Protonation of trimipramine salts by 1H, 13C and 15N NMR 167
and 1H–13C, 1H–15N gradient-enhanced heteronuclear mul-
tiple bond correlation12 (PFG-HMBC) experiments were
performed at room temperature. For assigning the broad
signals arising from acidic protons, 1H–13C HMBC, DQF-
COSY and ROESY13,14 experiments were performed for the
maleate salt at 223 K and for the mesylate salt at 263 K, both
in CDCl3.
were filtered and washed with cold acetone and dried under
vacuum to leave white crystals of trimipramine maleate
(7.34 g, 88.2%).
Synthesis of trimipramine mesylate (mesylate salt)
Trimipramine (6 g, 0.02 mol) was dissolved in 40 ml of
dry acetone, cooled to 268–270 K under nitrogen and dry
methanesulfonic acid (1.92 g, 0.02 mol) was added. The
mixture was stirred for 2 h under nitrogen and then cooled
to 258 K to give white crystals of trimipramine mesylate.
The crystals thus obtained were filtered and washed with
cold acetone and dried under vacuum to give white crystals
of trimipramine mesylate (4.5 g, 57.6%). This salt is highly
hygroscopic and, therefore, it was dried under nitrogen and
kept under a nitrogen atmosphere.
For DQF-COSY and ROESY experiments, a spectralwidth
of 10 700 Hz was used in both the dimensions. About 512
FIDs were obtained with t1 incrementation, each of 2048
complex data points. Phase-sensitive data were obtained by
the TPPI method.15 The relaxation delay and the number
of repetitions were 2.5 s and 16 for DQF-COSY and 1.5 s
and 40 for ROESY experiments. A spin lock time of 150 ms
was used for the ROESY experiment. For 1H–13C, HSQC
and HMBC experiments, spectral widths of 10 700 Hz and
1
24 000 Hz in the H and 13C dimensions, respectively, were
Synthesis of trimipramine hydrochloride
(hydrochloride salt)
used. For the HSQC and HMBC experiments, 256 and 400
FIDs, respectively, were collected with t1 incrementation,
each of 2048 data points, 32 transients and a 2 s recycle
delay. For 1H–15N HMBC experiments, a spectral width
of 4800 Hz and 3650 Hz in the 1H and 15N dimensions,
respectively, were used. About 400 FIDs were collected with
t1 incrementation, each of 2048 points, 64 transients and a 1.5 s
recycle delay. Phase-sensitive data for HSQC experiments
were obtained using the echo–antiecho method,16 and for
HMBC experiments, magnitude mode data were obtained.
The resulting data from all 2D experiments were zero-filled
to 1024 points in t1 and double Fourier transformed after
multiplying by a squared sine-bell window function shifted
by ꢀ/2 along both dimensions.
13C T1 relaxation measurements were made at 298 K
using the inversion–recovery method incorporating inverse
gated proton decoupling. Parameters used were spectral
width 24 000 Hz, time domain points 32 K, relaxation delay
50 s, number of scans 24, inversion–recovery delay varied
between 25 ms and 50 s in 20 steps, spectrum size 64K and
a line broadening of 3.0 Hz. From the resulting data, the
relaxation times of individual carbons were calculated using
Bruker Xwinnmr software version 3.1.
Trimipramine (6 g, 0.02 mol) was dissolved in 40 ml of dry
acetone, cooled to 268–270 K under nitrogen and dry HCl
gas was purged into the reaction mass until the pH of the
reaction medium was 2 as monitored with litmus paper. The
crystals thus obtained were filtered and washed with cold
acetone. The white crystals were dried under vacuum at
343–253 K for 4 h to give trimipramine hydrochloride (5.5 g,
83%).
NMR experiments
The NMR experiments were performed in deuterated
chloroform (99.8% D, Aldrich, Milwaukee, WI, USA). For
studying the broad acidic proton signals at temperatures
lower than the freezing-point of chloroform, CD2Cl2 (99.8%
D, Aldrich) was used and for 1D 15N experiments, a 1 : 5
mixture of deuterated (DMSO-d6; 99.8% D, Aldrich) and non-
deuterated dimethyl sulfoxide were used as solvents. The
experiments were performed on a Bruker Biospin Avance
400 spectrometer using 5 mm BBI and BBO probe heads.
Normal 1D 1H and 13C experiments were performed at room
temperature. 1D 1H NMR spectra at variable temperature
were obtained for all the three salts at 25 mM concentration,
varying the temperature in steps of 10 K from 298 to 218 K in
chloroform solution. For the maleate and the hydrochloride
salts, 1H spectra varying the temperature from 298 to 198 K
in steps of 10 K were also obtained in dichloromethane
solution. 1D 1H spectra were measured with a spectral
width, of 10 700 Hz, 32 K data points, 10 s relaxation delay,
NMR experiments on the free trimipramine base were
also performed, under identical conditions, for comparison
of the results.
