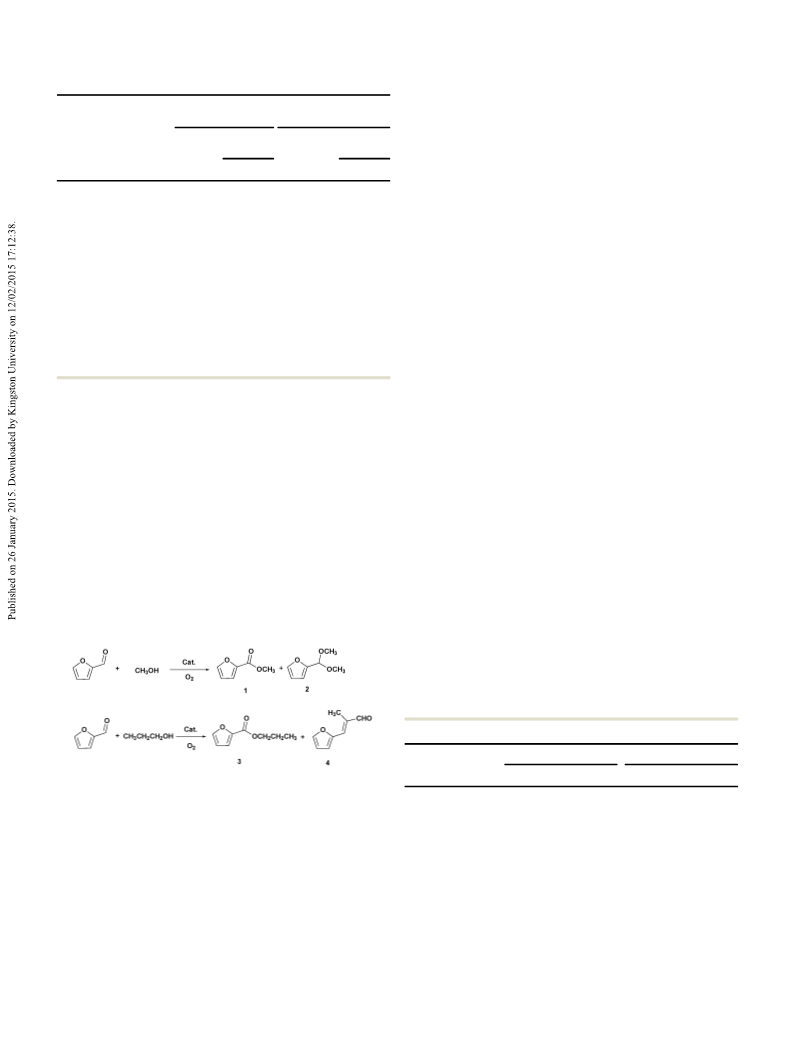Communication
ChemComm
4
2% conversion and 99% selectivity for 2-(dipropoxymethyl) furan Notes and references
were obtained when the reaction of FUR with n-propanol is
performed under a nitrogen atmosphere (entry 7). Particularly,
the reaction of FUR and propanal in the ratio of 10 : 1 was also
performed, in which 99% conversion of propanal and 99%
selectivity for 4 were attained in the presence of a Au catalyst
1
2
3
Y. Roman-Leshkov, J. N. Chheda and J. A. Dumesic, Science, 2006,
3
12, 1933; C. Zhou, X. Xia, C. Lin, D. Tong and J. Beltramini, Chem.
Soc. Rev., 2011, 40, 5588; J. C. Serrano-Ruiz and J. A. Dumesic, Energy
Environ. Sci., 2011, 4, 83.
