Paper
RSC Advances
the third cycle. Thus, the new [GLY(mim)3][NTf2]3 IL/DBU catalyst 11 R. Nomura, Y. Hasegawa, M. Ishimoto, T. Toyosaki and
system can be reused at least three times without any prominent
change in its DMC selectivity.
H. Matsuda, J. Org. Chem., 1992, 57, 7339–7342.
12 P. P. Pescarmona and M. Taherimehr, Catal. Sci. Technol.,
2012, 2, 2169–2187.
13 B. A. V. Santos, V. M. T. M. Silva, J. M. Loureiro and
A. E. Rodrigues, ChemBioEng Rev., 2014, 1, 214–229.
14 M. North, R. Pasquale and C. Young, Green Chem., 2010, 12,
1514–1539.
15 M. North and R. Pasquale, Angew. Chem., Int. Ed., 2009, 48,
2946–2948.
16 F. Shi, Y. Deng, T. SiMa, J. Peng, Y. Gu and B. Qiao, Angew.
Chem., Int. Ed., 2003, 42, 3257–3260.
17 S. Fang and K. Fujimoto, Appl. Catal., A, 1996, 142, L1–L3.
18 S. Wada, K. Oka, K. Watanabe and Y. Izumi, Front. Chem.,
2013, 1, 8.
19 M. Honda, M. Tamura, Y. Nakagawa and K. Tomishige,
Catal. Sci. Technol., 2014, 4, 2830–2845.
20 A. A. Chaugule, A. H. Tamboli, F. A. Sheikh, W. J. Chung and
H. Kim, J. Mol. Liq., 2015, 208, 314–321.
21 H. J. Buysch, in Ullmann's Encyclopedia of Industrial
Chemistry, Wiley-VCH Verlag GmbH & Co. KGaA, 2000, vol.
26, pp. 521–576.
22 F. Bustamante, A. S. F. Orrego, S. N. Villegas and
A. D. L. Villa, Ind. Eng. Chem. Res., 2012, 51, 8945–8956.
23 E. I. Izgorodina, J. L. Hodgson, D. C. Weis, S. J. Pas and
D. R. MacFarlane, J. Phys. Chem. B, 2015, 119, 11748–
11759.
4. Conclusions
In summary, we have proposed a strategy to enhance the
conversion of a thermodynamically unfavorable chemical
reaction using the properties of novel tri-cationic RTILs. The tri-
cationic RTILs/DBU catalyst system has been successfully used
for the transformation of CO2 to DMC using MeOH. The
[GLY(mim)3][NTf2]3 IL/DBU system was found to be an excellent
catalyst with 37% methanol conversion and 93% selectivity. In
these reactions, the tri-cationic IL forms an amidinium
carboxylate salt by chemical or physical adsorption of CO2 upon
reaction with DBU. Then, DMC is produced upon the reaction of
methanol with the amidinium carboxylate salt intermediate
with regeneration of the tri-cationic RTIL that can be reused.
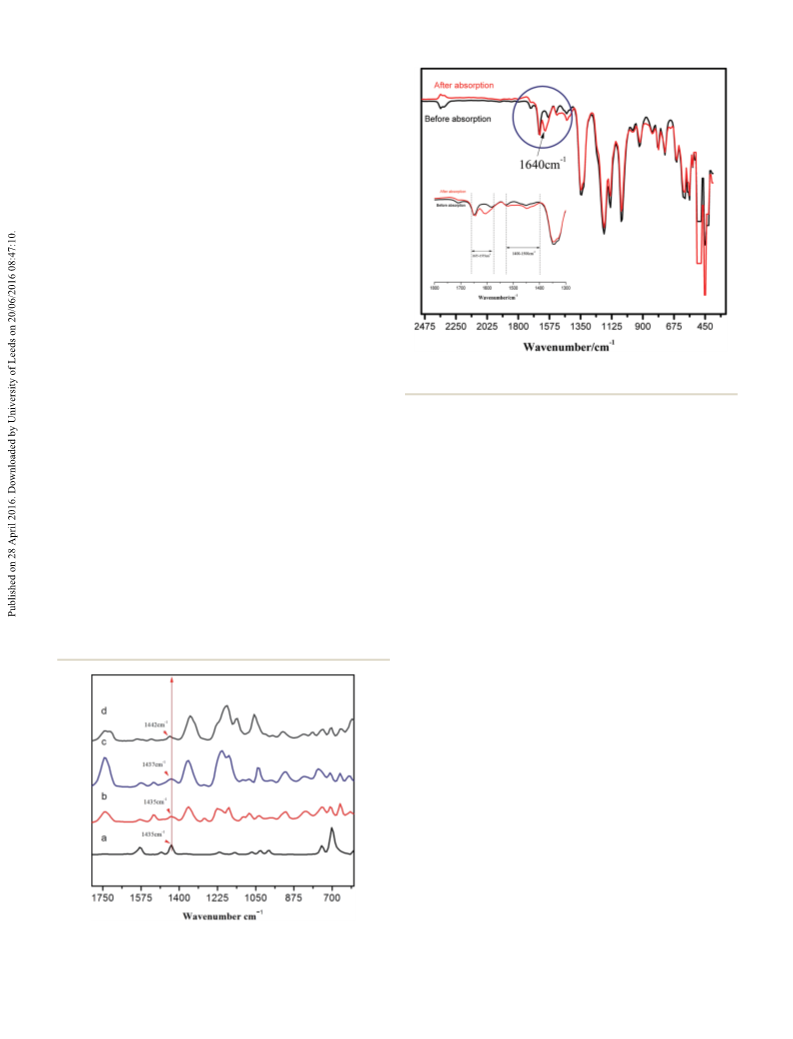
Using IR, 13C NMR, and XPS studies, it was conrmed that the
tri-cationic IL/DBU catalytic system chemically adsorbs CO2.
