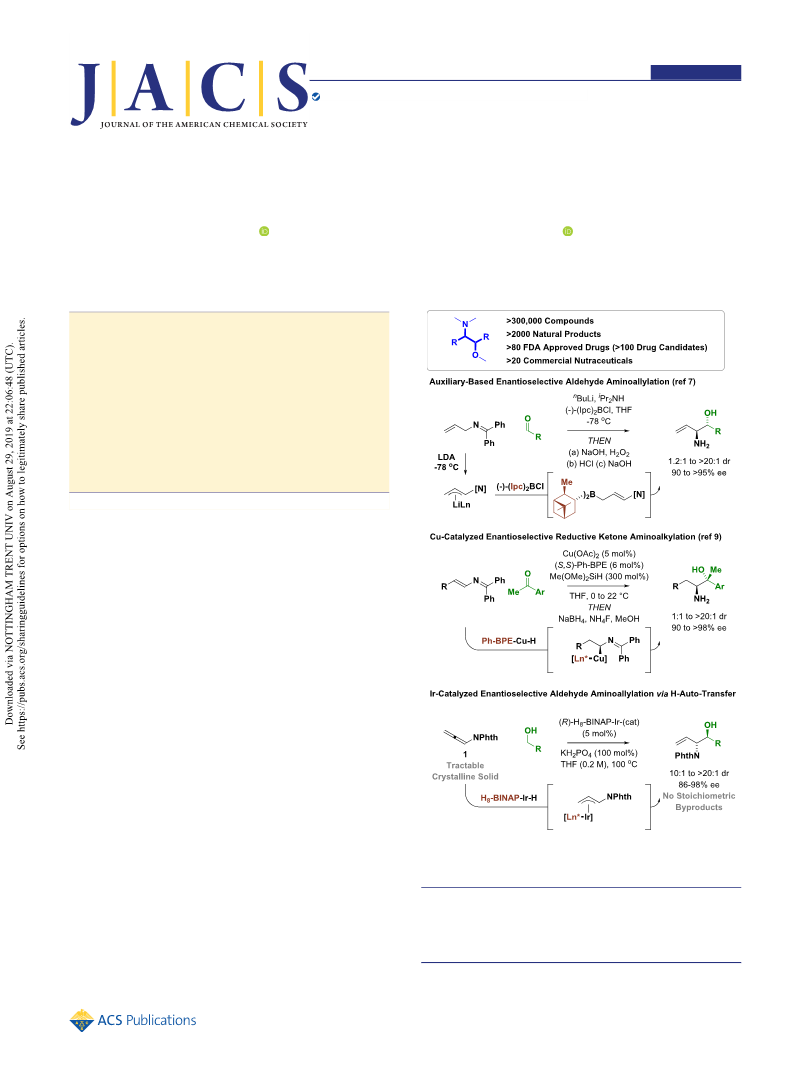
Journal of the American Chemical Society
Communication
metallic Reagents to Carbonyl Compounds: Chirality Transfer,
Multiplication and Amplification. Angew. Chem., Int. Ed. Engl. 1991,
Enantioselective Addition of Allylic Organometallic Reagents to
Aldehydes and Ketones. Chem. Rev. 2003, 103, 2763. (c) Yu, C.-M.;
Youn, J.; Jung, H.-K. Regulation of Stereoselectivity and Reactivity in
the Inter- and Intramolecular Allylic Transfer Reactions. Bull. Korean
Chem. Soc. 2006, 27, 463. (d) Marek, I.; Sklute, G. Creation of
Quaternary Stereocenters in Carbonylallylation Reactions. Chem.
Commun. 2007, 1683. (e) Hall, D. G. Lewis and Brønsted Acid
Catalyzed Allylboration of Carbonyl Compounds: From Discovery to
Mechanism and Applications. Synlett 2007, 11, 1644. (f) Hargaden, G.
