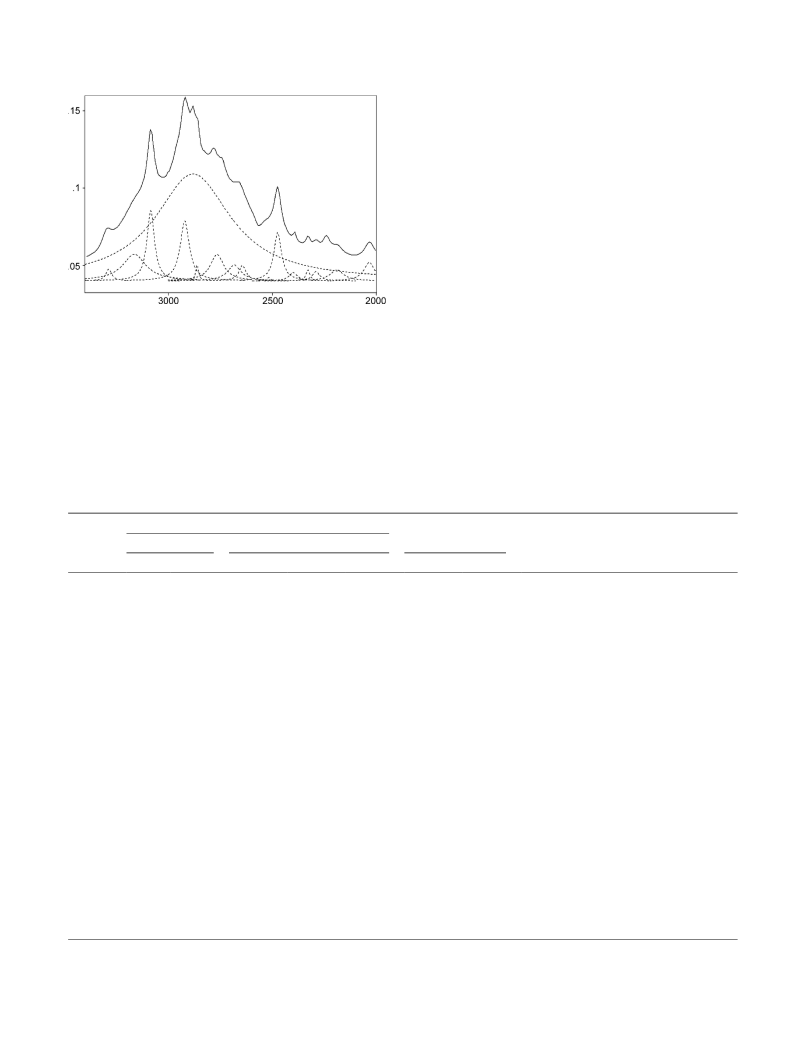
2-Nitromalonaldehyde (NO2MA), a simple compound to study the intramolecular hydrogen bond, has been synthesized and deuterated at the enolated proton. Molecular structure and vibrational frequencies of NO 2MA have been investigated by means of density functional theory (DFT) calculations. The geometrical parameters obtained in the B3LYP level using 6-31G**, 6-311G**, and 6-311++G** basis sets and compared with the corresponding parameters of malonaldehyde (MA). Frequencies calculated at B3LYP level using the 6-311G** and 6-311++G** basis sets are in good agreement with the corresponding experimental results for light and deuterated compounds in CCl 4/CS2 solution. The percentage of deviation of the bond lengths and bond angles was used to give a picture of the normal modes, and serves as a basis for the assignment of the wavenumbers. Theoretical calculations show that the hydrogen bond strength of NO2MA is slightly stronger than that of MA, which is in agreement with the spectroscopic results. The observed νOH/νOD and γOH/γOD appears at about 2880/2100 and 911/695 cm-1, respectively, are consistent with the calculated geometry and proton chemical shift results. To investigate the effect of NO2 group on the hydrogen bond strength, the charge distributions, steric effects, and electron delocalization in NO2MA and MA were studied by the Natural Bond Orbital (NBO) method for optimized model compounds at B3LYP/6-311++G** level of theory. The results of NBO analysis indicate that the electron-withdrawing effect of NO2 group decreases the hydrogen bond strength, but the steric and resonance effects increase the strength of the bond.
View More





Changyi Xinxing Technology Development Co., Ltd.
Contact:0086-510-86651065(Time Zone:8),86651656
Address:94 Yingbinxilu WestRoad, Huangtu Town, Jiangyin City, Jiangsu, China
Guangxi Nanning Guangtai Agriculture Chemical Co.,Ltd
Contact:+86-771-2311266
Address:Room703,Building12, Software Park Phase II,NO.68,Keyuan Road,Nanning City,Guangxi,China
Zhangjiagang Golden Reach Fine Chemical Co.,LTD.
Contact:+86-512-6585 6968
Address:Changfu Road, Dongsha Chemical Industry Park, Zhangjiagang City, Jiangsu Province, China
Anhui Xinyuan Technology Co.,Ltd
website:http://www.ahxytech.com/
Contact:0086-559-3515800
Address:Huangshan City of Anhui Province Huizhou Huizhou District Road, 21-9
Wuhan Chemwish Technology Co., Ltd
website:http://www.chemwish.com/
Contact:+86-27-67849912
Address:Room 1311, Unit 2, Block1, Innovation Road East Lake High-tech Development Zone Wuhan, Hubei,P.R. China
Doi:10.1016/S0022-1139(00)81520-1
(1982)Doi:10.1055/s-2003-41432
(2003)Doi:10.1021/ja01184a039
(1948)Doi:10.1080/10426500600732808
(2006)Doi:10.1016/0022-328X(96)06203-1
(1996)Doi:10.1016/S0040-4039(96)02181-8
(1996)