RESULTS AND DISCUSSION
°
45 pulse angle, 16 scans, and a spectral size of 32 K with
All three trimipramine salts show a single set of signals with
an anionic and cationic ratio of 1 : 1 as seen from the intensity
of the NMR signals. Analyses of the 1H, 13C resonances
were made using the combination of two-dimensional DQF-
COSY, ROESY, PFG-PEP-HSQC, PFG-HMBC and variable-
temperature experiments (Table 1). The structures of the
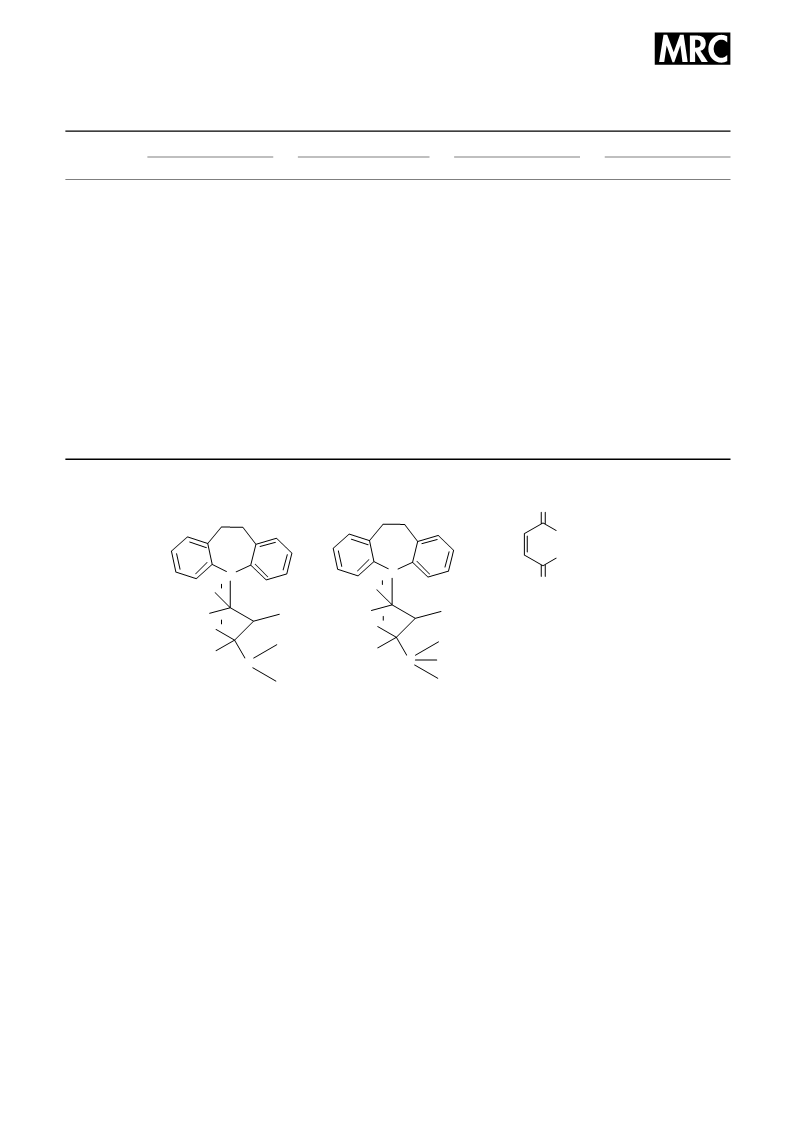
free base and its three salts and the labeling are shown in
Scheme 1.
0.3 Hz line broadening. 1D 13C spectra were obtained at
room temperature using WALTZ-16 1H decoupling with
spectral width 24 000 Hz, relaxation delay 3 s, time domain
points 32 K, pulse angle 45 , number of scans 32 and a line
broadening of 3 Hz. 1D 15N experiments in DMSO-d6 were
°
performed at 323 K using a spectral width of 8090 Hz, 20 s
relaxation delay, 32 K time domain points, 45 pulse angle,
°
4000 scans and a line broadening of 3 Hz.
For the maleate salt, a broad signal around 12 ppm
(75 Hz linewidth) was observable at 298 K arising from one
of the acidic protons, H-a or H-f, in both chloroform and
dichloromethane. The other acidic proton signal appeared
around 18 ppm, which was too broad to be clearly seen at
1
For complete, unambiguous assignments of the H, 13C
and 15N signals, homonuclear and heteronuclear 2D experi-
ments such as DQF-COSY,7,8 1H–13C gradient-enhanced het-
eronuclear single quantum correlation9–11 (PFG-PEP-HSQC)
Copyright 2004 John Wiley & Sons, Ltd.
Magn. Reson. Chem. 2005; 43: 166–170

 Somashekar
Somashekar
 Nagana Gowda
Nagana Gowda
 Ramesha
Ramesha
 Khetrapal
Khetrapal