D.S. Doke, et al.
Applied Catalysis A, General 587 (2019) 117246
Pyruvic acid or esters can be synthesized via catalytic oxidative dehy-
drogenation of lactic acid/esters, e.g., ethyl lactate to ethyl pyruvate. In
this case, the whole process would be based on biomass-derived feed-
stock, and the reaction could in theory proceed directly from lactic acid
to pyruvic acid. As a possible candidate for the synthesis of pyruvic
acid, oxidative dehydrogenation of lactates to the corresponding pyr-
uvates in both gas and liquid phases on solid catalysts has received
significant attention. Oxidative dehydrogenation of ethyl lactate to
ethyl pyruvate either in liquid or gas phase using various binary oxides
2. Experimental section
2.1. Materials and instrumentation
Ethyl lactate, vanadyl acetylacetonate VO(acac)2, ethyl pyruvate
(Aldrich Chemicals), FeCl , copper acetate (Avra Chemicals India),
3
V
2
O , tert-butyl hydroperoxide (TBHP, 70% aqueous) (Avra Chemicals
5
India) and 5.5 M in decane (Aldrich Chemicals), acetonitrile (Finar
Chemicals India) were used as received without further purification.
containing molybdenum such as Fe
2
O
3
-MoO
3
,
TeO
2
-MoO
3
and
MoO
2
Cl
2
(DMSO)
2
and MoO
2
Cl
2
(DMF) were prepared using literature
2
MoVNbO , vanadium oxide species, and iron phosphate have been in-
x
method [18].
vestigated by various research groups [1,9,10]. These processes achieve
high yields of pyruvate from lactate. In the case of vapor phase dehy-
dration of lactates, besides the requirement for vaporizing lactate at
high temperature, a reaction temperature above 473 K is also needed,
which increases the running cost of the process. High reaction tem-
peratures may lead to product decomposition and then to low pro-
ductivity along with high energy demand. There are several reports on
liquid phase oxidative dehydrogenation (ODH) of lactates to pyruvates.
2
.2. Typical reaction procedure
In a typical ODH reaction, a two-necked round bottom flask (50 ml)
was charged with ethyl lactate (0.23 g, 2 mmol), 70% aqueous or or-
ganic (5.5 M in decane) TBHP (4 mmol), catalyst (0.04 mmol, 2 mol%),
and 6 g CH CN as a solvent. The reaction mixture was stirred at desired
3
temperature for specified time. The reaction was monitored by GC. The
samples were taken periodically and analyzed using a GC-Hewlett
Packard 6890 equipped with an HP-5 column (50 m length, 0.25 mm
internal diameter and 1 μm film thickness) and a flame ionization de-
tector (FID). Conversion of ethyl lactate was calculated based on GC-
FID results, where substrate conversion (%) = [moles of substrate re-
acted]/[Initial moles of substrate used] × 100, and selectivity (%) to
products was calculated by [total moles of the product formed]/[total
moles of substrate converted] × 100. The produced ethyl pyruvate was
quantified using external calibration curve plotted using commercial
ethyl pyruvate. Individual yields were calculated and normalized with
respect to the GC response factors. The product identification was
carried out by injecting commercial samples in the GC.
Kazuhiro et al. have reported VOCl catalyst in the presence of various
3
oxidants with almost 50% conversion and < 50% selectivity to pyr-
uvate. Water is the by-product of this reaction and the catalyst was not
stable in the presence of water. VOCl is known to be highly sensitive to
3
hydrolysis in presence of water/moisture leading to the formation of
V
2
O . Hence it is essential to remove water formed in the reaction to
5
maintain the activity of the catalyst [11]. Zhou and others have used
TS-1 as a catalyst and peroxide as an oxidant with excellent conversion
(
˜100%) and maximum 98.6% selectivity to pyruvate [12]. Shiju and
co-workers have reported titanium-based catalysts for ODH using
oxygen as an oxidant, but this reaction requires high pressure (1 MPa)
and temperature (130 °C). However, selectivity to pyruvate was an
issue: Only at lower conversions (˜20%) high selectivity (˜70%) to
pyruvate was reported; with an increase in conversion to 60%, pyruvate
selectivity dropped drastically (˜25%) due to polymerisation of pyr-
