ChemCatChem
10.1002/cctc.201600765
COMMUNICATION
spontaneously exchanges with formate (I
4
), forming the more
support a simple mechanism through a carbonite intermediate,
stable intermediate I
5
.
2
generated upon H evolution by reaction of formate with the
catalytic hydride. The mechanism is consistent with
thermodynamic parameters, obtained from the temperature
dependence analysis and with Raman spectroscopy of the
carbonite intermediate. The reported findings are particularly
relevant for industrial-scale generation of multi-carbon
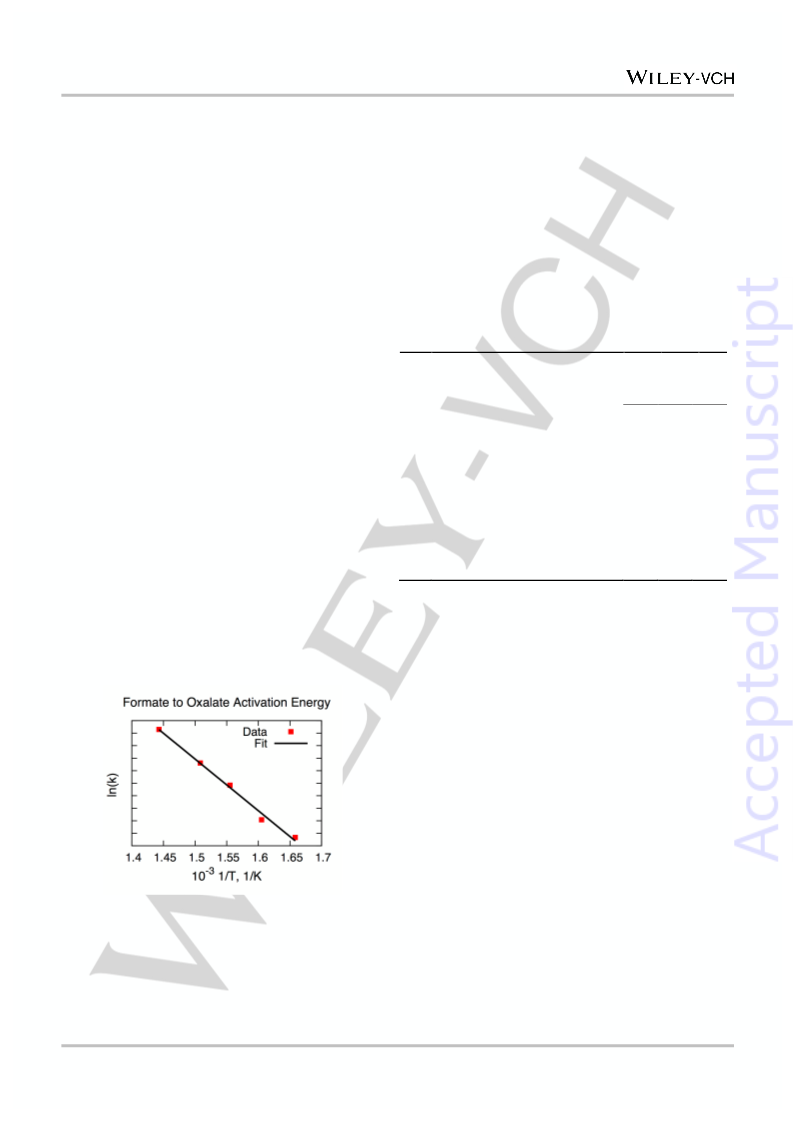
Our calculations of the complete thermodynamic cycle
Supporting Information, Table S1 and Table S2) show that I
TS is the rate-limiting step for oxalate formation (Table S2), with
(
2
→
1
-
1
a barrier of 41.0 kcal mol in excellent agreement with the
experimental value estimated from the observed temperature
dependence (Figure 2).
compounds from CO
2
since formate can be efficiently generated
by electrochemical reduction of CO
2
.
