Ding et al.
Selective Reduction of Nitro Group in Aryl Halides Catalyzed by Silver Nanoparticles Modified with ꢀ-CD
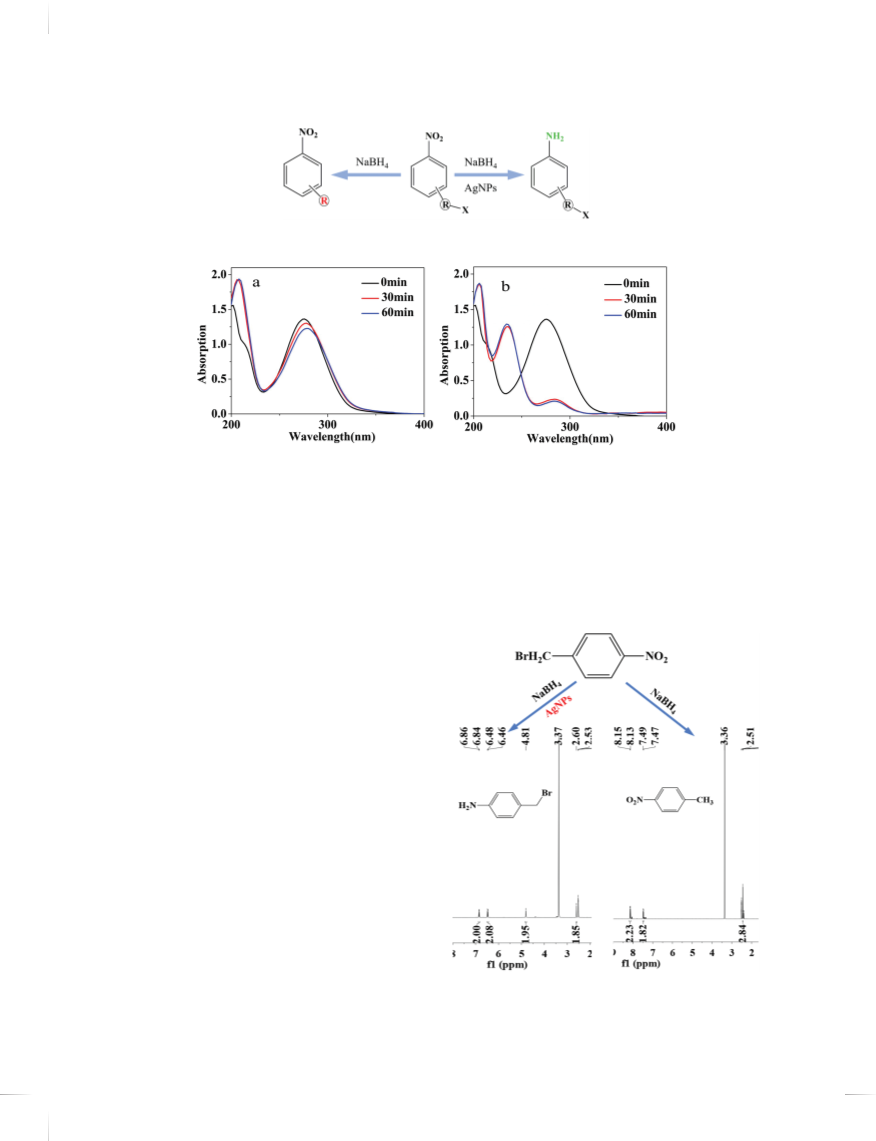
Figure 7. UV-vis spectra of reduction process of 2-nitrobenzyl bromide and 4-nitrophenylethyl bromide catalyzed by AgNPs modified with ꢀ-CD.
of the system catalyzed by AgNPs modified with ꢀ-CD
was located at 283 nm, whereas that catalyzed by AgNPs
prepared by sodium citrate shifted to 288 nm accompa-
nied with the increase in absorption intensity. The phe-
nomenon may be due to the simultaneous reduction of
methyl bromide to methyl and then the push electron
effect of methyl promotes the red shift of B band. This
claim proposed by us was further confirmed by H NMR
spectra.
Finally, in order to investigate the suitable range of
selective catalysis of the AgNPs modified with ꢀ-CD,
References and Notes
1. V. V. Annenkov, O. N. Verkhozina, T. A. Shishlyannikova, and E. N.
Danilovtseva, Anal. Biochem. 486, 5 (2015).
2
3
. M. S. Khan, A. Hanan, and A. Majeed, J. Chem. Soc. 10, 393 (1988).
. R. S. Downing, P. J. Kunkeler, and H. Vanbekkum, Catal. Today
37, 121 (1997).
4. P. Ji, J. H. Atherton, and M. I. Page, J. Org. Chem. 77, 7471 (2012).
5. C. E. Hendrick and Q. Wang, J. Org. Chem. 80, 1059 (2015).
1
6
7
8
. D. Herrmannsdörfer, M. Kaaz, O. Puntigam, J. Bender, M. Nieger,
and D. Gudat, Eur. J. Inorg. Chem. 2015, 4819 (2015).