C.; Guiry, P. J. The Development of the Asymmetric Nozaki−Hiyama−
Kishi Reaction. Adv. Synth. Catal. 2007, 349, 2407. (g) Lachance, H.;
Hall, D. G. Allylboration of Carbonyl Compounds. Org. React. 2009, 73,
3
0, 49. (b) Soai, K.; Shibata, T. Alkylation of Carbonyl Groups. In
Comprehensive Asymmetric Catalysis I-III; Jacobsen, E. N., Pfaltz, A.,
Yamamoto, H., Eds.; Springer-Verlag Berlin Heidelberg: Germany,
1
999; Vol. 2, pp 911. (c) Pu, L.; Yu, H.-B. Catalytic Asymmetric
Organozinc Additions to Carbonyl Compounds. Chem. Rev. 2001, 101,
57. (d) Trost, B. M.; Weiss, A. H. The Enantioselective Addition of
Alkyne Nucleophiles to Carbonyl Groups. Adv. Synth. Catal. 2009, 351,
63. (e) Comprehensive Organic Synthesis, 2nd ed.; Knochel, P.;
Molander, G. A., Eds.; Elsevier: Oxford, 2014; Vols. 1 and 2.
2) Schneider, N.; Lowe, D. M.; Sayle, R. A.; Tarselli, M. A.; Landrum,
7
9
(
G. A. Big Data from Pharmaceutical Patents: A Computational Analysis
of Medicinal Chemists’ Bread and Butter. J. Med. Chem. 2016, 59, 4385.
1. (h) Yus, M.; Gonzalez-Gomez, J. C.; Foubelo, F. Catalytic
́ ́
Enantioselective Allylation of Carbonyl Compounds and Imines.
Chem. Rev. 2011, 111, 7774. (i) Yus, M.; Gonzalez-Gomez, J. C.;
Foubelo, F. Diastereoselective Allylation of Carbonyl Compounds and
Imines: Application to the Synthesis of Natural Products. Chem. Rev.
2013, 113, 5595. (j) Huo, H.-X.; Duvall, J. R.; Huang, M.-Y.; Hong, R.
Catalytic Asymmetric Allylation of Carbonyl Compounds and Imines
with Allylic Boronates. Org. Chem. Front. 2014, 1, 303. (k) Kumar, P.;
Tripathi, D.; Sharma, B. M.; Dwivedi, N. Transition Metal Catalysis - A
Unique Road Map in the Stereoselective Synthesis of 1,3-Polyols. Org.
Biomol. Chem. 2017, 15, 733. (l) Spielmann, K.; Niel, G.; de Figueiredo,
R. M.; Campagne, J. M. Catalytic Nucleophilic ‘Umpoled’ π-Allyl
Reagents. Chem. Soc. Rev. 2018, 47, 1159.
(
3) For a recent review on metal-catalyzed carbonyl reductive
coupling, see: Holmes, M.; Schwartz, L. A.; Krische, M. J.
Intermolecular Metal-Catalyzed Reductive Coupling of Dienes,
Allenes, and Enynes with Carbonyl Compounds and Imines. Chem.
Rev. 2018, 118, 6026.
(
4) For selected reviews on carbonyl reductive coupling via hydrogen
auto-transfer, see: (a) Kim, S. W.; Zhang, W.; Krische, M. J. Catalytic
Enantioselective Carbonyl Allylation and Propargylation via Alcohol-
Mediated Hydrogen Transfer: Merging the Chemistry of Grignard and
Sabatier. Acc. Chem. Res. 2017, 50, 2371. (b) Doerksen, R. S.; Meyer, C.
C.; Krische, M. J. Feedstock Reagents in Metal-Catalyzed Carbonyl
(9) (a) Li, K.; Shao, X.; Tseng, L.; Malcolmson, S. J. 2-Azadienes as
Reagents for Preparing Chiral Amines: Synthesis of 1,2-Amino Tertiary
Alcohols by Cu-Catalyzed Enantioselective Reductive Couplings with
Ketones. J. Am. Chem. Soc. 2018, 140, 598. (b) Shao, X.; Li, K.;
Malcolmson, S. Enantioselective Synthesis of anti-1,2-Diamines by Cu-
Catalyzed Reductive Couplings of Azadienes with Aldimines and
Ketimines. J. Am. Chem. Soc. 2018, 140, 7083.
