G Model
CATTOD-9663; No. of Pages8
ARTICLE IN PRESS
2
J. Zhao et al. / Catalysis Today xxx (2015) xxx–xxx
Solid acid catalysts can overcome the drawbacks of homoge-
Furthermore, solid acid catalysts are capable of tuning the sur-
face acidity and working at harsh reaction conditions [19]. The
achieved over de-aluminated H-form mordenite at 165 ◦C in a
solvent consisting of water and MIBK [20,21]. In the presence
of vanadylphosphate (VOP), a 40% yield of HMF was obtained
in a 6 wt.% aqueous fructose solution [22]. Introducing different
yield and selectivity increased to 50% and 87%, respectively over a
Fe-containing VOP catalyst. Nb-based catalysts such as Nb2O5, nio-
also exhibited high catalytic activity [23–26]. Zr- and Ti-based cat-
fructose, HMF yield of 47% was obtained within 4 h at 130 ◦C over
SO42−/ZrO2 Al2O3 with Zr Al molar ratio of 1:1 [27]. Ion-exchange
The carbon-based solid acids, possessing high stability, low cost
and abundant strong protonic acid sites on surfaces, have been
tions [29]. In particular, carbon sphere (CS) solid acid catalysts can
be prepared by direct sulfonation of CS generated from various
resins, activated carbon, bio-char and lignin [30–32]. In the typical
synthesis of CS, glucose, sucrose, fructose or cellulose was heated to
400–600 ◦C under N2 flow to produce black powder. The obtained
black powder was then heated in concentrated sulfuric acid or fum-
ing sulfuric acid at 150–200 ◦C [33–35]. In addition to SO3H groups
on CS surfaces, there were also Ph OH and COOH functionalities,
compared to the sulfonated amorphous glassy carbon, activated
carbon and natural graphite [36,37], due to the compact carbon
structure of these precursors and the lack of functional groups, par-
ticularly acid sites on the surfaces [36,38]. In this work, a modified
preparation of carbon-based solid acid under mild conditions was
developed. The CS was prepared by hydrothermal carbonization of
glucose at 180 ◦C which was remarkably lower than the tempera-
ture in other CS synthetic routes. The resulted CS was sulfonated
by sulfuric acid aqueous solutions instead of concentrated H2SO4
or fuming sulfuric acid. Catalytic results showed that the catalysts
afforded high activity for the dehydration of fructose to HMF. Under
an optimized condition, fructose was converted into HMF with 90%
yield at 160 ◦C after reaction duration of 1.5 h.
filtered, washed and then dried following the same procedures in
CS preparation. Sulfuric acid solutions with different concentra-
CS solid acid catalysts were labeled as CS-1, CS-2, CS-3 and CS-4
according to the sulfuric acid and water volumetric ratios of 1:5, 1:2,
1:1 and 2:1, respectively. For comparison purposes, sulfonated CS
was also prepared in one-step following the procedures as reported
by Liu et al. [40]. Equal amounts of glucose dissolved in sulfuric
acid solutions of different sulfuric acid and water ratios (1:5, 1:2,
1:1, 2:1); the solutions were placed in teflon-sealed autoclaves and
heated at 180 ◦C for 4 h. The products were labeled as C-1, C-2, C-3
and C-4.
2.2. Catalysts characterization
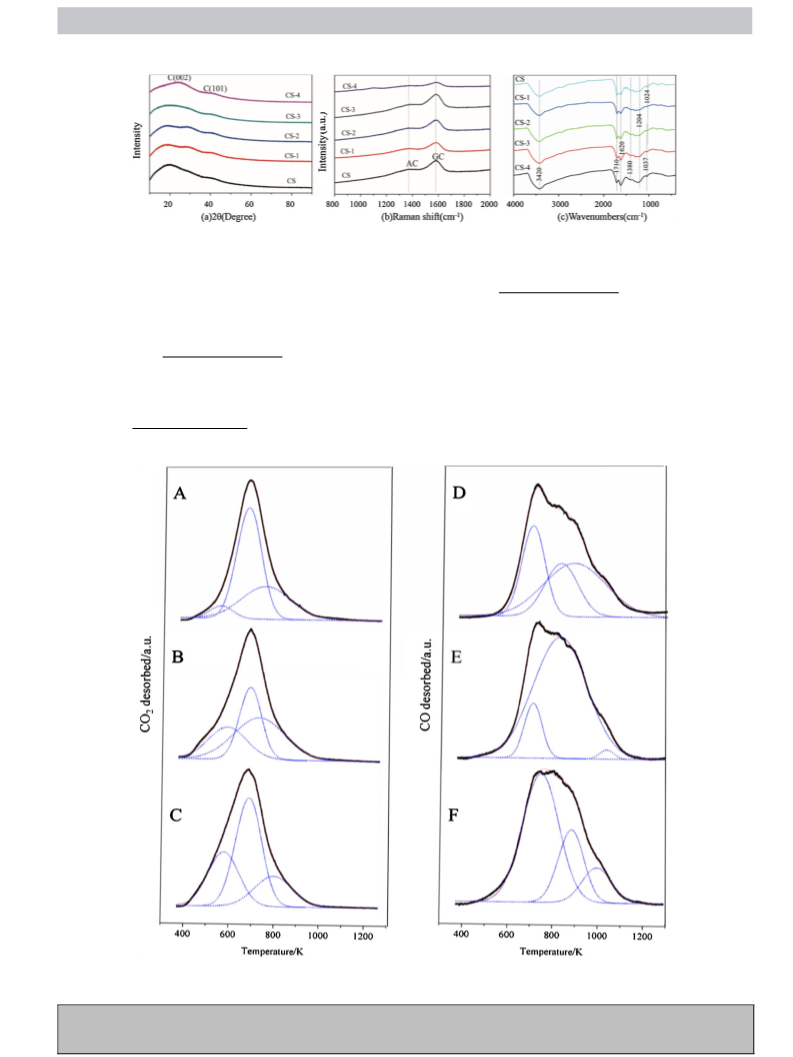
Fourier transform infrared (FTIR) spectra were obtained on
Digilab FTS 3100 FTIR with a 4 cm−1 resolution in the range
of 400–4000 cm−1 using a standard KBr disk technique. Raman
tests were carried out on a Renishaw 1000 Raman spectrome-
ter equipped with a 514 nm excitation from HeNe laser, of which
the detection depth is about 150 nm. The BET surface areas were
measured on Autosorb-6B (Quantachrome instruments) using the
liquid nitrogen adsorption method. Powder X-ray diffraction (XRD)
patterns were recorded on a Bruker Advance 8 X-ray diffractome-