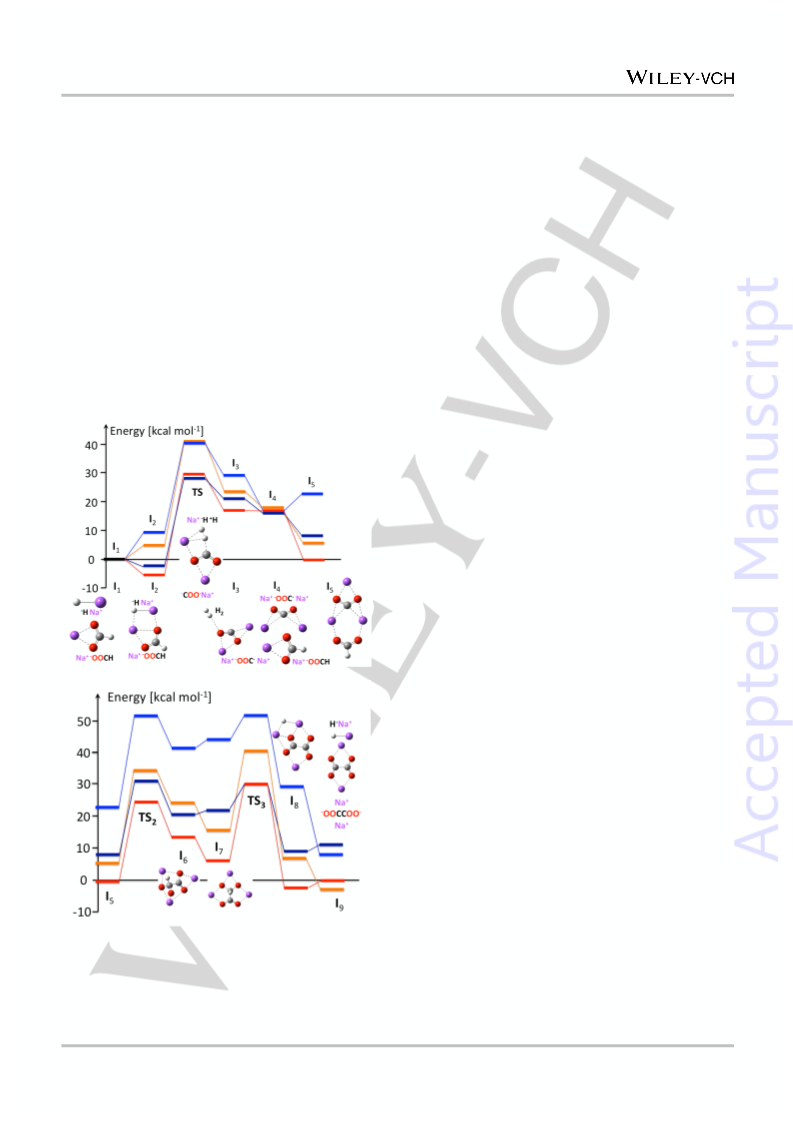
Figure 3 (top panel) shows that out-of-plane rotation of
Further, we are currently investigating the stability and
formate in I
transition state TS
OOCHCOO)Na complex (I
complex I through a 90 rotation of the O-C-C-O torsion angle
and a slight rearrangement of the Na counter ions. The C-H
bond is then elongated, forming NaH + Na (I ) through the
transition state TS . The separation of these electrostatically
bound species, upon precipitation of Na (s) forms I and
regenerates NaH for the next turn of the catalytic cycle.
5
, brings together the two carbon atoms, forming the
which forms the C-C bond in the
) before relaxing into the isomeric
13
reactivity of carbonite ions by Raman spectroscopy of C and
2
2
H enriched samples, and DFT studies. These studies could
(
3
6
have significant impact on the utilization of formate as a potential
carboxylating reagent via carbonite nucleophile.
o
7
+
2
C
2
O
4
8
3
Experimental Section
2
C
2
O
4
9
Experimental Details. The thermal reactions were explored by using a
Thermo Scientific Thermolyne Benchtop Muffle Furnace that could reach
a maximum temperature of 1200°C. Reactions were performed under a
2 2
flowing N atmosphere by introducing N gas through a vent port since
oxygen lowers the yield of oxalate formed. A series experiments were
designed using reaction temperature, reaction time and the amount of
catalyst as the reaction condition variables to obtain the best possible
yields. A typical bench-scale reaction was conducted using a 4.0 g.
formate sample placed into a 50 mL nickel crucible and calcined between
3
00 - 480°C. All chemicals were reagent grade obtained from Sigma
Aldrich including NaOH, sodium hydride, KOH, sodium and potassium
formate, sodium and potassium oxalate, sulfuric acid, and potassium
permanganate (J. T. Baker). The catalyst (e.g., NaH, NaOH, KOH) in
weighed amounts of 2.5% by mass were mixed thoroughly using a
mortar and pestle in a nitrogen glove box. The quantitative analysis of
oxalate formation was performed by volumetric titrations using
[
22,23]
standardized KMnO
4
solutions
as well as by ion chromatography
methods for the analysis of formate and oxalate.
