306
LEIVA ET AL.
initial thermolysis. This means that the corresponding
activation enthalpies of the reaction (ꢀH , Table II)
ing the diradical generation, facilitating further C O
cleavages.
#
are almost compensated by the entropies of activation.
All these findings suggest that the ACDP thermoly-
sis conforms a genuine reaction series [6,18] where
the solvent affects the tetroxacyclohexane ring O O
bond rupture of their molecule. Therefore, these val-
ues are in agreement with a stepwise reaction mecha-
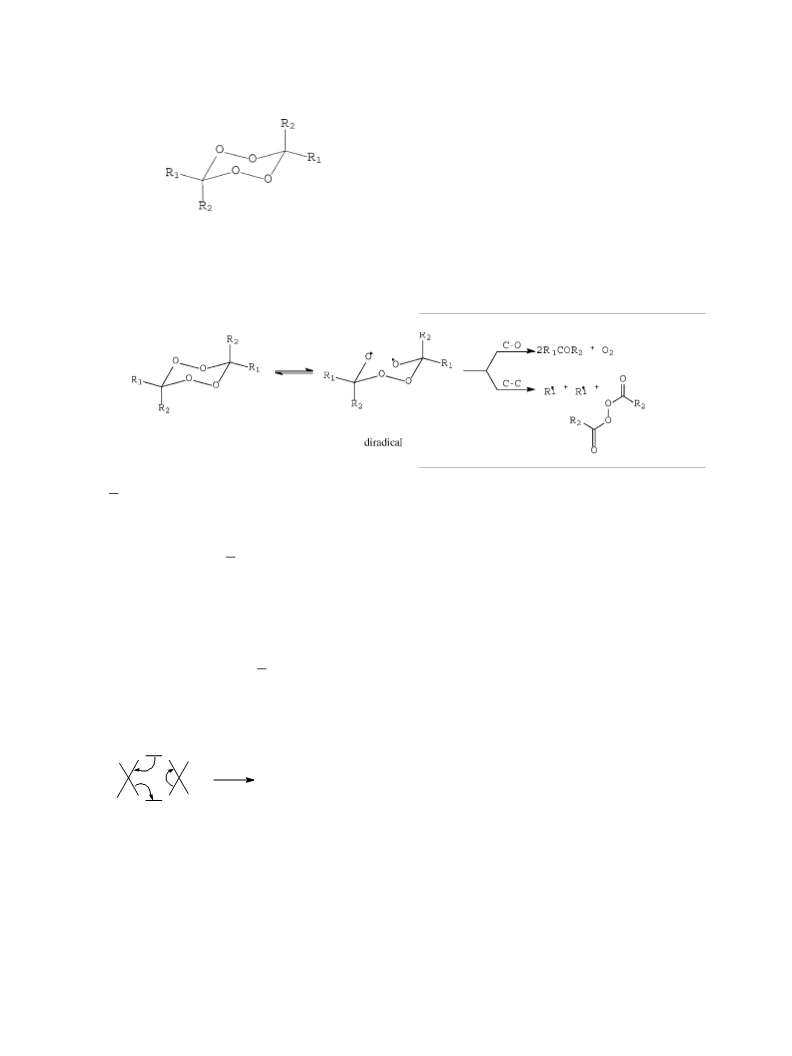
nism with homolytic rupture of one peroxidic bond of
ACDP molecule given a diradical, as it is observed in
other analogous ACDP reactions [6,8,9].
However a large variation in the enthalpies and en-
tropies of activation and the rate constant values can be
observedincomparingACDPthermolysisinMTBEre-
spect to other oxygenated solvents already investigated
CONCLUSIONS
1
2
3
. The thermolysis of ACDP in MTBE solution fol-
lows first-order kinetic law up to at least 60%
diperoxide conversion.
. Under the experimental conditions, the activation
parameters correspond to the unimolecular ther-
mal decomposition reaction of ACDP molecule.
