G Model
CATTOD-10171; No. of Pages10
ARTICLE IN PRESS
C. Ampelli et al. / Catalysis Today xxx (2016) xxx–xxx
2
incorporation of Zr4+ into the ceria lattice improves the activa-
tion of methanol (to form methoxy species) and facilitates the -H
elimination of hemiacetal, an intermediate in the reaction. Gold-
graphene nanocomposite catalysts were also reported recently to
be highly efficient in the oxidative esterification [20], although in
contrast with the role of the support indicated by Li et al. [19].
The optimal type of support in the reaction is also unclear.
Menegazzo et al. [15] indicated zirconia as the best support for
gold nanoparticles by comparing the performances of ceria, tita-
nia and zirconia based catalysts. However, later the same authors
ammonia solution was added under vigorous stirring until a pH
of 8.6 was reached. After the complete precipitation, the hydroxide
suspension was aged for 20 h at 90 C. The aged hydroxide was fil-
◦
tered and washed with warm water until removal of chloride ions
◦
(confirmed by AgNO3 test). The solid was dried at 110 C overnight
◦
and calcined at 650 C for 3 h.
About 0.4 wt.% of gold was added by deposition-precipitation
(DP) at pH = 8.6. In a typical DP technique, the oxide support (2 g)
was suspended in 80 mL of an aqueous solution containing the gold
precursor (HAuCl4·3H2O, in a concentration corresponding to a the-
oretical Au loading of 1% wt) for 3 h and the pH was adjusted by the
addition of aqueous NaOH (0.5 M). After deposition of gold onto the
support (in the form of gold-hydroxide), the solid was washed, fil-
tered and dried at room temperature for 12 h and finally annealed
[
16] remarked the use of Au-CeO2 catalysts and Au supported on
sulphated ZrO catalysts [17]. Au nanoparticles supported on ceria-
2
alumina mixed oxides were instead suggested recently as active
catalysts by Smolentseva et al. [21], indicating that the oxygen stor-
age capacity of the support (possible only in ceria-based catalysts)
is the key element for the catalytic performances. In contrast with
this conclusion, Wang et al. [22] showed that a support like MgO for
gold nanoparticles, thus without oxygen storage capacity and acid
sites, has also excellent performances in the oxidative esterification
of aldehydes. Suzuki et al. [23] showed that supported gold-nickel
oxide nanoparticles have excellent performances in the oxidative
esterification. In this case, a core-shell structure is present, with
the Au nanoparticles at the core and the surface covered by highly
oxidized NiOx. This result may thus question the idea proposed by
many authors [24] that sites at the gold nanoparticle-support inter-
face are those active in selective oxidation reactions. Mononuclear
gold species present in the solution, deriving from the dissolution
of gold nanoparticles, are instead proposed by Hashmi et al. [25] as
the active species in the oxidative esterification.
There is thus a relevant interest on this reaction, although still
aspects are under discussion related to the exact nature of the active
sites and the reaction mechanism. In a high-throughput study on
this reaction [26] we observed that gold on zirconia is effectively
the catalyst giving the best performances under diluted conditions,
but with the performances strongly depending on the modalities
of preparation of the zirconia support. In addition, tests using a
much higher furfural to methanol ratio (no solvent and bases), as
necessary for the industrial development of the process, resulted
in a quite different ranking of the catalysts.
◦
◦
at two different temperatures (200 C, 400 C) in air. These catalysts
are indicated hereafter as AuZ200 and AZ400, respectively.