Acknowledgements
Georgian Court gratefully acknowledges funding from Liquid
Light, Inc. for the undergraduate students who helped conduct
the experimental work for this paper. V.S.B. acknowledges the
AFOSR grant FA9550-13-1-0020 and high performance
computing facilities from NERSC and Yale University.
Keywords: Formate to Oxalate Conversion
• Carbon Chain
Generation • CO Reduction • Density Functional Theory
2
References
1
2
. M. Aresta, A. Dibenedetto, J. Chem. Soc. Dalton Transactions, 2007,
8, 2975-2992.; 2. E. J. Quadrelli, G. Centi, J. Duplan, S. Perathoner,
ChemSusChem, 2011, 4, 1194-1215.; 3. A. Paparo, J. S. Silvia, E. C.
Kefalidis, T. P. Spaniol, L. Maron, J. Okuda, C. C. Cummins, Angew.
Chem. 2015, 54, 9115-9119.; 4. J. Andrez, J. Pacaut, P. Bayle, M.
Mazzanti, Angew. Chem. 2014, 53, 10448-10452.; 5. X. Min, M. W.
Kanan, J. Am. Chem. Soc., 2015, 137, 4701-4708.; 6. J-P. Jones, G. K.
S. Prakash, G. A. Olah, Israel Journal of Chemistry, 2014, 54, 1451-
1
2
466.; 7. J. D. Watkins, A. B. Bocarsly, ChemSusChem, 2014, 7, 284-
90.; 8. D. Kopljar, A. Inan, P. Vindayar, N. Wagner, E. Klemm, Journal
of Applied Electrochemistry, 2014, 44, 1107-1116.; 9. R. Angamuthu, P.
Byers, M. Lutz, A. L. Spek, L. Anthony, E. Bouwman, Science, 2010, 327,
3
13-315.; 10. N. Hollingsworth, B. K. Holt, N. H. de Leeuw, R. S. F.
Figure 3. Free energy profile of catalytic conversion of formate
to oxalate as salts of sodium (red 663 K, orange 298 K) and
potassium (navy 713 K, blue 298 K), computed at the DFT
B3LYP level of theory (SI).
Taylor, J. Jocquemin, C. Hardaacre, M. T. Galante, C. Longo, Angew.
Chem. 2015, in press.; 11. Kaczur, J., Kramer, T., Keyshar, K., Majsztrik,
P. and Twardowski, Z., “Process and High Surface Area Electrodes for
the Electrochemical Reduction of Carbon Dioxide”, 14 Oct 2014, Patent:
US 8,858,777.; 12. Kaczur, J., Kramer, T., Keyshar, K., Majsztrik, P. and
Twardowski, Z., “System and Process and High Surface Area Electrodes
for the Electrochemical Reduction of Carbon Dioxide”, 2 May 2013,
Patent: US 2013/0105304.; 13. Kaczur, J., Teamey, K., “Integrated
Process for Producing Carboxylic Acids from Carbon Dioxide”, 21 Jul
Concluding Remarks: We found that quantitative conversion of
formate into oxalate can be achieved by simple calcination of
molten formate salts in the presence of NaH. DFT calculations
This article is protected by copyright. All rights reserved.

 Lakkaraju, Prasad S.
Lakkaraju, Prasad S.
 Askerka, Mikhail
Askerka, Mikhail
 Beyer, Heidie
Beyer, Heidie
 Ryan, Charles T.
Ryan, Charles T.
 Dobbins, Tabbetha
Dobbins, Tabbetha
 Bennett, Christopher
Bennett, Christopher
 Kaczur, Jerry J.
Kaczur, Jerry J.
 Batista, Victor S.
Batista, Victor S.