F. Lu et al. / Molecular Catalysis 444 (2018) 22–33
23
tions. The Zr-doped CeO2 has recently been used as an efficient
ketonization catalyst. The activity enhancement is likely due to the
solid solution with abundant Lewis acid-base M-O (M = Ce, Zr) sites
microscopy (TEM) study was performed on Tecnai F20 instrument
operating at 120 kV and 200 kV. The BET surface area, pore vol-
ume, and pore size distribution measurements were performed
using a Quantachrome Autosorb-iQ instrument. Raman spectra
were obtained on an ultraviolet high-resolution Raman spectrom-
eter with a He-Cd laser of excitation wavelength 514.5 nm, which
and oxygen vacancies. The introduction of ZrO could also result in
2
a lower reduction temperature of the bulk ceria and thus enhanced
redox properties [22,23]. Actually, it is generally believed that the
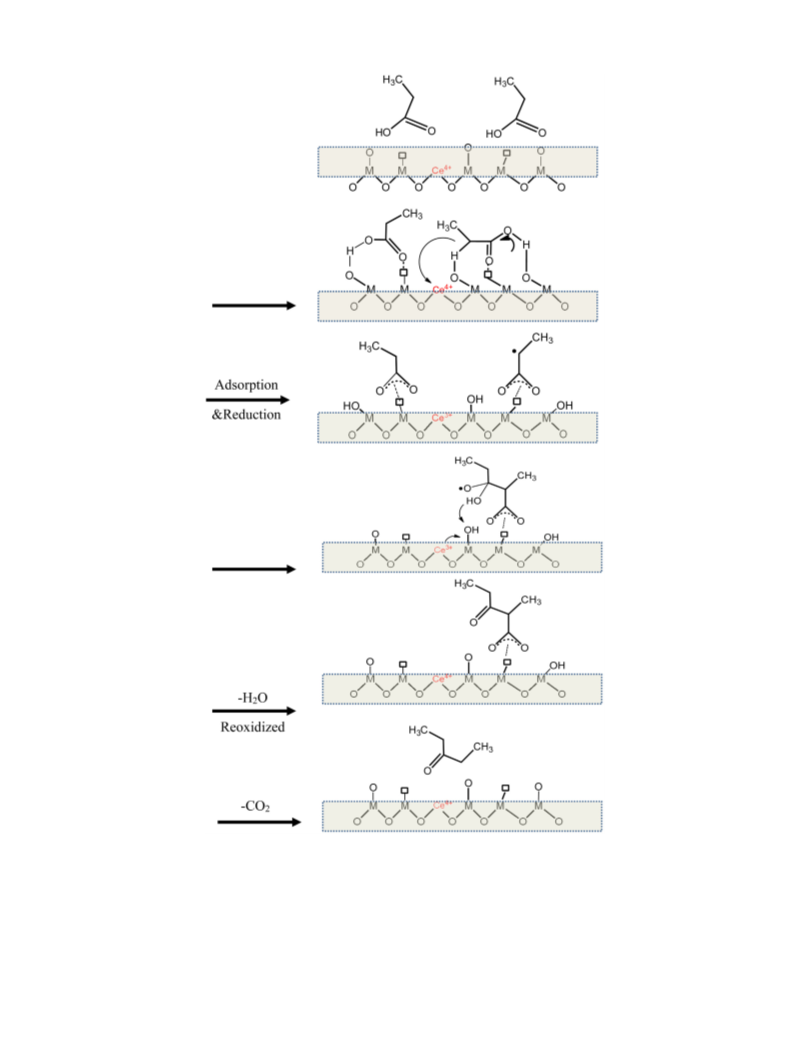
surface ketonization mechanism involves consecutive reduction-
oxidation cycles of the amphoteric oxides [7,24]. Hence, the catalyst
redox properties are assumed to be critical to the improvement
of ketonization activity. Despite the above reports, the underlying
reasons of the promotional effect of doped CeO2 catalysts espe-
cially for the role of solid solution were not well understood so
far. Furthermore, the redox cycle during the ketonziation reaction
has not been verified experimentally. Whether the redox proper-
−
1
consisted of two accumulations of 30 s with a resolution of 2 cm
.
Temperature-programmed reductions by hydrogen (H -TPR)
2
were recorded using a thermal conductivity detector and a reduc-
tion gas of 10 vol.% H2 in Ar, set at a flow rate of 40 mL/min.
◦
Approximately 50 mg samples were pre-treated at 450 C for
60 min in pure He, then the sample cell was purged with H /Ar
2
◦
◦
mixture for 30 min and the analysis ran from 50 C to 900 C at a
heating rate of 10 C/min. Water produced by the reduction was
◦
ties of CeO -like amphoteric oxides are key in maintaining the high
ketonization activity is still not very clear.
trapped in an ice bath upstream of the TCD detector. The sample
after a TPR run was reoxidized and then a second TPR run was con-
ducted in order to reveal the redox properties of the sample. The
2
Giving the issues raised above, the current work focused on
exploring the structure-activity relationship of mixed oxides and
the role of solid solution during the surface ketonization process.
To achieve these purposes, we prepared a series of Ce-Fe solid
solutions and then the as-prepared catalysts were applied in the
vapor-phase ketonization reaction of carboxylic acid. As is well
known, Fe-modified ceria systems present a remarkable improve-
ment of their oxygen exchange abilities as compared with the pure
◦
first run was performed up to 900 C, and then the sample was
◦
cooled to a desired temperature (100, 300, 500 C). Thereafter, the
◦
reduced sample was exposed to air at 100, 300, 500 C for 30 min.