2.2. Characterization of the catalysts
Atomic absorption spectroscopy (AAnalyst 200 Perkin Elmer)
was used to determine the gold amount after dispersion of the sam-
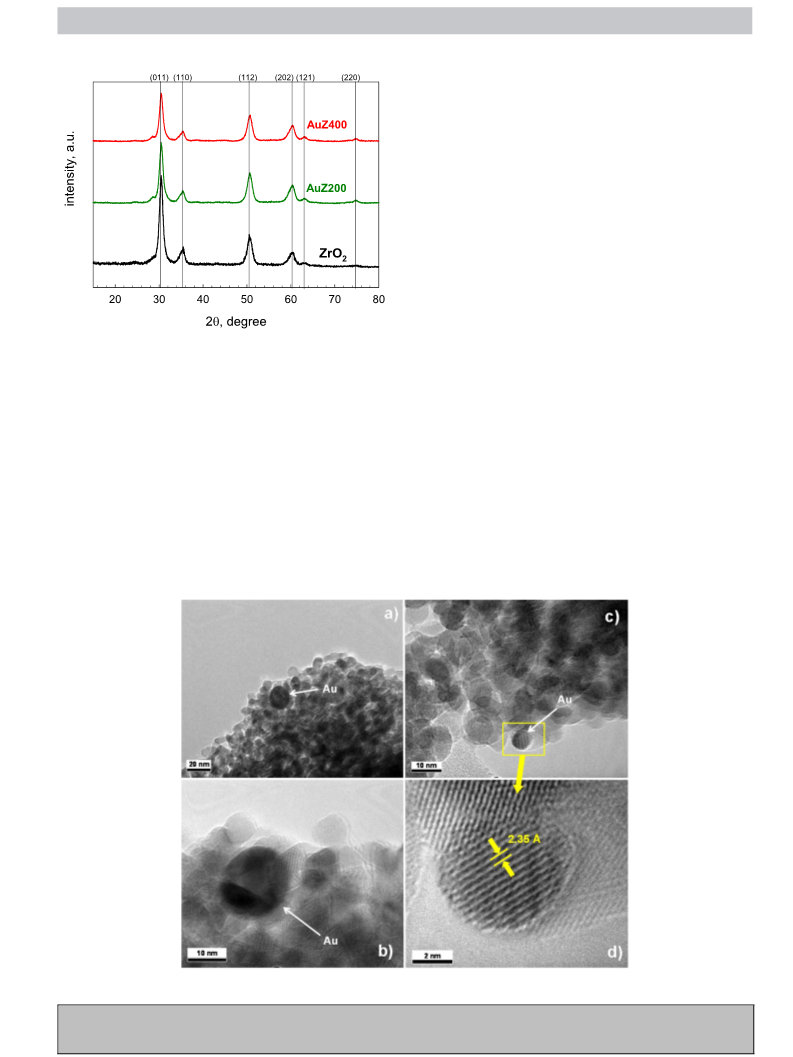
ples in HF by ultrasounds. The phase composition and the degree
of crystallinity were analysed by X-ray diffraction analysis with a
ADP 2000 diffractometer using a Cu-K␣ radiation. Data were col-
◦
−1
◦
lected at a scanning rate of 0.025 s in a 2 range from 15 to 80 .
Diffraction peak identification was made on the basis of the JCPDS
database of reference compounds.
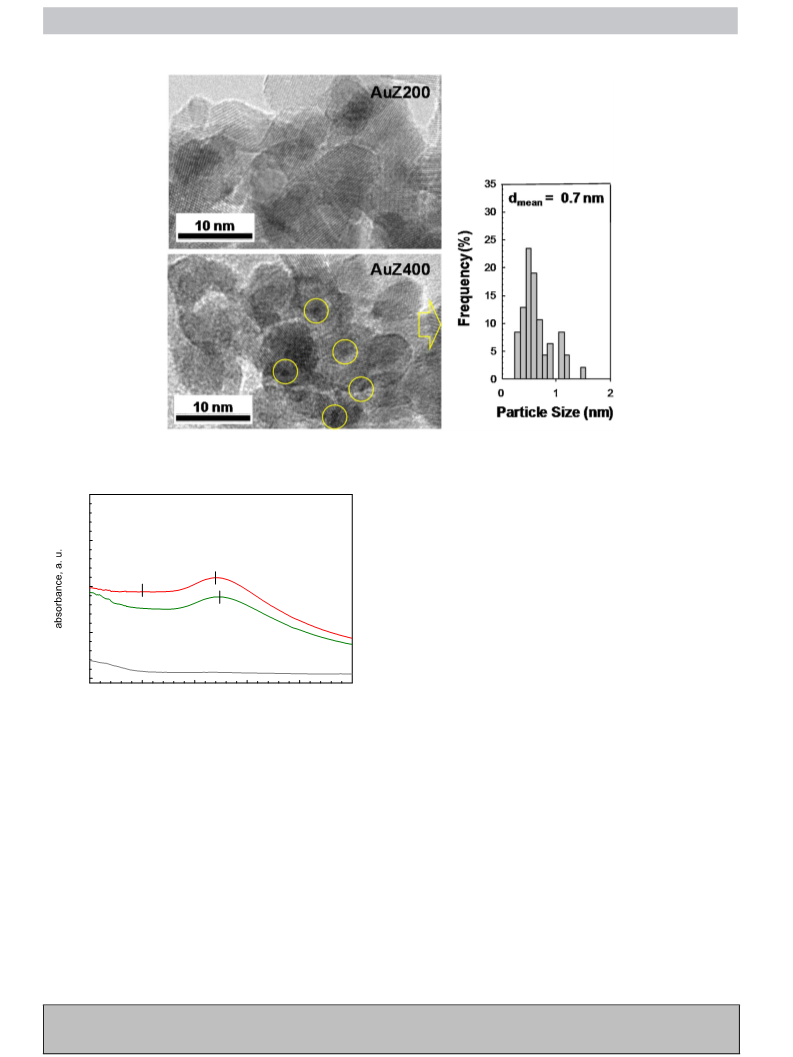
Transmission electron microscopy (TEM) images were acquired
by using a Philips CM12 microscope (resolution 0.2 nm) with
an accelerating voltage of 120 kV, while the elemental analysis
was investigated by using a Philips XL-30-FEG scanning electron
microscope equipped with an energy dispersive X-ray analyser.