(10) Only five catalytic enantioselective allene-carbonyl reductive
couplings have been reported, see: (a) Han, S. B.; Kim, I. S.; Han, H.;
Krische, M. J. Enantioselective Carbonyl Reverse Prenylation from the
Alcohol or Aldehyde Oxidation Level Employing 1,1-Dimethylallene as
the Prenyl Donor. J. Am. Chem. Soc. 2009, 131, 6916. (b) Holmes, M.;
Nguyen, K. D.; Schwartz, L. A.; Luong, T.; Krische, M. J.
(
5) (a) Ghislieri, D.; Turner, N. J. Biocatalytic Approaches to the
Synthesis of Enantiomerically Pure Chiral Amines. Top. Catal. 2014,
7, 284. (b) Breuer, M.; Ditrich, K.; Habicher, T.; Hauer, B.; Kesseler,
M.; Sturmer, R.; Zelinski, T. Industrial Methods for the Production of
Optically Active Intermediates. Angew. Chem., Int. Ed. 2004, 43, 788.
c) Sehl, T.; Maugeri, Z.; Rother, D. Multi-step Synthesis Strategies
5
̈
(
towards 1,2-Amino Alcohols with Special Emphasis on Phenyl-
propanolamine. J. Mol. Catal. B: Enzym. 2015, 114, 65 and references
cited therein .
(
6) For racemic vicinal amino alcohols via metal-catalyzed carbonyl
(α-amino)allylation, see: (a) Skucas, E.; Zbieg, J. R.; Krische, M. J. anti-
Enantioselective Formation of CF -Bearing All-Carbon Quaternary
3
Aminoallylation of Aldehydes via Ruthenium Catalyzed Transfer
Hydrogenative Coupling of Sulfonamido-Allenes: 1,2-Aminoalcohols.
J. Am. Chem. Soc. 2009, 131, 5054. (b) Zbieg, J. R.; McInturff, E. L.;
Krische, M. J. Allenamide Hydro-Hydroxyalkylation: 1,2-Amino-
alcohols via Ruthenium Catalyzed Carbonyl anti-Aminoallylation.
Org. Lett. 2010, 12, 2514. (c) Zhang, W.; Chen, W.; Xiao, H.; Krische,
M. J. Carbonyl anti-(α-Amino)allylation via Ruthenium Catalyzed
Hydrogen Auto-Transfer: Use of an Acetylenic Pyrrole as an Allylmetal
Stereocenters via C-H Functionalization of Methanol: Iridium
Catalyzed Allene Hydrohydroxymethylation. J. Am. Chem. Soc. 2017,
139, 8114. (c) Tsai, E. Y.; Liu, R. Y.; Yang, Y.; Buchwald, S. L. A Regio-
and Enantioselective CuH-Catalyzed Ketone Allylation with Terminal
Allenes. J. Am. Chem. Soc. 2018, 140, 2007. (d) Schwartz, L. A.; Holmes,
M.; Brito, G. A.; Gonc a̧ lves, T. P.; Richardson, J.; Ruble, J. C.; Huang,
K.-W.; Krische, M. J. Cyclometallated Iridium-PhanePhos Complexes
Are Active Catalysts in Enantioselective Allene-Fluoral Reductive
Coupling and Related Alcohol-Mediated Carbonyl Additions that
Form Acyclic Quaternary Carbon Stereocenters. J. Am. Chem. Soc.
2019, 141, 2087. (e) Liu, R. Y.; Zhou, Y.; Yang, Y.; Buchwald, S. L.
Enantioselective Allylation Using Allene, a Petroleum Cracking
Byproduct. J. Am. Chem. Soc. 2019, 141, 2251.