J. Q. Bond, A. A. Upadhye, H. Olcay, G. A. Tompsett, J. Jae, R. Xing,
D. M. Alonso, D. Wang, T. Zhang, R. Kumar, A. Foster, S. M. Sen,
C. T. Maravelias, R. Malina, S. R. H. Barrett, R. Lobo, C. E. Wyman,
J. A. Dumesic and G. W. Huber, Energy Environ. Sci., 2014, 7, 1500.
Biorefineries—Industrial Processes and Products, ed. B. Kamm,
P. R. Gruber and M. Kamm, Wiley-VCH, Weinheim, 2006.
2 3
and K CO (entry 8). This shows that highly selective production
of 4 is achieved when the reaction was carried out between a large
number of FUR and little propanal. So, it can be concluded that
the first step is the oxidation of little propanol to propanal, and
the next step is the rapid condensation happening between FUR
and very little in situ generated propanal in the FPO system
4 K. J. Zeitsch, The Chemistry and Technology of Furfural and its Many
By- Products, Elsevier Science, 2000; S. Lima, M. Pillinger and
A. A. Valente, Catal. Commun., 2008, 9, 2144.
5
H. Guo and G. Yin, J. Phys. Chem. C, 2011, 115, 17516.
(
see the lower-right route in Scheme 1). Correspondingly, in this
6 B. Kamm, P. R. Gruber and M. Kamm, Biorefineries - Industrial
Processes and Products Status Quo and Future Directions, Wiley-VCH,
reaction, the Au/FH catalyst is responsible for the selective oxida-
tion of propanol; while the role of K
subsequent condensation process.
In summary, tunable and efficient transformation of FUR with
aliphatic alcohols in the presence of molecular oxygen has been
achieved. In the presence of the Au/FH catalyst and potassium
carbonate, a oxidative esterification process occurs in the FMO
system, in which 91.8% yield of methyl 2-furoate with 98.7%
2005.
2 3
CO is to promote the
7
G. W. Huber, J. N. Chheda, C. J. Barrett and J. A. Dumesic, Science,
2005, 308, 1446; J. N. Chheda, G. W. Huber and J. A. Dumesic,
Angew. Chem., Int. Ed., 2007, 46, 2; R. West, Z. Y. Liu, M. Peter and
J. A. Dumesic, ChemSusChem, 2008, 1, 417; W. Shen, G. A. Tompsett,
K. D. Hammond, R. Xing, F. Dogan, C. P. Grey, W. C. Conner,
S. M. Auerbach and G. W. Huber, Appl. Catal., A, 2011, 392, 57;
S. Dutta, S. De, B. Saha and Md. I. Alam, Catal. Sci. Technol., 2012,
2
, 2025.
8
T. Mallat and A. Baiker, Chem. Rev., 2004, 104, 3037–3058.
selectivity is obtained at 140 1C for 4 h under 0.3 MPa O
2
, while a
9 E. Taaring, I. S. Nielsen, K. Egeblad, R. Madsen and C. H. Christensen,
ChemSusChem, 2008, 1, 75.
highly selective oxidative condensation process arises in the FPO
system where a 91.4% yield of 3-(furan-2-yl-)-2-methylacryaldehyde
with 97.2% selectivity is attained under the same conditions. This
will provide a promising approach for the efficient valorization of
hemicellulose in the biomass transformation.
We are grateful for the support of the National Natural
Science Foundation of China (No. 21003093) and the State
Key Programme of National Natural Science Foundation of
China (No. 21336008).
1
0 F. Pinna, A. Olivo, V. Trevisan, F. Menegazzo, M. Signoretto, M. Manzoli
and F. Boccuzzi, Catal. Today, 2013, 203, 196; M. Signorettoa,
F. Menegazzoa, L. Contessottoa, F. Pinnaa, M. Manzolib and
F. Boccuzzi, Appl. Catal., B, 2013, 129, 287; F. Menegazzo, T. Fantinel,
M. Signoretto, F. Pinna and M. Manzoli, J. Catal., 2014, 319, 61;
F. Menegazzo, M. Signoretto, F. Pinna, M. Manzoli, V. Aina, G. Cerrato
and F. Boccuzzi, J. Catal., 2014, 309, 241.
11 N. Zheng and G. D. Stucky, Chem. Commun., 2007, 3862; L. Wang,
J. Li, W. Dai, Y. Lv, Y. Zhang and S. Gao, Green Chem., 2014, 16,
2
164–2173; X. Bai, F. Ye, L. Zheng, G. Lai, C. Xia and L. Xu, Chem.
Commun., 2012, 48, 8592.
This journal is ©The Royal Society of Chemistry 2015
Chem. Commun., 2015, 51, 3674--3677 | 3677

 Tong, Xinli
Tong, Xinli
 Liu, Zonghui
Liu, Zonghui
 Yu, Linhao
Yu, Linhao
 Li, Yongdan
Li, Yongdan