Ultraviolet visible diffuse reflectance spectra were recorded by a
Jasco V570 spectrometer equipped with an integrating sphere for
solid samples using BaSO4 as reference. BET surface area and pore
size distributions were determined using N adsorption/desorption
2
isotherm at 77 K with an Autosorb iQ (Quantachrome Instruments)
system.
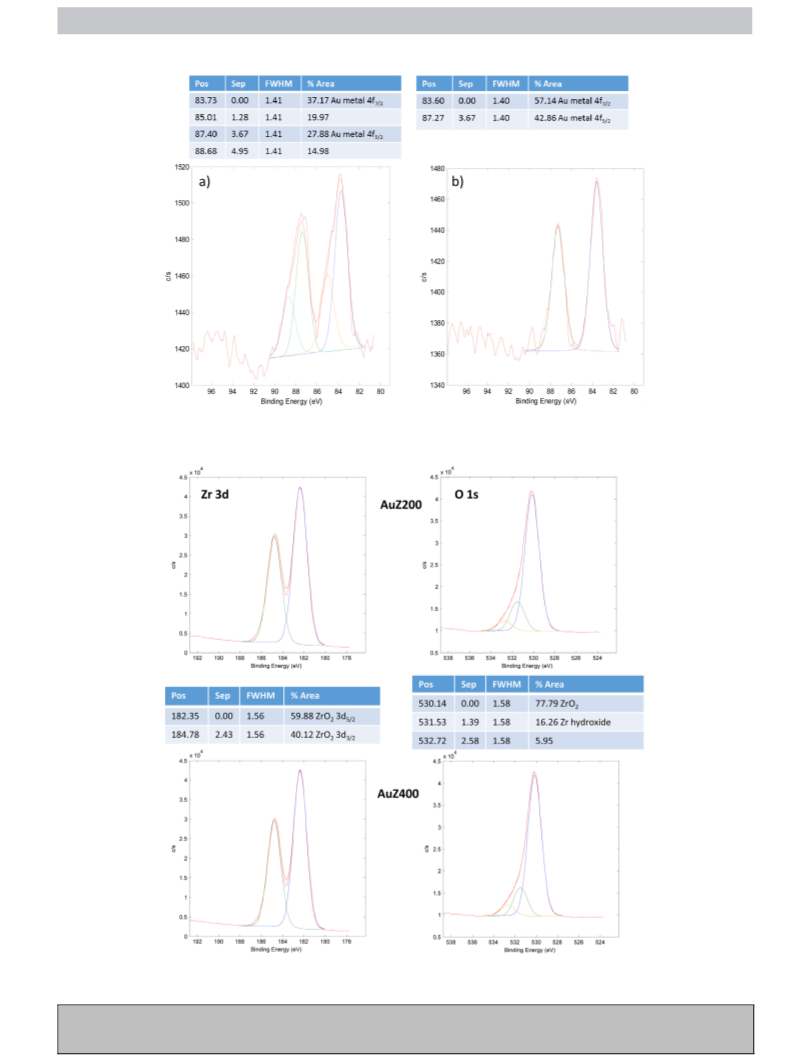
XPS measurements were performed on a PHI Quantera (Physical
Electronics). The survey and high resolution spectra were taken
using the “large area analysis” which is a 100 W 100 m diameter
X-ray beam scanned over a 1300 × 100 m area at high speed. High
resolution spectra were obtained with a pass energy of 26 eV. The
energy scale for all samples were adjusted by placing the C 1 s peak
from adventitious carbon at 284.8 eV. The spectra were fitted by
software and the resulting peak position, FWHM and percentage of
the total area are given.