Pronucleophile. Org. Lett. 2017, 19, 4876. (d) Schwarz, J. L.; Schaf
̈
ers,
F.; Tlahuext-Aca, A.; Luckemeier, L.; Glorius, F. Diastereoselective
̈
Allylation of Aldehydes by Dual Photoredox and Chromium Catalysis.
J. Am. Chem. Soc. 2018, 140, 12705.
(
7) (a) Barrett, A. G. M.; Seefeld, M. A. B-[(E)-3-(Diphenylamino)-
allyl]diisopinocampheylborane: An Excellent Reagent for The Stereo-
selective Synthesis of anti-β-Diphenylamino Alcohols. J. Chem. Soc.,
Chem. Commun. 1993, 339. (b) Barrett, A. G. M.; Seefeld, M. A. The
Use of B-[(E)-3-(Diphenylamino)allyl]diisopinocampheylborane as A
Reagent for The Stereoselective Synthesis of anti-β-Diphenylamino
Alcohols and trans-1-Diphenylamino-2-(1-hydroxylalkyl)-
cyclopropanes. Tetrahedron 1993, 49, 7857. (c) Barrett, A. G. M.;
Seefeld, M. A.; Williams, D. J. Convenient Asymmetric Synthesis of
anti-β-Amino Alcohols: An X-Ray Crystallographic Study of (4R)-2,2-
Dimethyl-4-[(2S)-(diphenylmethyleneamino)-(1S)-hydroxy-3-buten-
(11) (a) Fenandez, I.; Monterde, M. I.; Plumet, J. On the Base-
Induced Isomerization of Cyclic Propargylamides to Cyclic Allena-
mides. Tetrahedron Lett. 2005, 46, 6029. (b) Lindsay, V. N. G.; Fiset,
D.; Gritsch, P. J.; Azzi, S.; Charette, A. B. Stereoselective Rh (S-IBAZ) -
Catalyzed Cyclopropanation of Alkenes, Alkynes, and Allenes:
Asymmetric Synthesis of Diacceptor Cyclopropylphosphonates and
Alkylidenecyclopropanes. J. Am. Chem. Soc. 2013, 135, 1463.
(12) Bower, J. F.; Skucas, E.; Patman, R. L.; Krische, M. J. Catalytic C-
C Coupling via Transfer Hydrogenation: Reverse Prenylation,
Crotylation and Allylation from the Alcohol or Aldehyde Oxidation
Level. J. Am. Chem. Soc. 2007, 129, 15134.
2
4
1
-yl]-1,3-dioxolane. J. Chem. Soc., Chem. Commun. 1994, 1053.
(
d) Barrett, A. G. M.; Seefeld, M. A.; White, A. J. P.; Williams, D. J.
(13) (a) Zhang, X.; Mashima, K.; Koyano, K.; Sayo, N.; Kumobayashi,
H.; Akutagawa, S.; Takaya, H. Synthesis of Partially Hydrogenated
BINAP Variants. Tetrahedron Lett. 1991, 32, 7283. (b) Zhang, X.;
Taketomi, T.; Yoshizumi, T.; Kumobayashi, H.; Akutagawa, S.;
Mashima, K.; Takaya, H. Asymmetric Hydrogenation of Cyclo-
alkanones Catalyzed by BINAP-Ir(I)-Aminophosphine Systems. J.
Convenient Asymmetric Syntheses of anti-β-Amino Alcohols. J. Org.
Chem. 1996, 61, 2677.
(
8) For selected reviews on enantioselective carbonyl allylation, see:
a) Ramachandran, P. V. Pinane-based versatile ″allyl″ boranes.
Aldrichimica Acta 2002, 35, 23. (b) Denmark, S. E.; Fu, J. Catalytic
(
E
J. Am. Chem. Soc. XXXX, XXX, XXX−XXX

 Spielmann, Kim
Spielmann, Kim
 Xiang, Ming
Xiang, Ming
 Schwartz, Leyah A.
Schwartz, Leyah A.
 Krische, Michael J.
Krische, Michael J.