uvate [13]. In a recent work reported by Shiju et al. on the use of
mesoporous vanadia-titania catalysts for atmospheric pressure ODH at
3
. Results and discussion
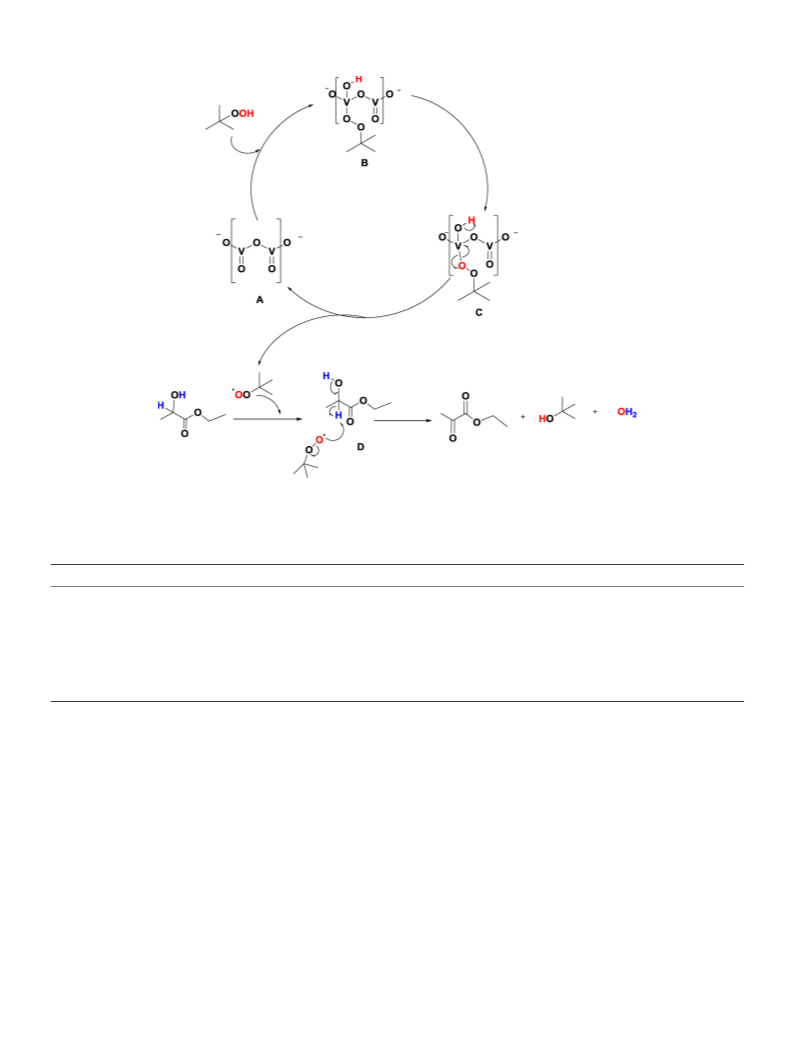
Oxidative dehydrogenation of ethyl lactate (Scheme 1) was carried
out using various commercial metal salts like vanadyl acetylacetonate,
copper acetate, iron chloride as well as simple complexes like
1
30 °C using diethyl succinate as a solvent, a pyruvate selectivity
of > 90% at ˜20% lactate conversion was observed. However, in this
case also the selectivity dropped to 60% at 70% conversion [14]. The
same group has again reported vapor phase ODH of ethyl lactate using
vanadia supported on different oxides as catalysts in a fixed bed reactor
at 180 °C with a maximum 48% yield of ethyl lactate [15]. The liquid
phase ODH of lactic acid using solid catalysts such as Pd/C modified by
Te and Pb has been reported by Hayashi et al. [16] with maximum 80%
conversion and 50% pyruvic acid yield at 90 °C. However, high cost of
precious metal is a matter of concern due to high Pd loading (5 wt.%)
essential for the reaction. Wang et al. have reported ethyl lactate ODH
using carbon nanotubes as catalysts and TBHP as an oxidant with
maximum yield of 40% at 90 °C [17]. Hence, development of a catalytic
system for efficient oxidative dehydrogenation of lactate to pyruvate
under mild reaction conditions using simple and inexpensive catalysts is
still highly desired.
MoO
2
Cl
2
(DMSO)
2
, MoO
2
Cl
2
(DMF) as catalysts in the presence of or-
2
ganic TBHP (5.5 M in decane) as an oxidant in acetonitrile as a solvent
at 80 °C. The results are reported in Table 1S. The acetonitrile solvent
was chosen based on our previous experience in oxidation reactions
using Mo-based complexes as homogeneous catalyst and H
2
2
O or TBHP
as oxidant, where better activity was obtained in acetonitrile compared
to other solvents.
Oxidative dehydrogenation of ethyl lactate without catalyst at 80 °C
using organic TBHP as an oxidant in acetonitrile solvent did not show
any conversion even after 11 h. Under the same conditions, various
Mo-, Cu-, and Fe-based catalysts yielded moderate to excellent con-
version of ethyl lactate (31–92%) with 100% selectivity to ethyl pyr-
uvate. It is interesting to note that in all cases ethyl pyruvate was
formed as a sole product and no other by-products were observed. Mo-
based complexes showed 92% ethyl lactate conversion with organic
TBHP; however, under identical conditions no reaction was observed
with aqueous TBHP, due to catalyst deactivation in the presence of
water. All the Mo complexes in presence of water formed blue com-
plexes which were found to be inactive for ODH reaction. It is well
documented in the literature that molybdenum blue are the complexes
with dimeric structure with very facile electron transfer between both
Vanadium-based catalysts have been extensively studied for selec-
tive oxidation of various organics including ethyl lactate. Various va-
nadium-based homogeneous catalysts including VOCl have been stu-
3
died for the oxidation of ethyl lactate to ethyl pyruvate using gas-liquid
micro flow system [11,14]. A maximum yield of 31% has been reported
at room temperature using oxygen as an oxidant. Molecular sieves were
essential to remove water generated during the reaction as a by-product
to avoid hydrolysis of the sensitive catalyst, VOCl .
3
Considering the importance of pyruvate and recognizing the pre-
vious efforts in this area, we have attempted oxidative dehydrogenation
of ethyl lactate to ethyl pyruvate using vanadium-based catalysts in the
liquid phase using t-butyl hydroperoxide as an oxidant. Interestingly,
very high conversion with nearly 100% selectivity to ethyl pyruvate
was observed, and the results are presented herein.
Scheme 1. Oxidative dehydrogenation of ethyl lactate to ethyl pyruvate.
2

 Doke, Dhananjay S.
Doke, Dhananjay S.
 Khomane, Sonali B.
Khomane, Sonali B.
 Pandhare, Swati L.
Pandhare, Swati L.
 Dongare, Mohan K.
Dongare, Mohan K.
 Dumeignil, Franck
Dumeignil, Franck
 Umbarkar, Shubhangi B.
Umbarkar, Shubhangi B.