. Analysis of the reaction products and the acti-
vation parameter values contributes to postulate
the mechanism for the thermolysis of the ACDP
in MTBE solution. The thermolyses would occur
through a common mechanism of decomposition
already advanced for the tetroxanes, which be-
gins with the homolytic rupture of the peroxydic
bond leading to the formation of an intermediate
diradical and further C O bond ruptures giving
acetone and oxygen as final products.
(methanol, THF, and 2-propanol), while small varia-
tion can be observed in the hydrocarbons (n-octane,
benzene). All of these findings suggest that the low po-
larity of MTBE determines the low rate found in the
ACDP thermolysis reaction.
Quantitative analysis of the acetone produced in the
thermolysis (Fig. 2) shows a molar yield practically
of 2 moles of carbonylic product for mole of ACDP
decomposed, which is not dependent on the temper-
ature and initial concentration of the tetroxane. Since
the concerted type decomposition could be discarded,
these results suggests that the formation of the prod-
ucts of the thermolysis can be interpreted in terms of an
initial O O homolysis to give the diradical [Eq. (1)],
which can rebuild the ACDP molecules undergoing
C O cleavages to give exclusively acetone plus oxy-
gen [Eq. (1)]. Thus, this suggests thatsolventmolecules
participate in the “reaction cage solvent” formed dur-
4
. ThekineticsofACDPthermolysisissignificantly
affected by the physicochemical characteristics
of the reaction media. Probably, the low polar-
ity of the MTBE solvent determinates the low
decomposition rates observed.
BIBLIOGRAPHY
1. Jorge, N. L. Theoretical conformationals studies, elec-
tronic structures and chemical reactivity of cyclic per-
oxides; Doctoral thesis, 1997, UNLP, Argentina.
2. Jorge, N. L.; G o´ mez Vara, M. E.; Castro, E. A.; Autino,
J. C.; Cafferata, L. F. R. J Mol Struct (Theochem) 1999,
4
59, 29.
3
4
. Jorge, N. L.; Peruchena, N.; Castro, E. A.; Cafferata, L.
F. R. J Mol Struct (Theochem) 1988, 433, 311.
. Cafferata, L. F. R.; Furlong, J. J. In Advances in Oxy-
genated Processes; Baumstark, A. L. (Ed.); JAI Press:
Greenwich, CT, 1995; Vol. 4, p. 81.
5
6
7
8
. Cafferata, L. F. R.; Eyler, G. N.; Mir ´ı fico, M. V. J Org
Chem 1984, 49, 2107.
. Cafferata, L. F. R.; Eyler, G. N.; Svartman, E. L.; Ca n˜ izo,
A. I.; Alvarez, E. E. J Org Chem 1991, 56, 411.
. Ca n˜ izo, A. I.; Cafferata, L. F. R. An Asoc Quim Ar-
gentina 1992, 80(4), 345.
. Leiva, L. C.; Castellanos, M. G.; Jorge, N. L.; Cafferata,
L. F. R.; G o´ mez Vara, M. E. Revista de la Sociedad
Qu ´ı mica de M e´ xico 1998, 42, 223.
Figure 2 Acetone molar yields (moles of acetone per mole
9. Leiva, L. C.; Cafferata, L. F. R.; G o´ mez Vara, M. E. An
Asoc Qu ´ı m Argentina 2000, 88(1/2), 9.
10. Reichardt, C. Solvent Effects in Organic Chemistry; Ver-
lag Chemie: New York, 1979.
of ACDP decomposed) of acetone diperoxide in MTBE solu-
◦
◦
◦
tion at different temperatures. 130 C (◦), 140 C (ꢀ), 150 C
◦
(
ꢁ), and 166 C (×).

 Leiva, Laura C.
Leiva, Laura C.
 Jorge, Nelly L.
Jorge, Nelly L.
 Romero, Jorge M.
Romero, Jorge M.
 Cafferata, Lazaro F.R.
Cafferata, Lazaro F.R.
 Gomez Vara, Manuel E.
Gomez Vara, Manuel E.