The aim of this work is to investigate in detail two Au-ZrO2 cat-
◦
alysts, with the second derived from the first, calcined at 400 C
◦
rather than at 200 C. Giving these two catalysts a quite different
behavior in the oxidative esterification of furfural, the comparison
of their physico-chemical properties provides an excellent basis to
derive indications on the nature of the active sites in this reaction,
and about the possible role of the support as well as of the reac-
tion conditions. In order to allow a more reliable comparison of
the catalytic performances of these samples, the reactivity of the
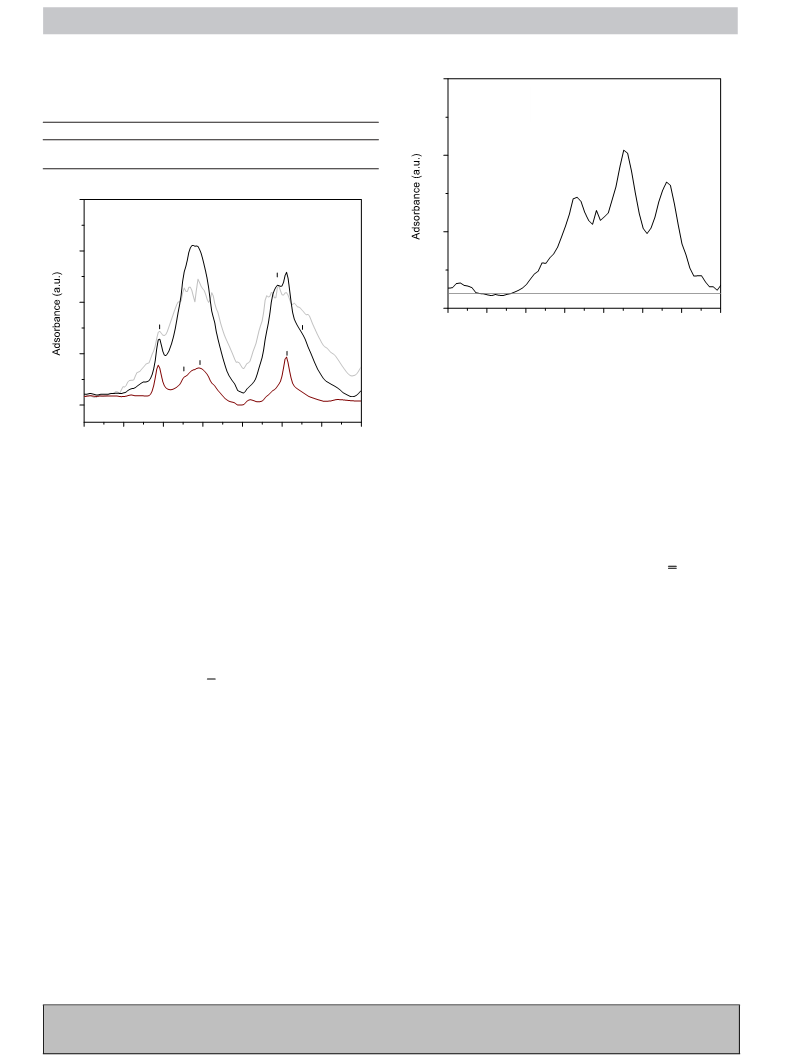
FTIR measurements were carried out on self-supporting wafers
TM
−1
reference AUROlite
Au/TiO2 catalyst (a commercially available
and the spectra were collected at a resolution of 4 cm
with a
standard reference gold-based catalyst) is also reported.
Bruker IF55 FTIR spectrophotometer equipped with a MCT detec-
tor. The self-supporting wafers were prepared and activated under
−
4
◦
vacuum (10 Torr) for 2 h at 200 C in an IR cell allowing in situ
thermal treatments, and pyridine dosage. Lewis and Brønsted acidic
sites quantification was performed by integrating the area under-
2
. Experimental
−
1
2.1. Preparation of the catalysts
neath the bands at 1450 and 1545 cm . The integrated molar
extinction coefficients (IMEC) were calculated, according to the
Beer’s law, by dosing pyridine (Py) at increasing concentrations
over each sample under study, after evacuation at rt under high
vacuum: A = ·c·D, where A = absorbance, = molar extinction coef-
ficient, c = concentration and D = path length. The value was
obtained by plotting A vs c (mol Py), assuming a constant D (since
the sample is in a pellet form and the thickness is always constant).
Hence, Brønsted and Lewis acid sites were quantified using the
AUROliteTM Au/TiO2 (1 wt.% Au) was a commercial sample pro-
vided by Strem Chemicals Inc. in the form of small extrudates.
Before testing the catalytic activity, the pellets were crushed in
a mortar until a very fine powder was obtained and then sieved
to particle size distribution of 120–250 mesh, i.e. 0.063–0.125 mm.
This particle size distribution was used also for the other catalysts.
All the reagents were of analytical grade and bought from Sigma
Zirconium(IV) oxychloride octahydrate (ZrOCl ·8H O), used as zir-
2
2
CPyonXsites = (Areapellet·IA(X))/(IMEC(X)·Weightpellet)
(1)
conia source, was dissolved in distilled water, then a diluted
Please cite this article in press as: C. Ampelli, et al., On the nature of the active sites in the selective oxidative esterification of furfural

 Ampelli
Ampelli
 Barbera
Barbera
 Centi
Centi
 Genovese
Genovese
 Papanikolaou
Papanikolaou
 Perathoner
Perathoner
 Schouten
Schouten
 van der Waal
van der Waal