. M. R. Naffziger, B. O. Ashburn, J. R. Perkins, and R. G. Carter,
J. Org. Chem. 72, 9857 (2007).
. W. Xu, J. J. Pignatello, and W. A. Mitch, Sci. Technol. 49, 3419
(2015).
4
-nitrophenylethyl bromide and 2-nitrobenzyl bromide
were chose to conduct the same reaction. The UV-vis spec-
9. F. Li, B. Frett, and H. Li, Synlett 25, 1403 (2014).
1
0. T. T. Li, J. J. Yang, Z. Ali, Z. L. Wang, X. B. Mou, N. Y. He, and
tra of reduction process of the t IwP o: 1c 0o 9m . 9p o4 u. 1n 7d 4s . r9 e 4fl eOc nt e:dMon, 17 Dec 2018 18:34:32
Z. F. Wang, Sci. China Chem. 60, 370 (2017).
Copyright: American Scientific Publishers
the same selective catalytic features as shown in Figure 7.
1
1. N. Kristian, Y. Yan, and X. Wang, Chem. Commun. 3, 353 (2008).
1
Delivered by Ingenta
Similarly, H NMR spectra were used to verify that
the halogenated alkyl group was protected by ꢀ-CD in
the reduction process. Therefore, we can conclude that the
position of halogenated alkyl group on the aromatic ring
and the length of the halogenated alkyl group have little
effect on the selective catalysis of AgNPs modified with
1
2. A. Aijaz, A. Karkamkar, Y. J. Choi, N. Tsumori, E. Rönnebro,
T. Autrey, H. Shioyama, and Q. Xu, J. Am. Chem. Soc. 134, 13926
(2012).
1
1
3. S. K. Hanson and R. T. Baker, Acc. Chem. Res. 48, 2037 (2015).
4. X. Hong, D. Wang, S. Cai, H. Rong, and Y. Li, J. Am. Chem. Soc.
134, 18165 (2012).
1
1
5. S. N. Mohammad, J. Mater. Chem. 22, 21560 (2012).
6. R. W. J. Scott, O. M. Wilson, and R. M. Crooks, Chem. Mater.
16, 5682 (2004).
ꢀ
-CD. Without doubt, the results of this study can greatly
enhance the application value of AgNPs modified with
ꢀ
-CD in the synthesis of halogenated aromatic amines.
17. Q. Xiao, Z. Liu, F. Wang, S. Sarina, and H. Y. Zhu, Appl. Catal. B
09, 69 (2017).
2
18. M. J. Climent, A. Corma, J. C. Hernández, A. B. Hungría, S. Iborra,
and S. Martínez-Silvestre, J. Catal. 292, 118 (2012).
4
. CONCLUSION
1
9. J. Macanás, L. Ouyang, M. L. Bruening, M. Muñoz, J. C. Remigy,
and J. F. Lahitte, Catal. Today 156, 181 (2010).
In summary, we first report a selective reduction of nitro
group in aryl halides catalyzed by silver nanoparticles
modified with ꢀ-CD. Taking the advantage of hydropho-
bic lumen and donor–acceptor behavior of ꢀ-CD, the
halogenated alkyl groups on the aromatic ring can be
enveloped in the inner cavity that thereby inhibits the
2
2
0. G. Budroni and A. Corma, J. Catal. 257, 403 (2008).
1. S. Gladiali and E. Alberico, Chem. Soc. Rev. 35, 226 (2006).
22. W. Cui, Q. Xiao, S. Sarina, W. Ao, M. Xie, H. Zhu, and Z. Bao,
Catal. Today 235, 152 (2014).
23. J. S. M. Samec, J. Bäckvall, P. G. Andersson, and P. Brandt, Chem.
Soc. Rev. 35, 237 (2006).
1
reduction of the halogen. UV-vis spectra and H NMR
24. W. Zhang, G. Lu, C. Cui, Y. Liu, S. Li, W. Yan, C. Xing, Y. Chi,
Y. Yang, and F. Huo, Adv. Mater. 26, 4056 (2014).
spectra were applied to verify the mechanism proposed
by us. This study will open a new strategy for the synthe-
sis of halogenated aromatic amines, and provides an idea
for fabrication of novel selective catalyst.
25. Y. Chen, J. Qiu, X. Wang, and J. Xiu, J. Catal. 242, 227 (2006).
26. A. Noschese, A. Buonerba, P. Canton, S. Milione, C. Capacchione,
and A. Grassi, J. Catal. 340, 30 (2016).
27. H. Fu, L. Zhang, Y. Wang, S. Chen, and Y. Wan, J. Catal. 344, 313
2016).
(
Acknowledgments: This work was supported by the
open project of the state key laboratory of bioelectronics
of southeast university (Grant No. 20170118).
2
8. R. K. Rai, A. Mahata, S. Mukhopadhyay, S. Gupta, P. Li, K. T.
Nguyen, Y. Zhao, B. Pathak, and S. K. Singh, Inorg. Chem. 53, 2904
(2014).
J. Nanosci. Nanotechnol. 18, 8201–8206, 2018
8205

 Ding, Xu
Ding, Xu
 Zhu, Geng
Zhu, Geng
 Guan, Jian-Ning
Guan, Jian-Ning
 Han, Guo-Zhi
Han, Guo-Zhi