◦
Then, the sample was cooled down to 50 C in air for the second
TPR experiment.
The number and the strength of the basic sites of the samples
were obtained from temperature-programmed desorption (TPD) of
CeO , which is due to the strong interaction of Fe and Ce species and
CO pre-adsorbed at room temperature on a Quantachrome Instru-
2
2
the formation of solid solution with different physical and chemi-
cal properties [25]. Ce-Fe mixed oxides as heterogeneous catalysts
for ketonization reaction can be easily separated from the products
and used in continuous-flow fixed-bed process, which is suitable
for large-scale industrial application [26,27]. Herein, the catalytic
performances of the as-prepared samples were correlated with the
observed structural evolution of the catalysts. Besides, the spent
catalysts were carefully characterized and analyzed. Our results
can furnish fresh perspective on the development of high-efficient
heterogeneous catalysts for vapor-phase ketonization reaction and
provide new grounds for the following fundamental and applied
ment. Approximately 100 mg samples were outgassed and dried at
450 C for 30 min under 40 mL/min He flow, cooled to room temper-
◦
ature, and then treated with a flow of 10% vol.%CO /Ar (40 mL/min)
2
for 120 min. The sample cell was then evacuated for 15 min and
purged with He (40 mL/min) for 60 min to remove the physisorbed
◦
CO . The sample was then heated from room temperature to 500 C
2
◦
at 10 C/min. The resulting CO2 peak was quantified by calculating
the areas under the curve on the basis of the known amounts of
CO2 using thermal conductivity.
X-ray photoelectron spectroscopy (XPS) measurements were
performed in an Escalab 250Xi with a monochromatized Al K
source (1486 eV). Narrow spectra were collected with pass energy
of 30 eV. The charge correction was made considering that the
C1 s signal of contaminating carbon (C C or C H bonds) was cen-
tered at 284.8 eV. All samples were dried before the XPS test. A
Fourier transform IR spectrometer (NICOLET 5700, USA) was used
to acquire the IR spectra of the samples. KBr was used to make the
samples for FTIR analysis. TG/DTA (Thermogravimetric/Differential
Thermal Analysis) experiments were performed using a Mettler
TGA (Thermogravimetric Analysis) system from room temperature
research using such CeO -based mixed oxides catalysts.
2
2
. Experimental details
2.1. Catalyst preparation
Ce1-xFexO2-␦ mixed oxide catalysts were prepared by a gen-
eral co-precipitation method and x just denotes the molar ratio
of Fe/(Ce + Fe) (x = 0.1, 0.2, 0.3, 0.5, 0.7). Solution A was prepared
by dissolving appropriate amounts of Fe (NO ) ·6H O and Ce
◦
◦
to 600 C at 10 C/min under N flow (30 mL/min).
2
3
3
2
Inductively coupled plasma atomic emission spectrometer (ICP-
AES) (Thermo iCAP6300) was used to measure the bulk Fe/(Ce + Fe)
ratios in the fresh samples. For the analysis, about 10 mg of the
catalyst was dissolved in an aqua regia solution. Afterward, the
solution was diluted to 500 mL using distilled water and 10 mL of
this solution was used for analysis.
(
NO ) ·6H O in the desired molar ratio. Solution B was prepared by
3
3
2
dissolving ammonia water (25–28 wt.%) in 100 mL deionized water.
In a typical procedure, solutions A and B were added at the same
time to a beaker containing 100 mL of deionized water while stir-
ring. The pH of the suspensions was maintained around 9 and then
◦
the solution was aged at 70 C for 4 h. The obtained precipitates
were filtered off, washed with distilled water three times and sub-
2
.3. Catalyst testing
◦
sequently dried at 90 C overnight. Finally, the dried sample was
◦
◦
−1
.
calcined at 500 C for 2 h in the air at a heating rate of 5 C min
Pure CeO2 and Fe O were also prepared for comparison by adopt-
The gas-phase ketonization of carboxylic acids was carried in
2
3
a down-flow quartz fixed-bed reactor (9 mm i.d.) under atmo-
spheric pressure. A HPLC pump was used to introduce the liquid
feed solution into the reactor. In a typical run, 0.3 g of catalyst
ing the same method. The raw chemicals used in this study were
purchased from Sinopharm Chemical. Reagent Co. Ltd., China and
of analytical reagent grade.
(40–60 mesh) was loaded into the reactor between two plugs of
quartz wool and fused SiO2 granules and the reaction was car-
ried out with liquid acetic acid or propionic acid flow rate of
2.2. Catalyst characterization
1
.8 mL/h with an N2 flow rate of 40 mL/min. Each sample was pre-
The powder X-ray diffraction (XRD) patterns of samples were
recorded on a Philips PW3050/60 vertical goniometer using Ni-
filtered Cu K␣1 radiation ( = 1.5406 Å). Transmission electron
treated in N2 flow at the lowest reaction temperature for 2 h prior
to the reaction. The feed and gas-phase reaction mixtures were
analyzed with online GC analysis equipped with two columns in

 Lu, Feipeng
Lu, Feipeng
 Jiang, BinBo
Jiang, BinBo
 Wang, Jingdai
Wang, Jingdai
 Huang, Zhengliang
Huang, Zhengliang
 Liao, Zuwei
Liao, Zuwei
 Yang, Yongrong
Yang, Yongrong