Furthermore, the mechanism of the reaction was proposed on
the basis of the 13C NMR study. We believe that this direct
synthesis route to produce DMC has great potential and the use
of an imidazolium containing tri-cationic RTIL/DBU catalytic
system as a reactive and renewable intermediate will open
a promising way to solve the many challenging chemical prob-
lems facing this reaction.
24 E. E. Benson, C. P. Kubiak, A. J. Sathrum and J. M. Smieja,
Chem. Soc. Rev., 2009, 38, 89–99.
25 J. E. Bara, C. J. Gabriel, S. Lessmann, T. K. Carlisle,
A. Finotello, D. L. Gin and R. D. Noble, Ind. Eng. Chem.
Res., 2007, 46, 5380–5386.
26 J. E. Bara, T. K. Carlisle, C. J. Gabriel, D. Camper,
A. Finotello, D. L. Gin and R. D. Noble, Ind. Eng. Chem.
Res., 2009, 48, 2739–2751.
Acknowledgements
This study was supported by the National Research Foundation
of Korea (NRF) – Grants funded by the Ministry of Science, ICT
and Future Planning (2014R1A2A2A01004352), Republic of
Korea.
27 J. E. Bara, D. E. Camper, D. L. Gin and R. D. Noble, Acc. Chem.
Res., 2010, 43, 152–159.
28 E. D. Bates, R. D. Mayton, I. Ntai and J. H. Davis, J. Am. Chem.
Soc., 2002, 124, 926–927.
29 H. Abimanyu, B. S. Ahn, C. S. Kim and K. S. Yoo, Ind. Eng.
Chem. Res., 2007, 46, 7936–7941.
References
1 Z. Z. Yang, Y. N. Zhao and L. N. He, RSC Adv., 2011, 1, 545–
567.
2 Z. Z. Yang and L. N. He, Beilstein J. Org. Chem., 2014, 10,
1959–1966.
3 Z. Xu, N. D. McNamara, G. T. Neumann, W. F. Schneider and
J. C. Hicks, ChemCatChem, 2013, 5, 1769–1771.
4 S. Wang and X. Wang, Angew. Chem., Int. Ed., 2015, 54, 2–15.
5 C. Wang, S. M. Mahurin, H. Luo, G. A. Baker, H. Li and S. Dai,
Green Chem., 2010, 12, 870–874.
30 A. A. Chaugule, H. A. Bandhal, A. H. Tamboli, W. J. Chung
and H. Kim, Catal. Commun., 2015, 75, 87–91.
31 Y. L. Yang and Y. Kou, Chem. Commun., 2004, 226–227.
32 D. J. Heldebrant, C. R. Yonker, P. G. Jessop and L. Phan,
Energy Environ. Sci., 2008, 1, 487–493.
33 N. Hollingsworth, S. F. R. Taylor, M. T. Galante,
J. Jacquemin, C. Longo, K. B. Holt, N. H. de Leeuw and
C. Hardacre, Angew. Chem., Int. Ed., 2015, 54, 14164–14168.
34 S. Huang, B. Yan, S. Wang and X. Ma, Chem. Soc. Rev., 2015,
44, 3079–3116.
6 P. Tundo and M. Selva, Acc. Chem. Res., 2002, 35, 706–716.
´
7 E. Torralba-Calleja, J. Skinner and D. Gutierrez-Tauste, J.
Chem., 2013, 2013, 16.
8 C. C. Tai, M. J. Huck, E. P. McKoon, T. Woo and P. G. Jessop,
J. Org. Chem., 2002, 67, 9070–9072.
9 A. T. Najafabadi, Int. J. Energy Res., 2013, 37, 485–499.
10 J. Sun, S. I. Fujita, F. Zhao and M. Arai, Green Chem., 2004, 6,
613–616.
35 S. Huang, B. Yan, S. Wang and X. Ma, J. Cheminf., 2012, 46,
44–52.
This journal is © The Royal Society of Chemistry 2016
RSC Adv., 2016, 6, 42279–42287 | 42287

 Chaugule, Avinash A.
Chaugule, Avinash A.
 Tamboli, Ashif H.
Tamboli, Ashif H.
 Kim, Hern
Kim, Hern