ter using a Ni filtered Cu K␣ radiation (ꢀ = 0.154 nm), operated at
40 kV and 40 mA. XRD data were collected between 10 and 90◦ (2ꢁ)
with a resolution of 0.02◦ (2ꢁ). The morphology and structure of the
spheres were investigated using a field emission scanning electron
microscope (FESEM, JOEL JSM 6701F). Temperature-programmed
desorption (TPD) was performed on a Micromeritics AutoChem II
2920 instrument. Typically, the pretreatment of the sample was
conducted in a quartz reactor by high-purity Ar at 200 ◦C for 2 h.
Then after the temperature cooled down to 100 ◦C, it was set to
rise to 1020 ◦C at a rate of 5 ◦C/min. A mass spectrometer (Ther-
moStar GSD 301 T2) was used to detect the desorbed CO and CO2.
In addition, 1H-NMR spectra were measured to analysis the sep-
arated product on a Bruker Avance II 300 MHz spectrometer with
CDCl3 as the solvent. The element composition of the catalyst was
estimated by Vario EL III CHNS Elemental Analyser.
The total number of acid sites on the catalyst surfaces were
determined by a well-established acid-base back neutralization
titration method [41]. The CS catalyst (30 mg) was suspended in
a sodium hydroxide aqueous solution (0.1 M, 20 ml). The mixture
was stirred using a magnetic stirrer for 4 h, followed by ultrasonica-
tion for 1 h at room temperature. The concentration of the OH− ions
in the supernatant solution was calibrated by oxalic acid standard
solution. The content of SO3H groups on the surfaces of the CSs
were determined by neutralization titration with sodium hydrox-
ide. A sodium chloride aqueous solution (0.2 M, 20 ml) was added to
the catalyst (30 mg). The mixture was stirred using a magnetic stir-
rer for 4 h, followed by ultrasonication for 1 h at room temperature.
The supernatant solution was titrated using a sodium hydroxide
(0.01 M) solution.
2. Experimental
2.1. Catalyst preparation
Fructose (≥99% purity), HMF (99% purity), Glucose (≥99%
purity), sulfuric acid (98% purity) and all the solvents were obtained
from Sigma-Aldrich. These commercial chemicals were used as
received without further purification. The CS was prepared by
hydrothermal carbonization of glucose [39]. In the typical synthe-
sis, 5 g of glucose was dissolved in 30 ml of deionized water to form a
clear solution under stirring. The solution was then transferred into
a 40 ml capacity teflon-lined autoclave and maintained at 180 ◦C for
10 h. The resulting solid products were filtered and washed with
deionized water and ethyl alcohol, followed by vacuum drying for
4 h at 60 ◦C to afford CS.
2.3. Catalytic reactions
The catalytic reaction was performed in a 25 ml capacity flask
equipped with a condenser. In a typical catalytic run, a DMSO solu-
tion of fructose (0.5 g of fructose in 10 ml of DMSO) was added into
the flask along with 100 mg of CS solid acid catalysts. The reac-
tion mixture was heated to the reaction temperature in oil bath
with a thermostat and an electronically controlled magnetic stir-
rer. The reaction samples were removed periodically and analyzed.
The reaction products were diluted in water and filtered through a
syringe filter (VWR, 0.22 um PTFE) prior to analysis by liquid chro-
matography (HPLC, Agilent 1100 series, Bio-Rad Aminex HPX-87H,
The as-synthesized CS was dispersed in a sulfuric acid solution
under stirring. The suspension was placed in a 40 ml teflon-sealed
autoclave and maintained at 180 ◦C for 4 h. The black products were
Please cite this article in press as: J. Zhao, et al., Efficient dehydration of fructose to 5-hydroxymethylfurfural over sulfonated carbon

 Zhao, Jun
Zhao, Jun
 Zhou, Chunmei
Zhou, Chunmei
 He, Chao
He, Chao
 Dai, Yihu
Dai, Yihu
 Jia, Xinli
Jia, Xinli
 Yang, Yanhui
Yang, Yanhui