Absorption Spectra of Ag0 and Ag2 in Water
J. Phys. Chem. A, Vol. 106, No. 13, 2002 3127
+
(2) Buxton, G. V.; Greenstock, C. L.; Helman, W. P.; Ross, A. B. J.
Phys. Chem. Ref. Data. 1988, 17, 513.
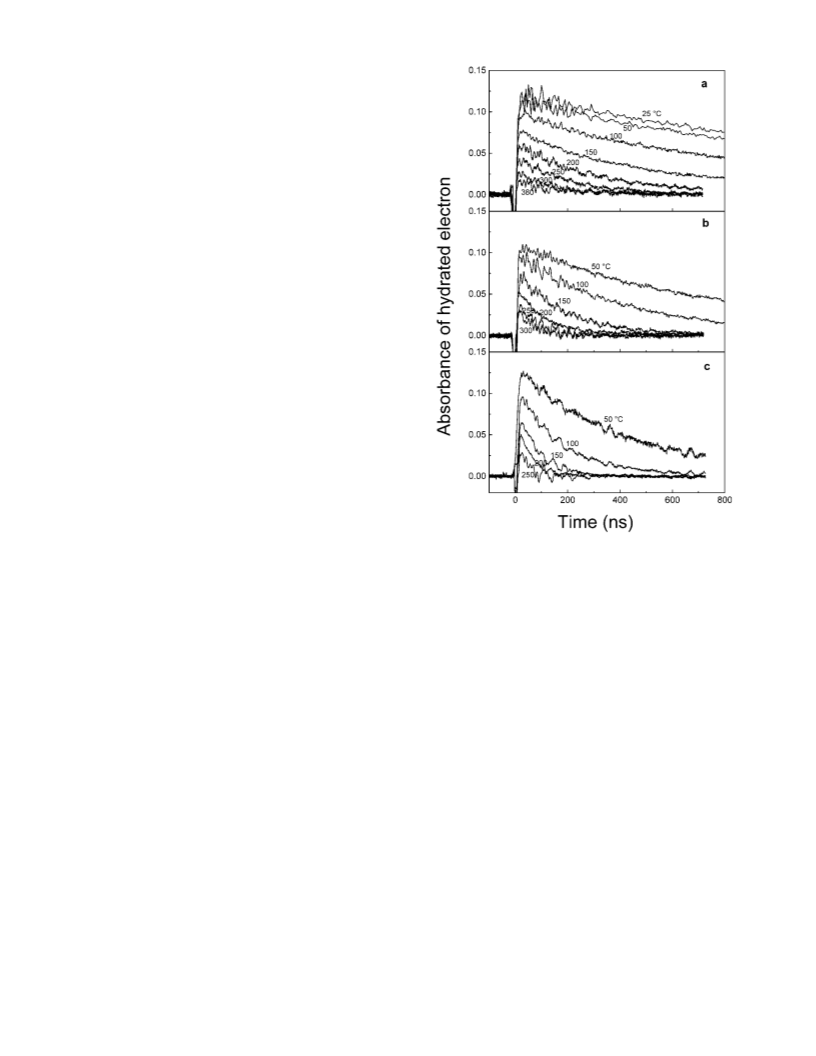
density of supercritical water at 380 °C (and 300 atm) is almost
half of that for water at room temperature. Therefore the reported
absorption spectra should be corrected taking into account the
change of water density.
(3) Henglein, A. Chem. ReV. 1989, 89, 1861.
(4) Belloni, J.; Amblard, J.; Marignier, J. L.; Mostafavi, M. In Clusters
of atoms and Molecules; Haberland, H., Ed.; Springer-Verlag II: New York,
1994; p 290.
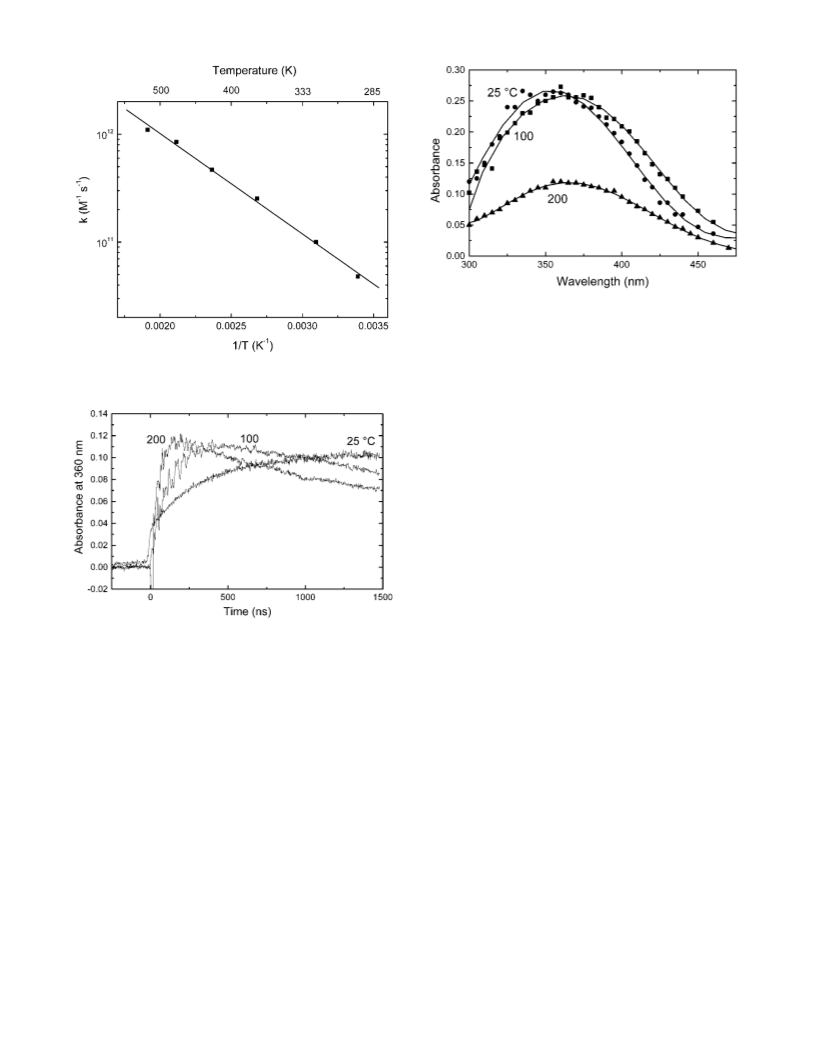
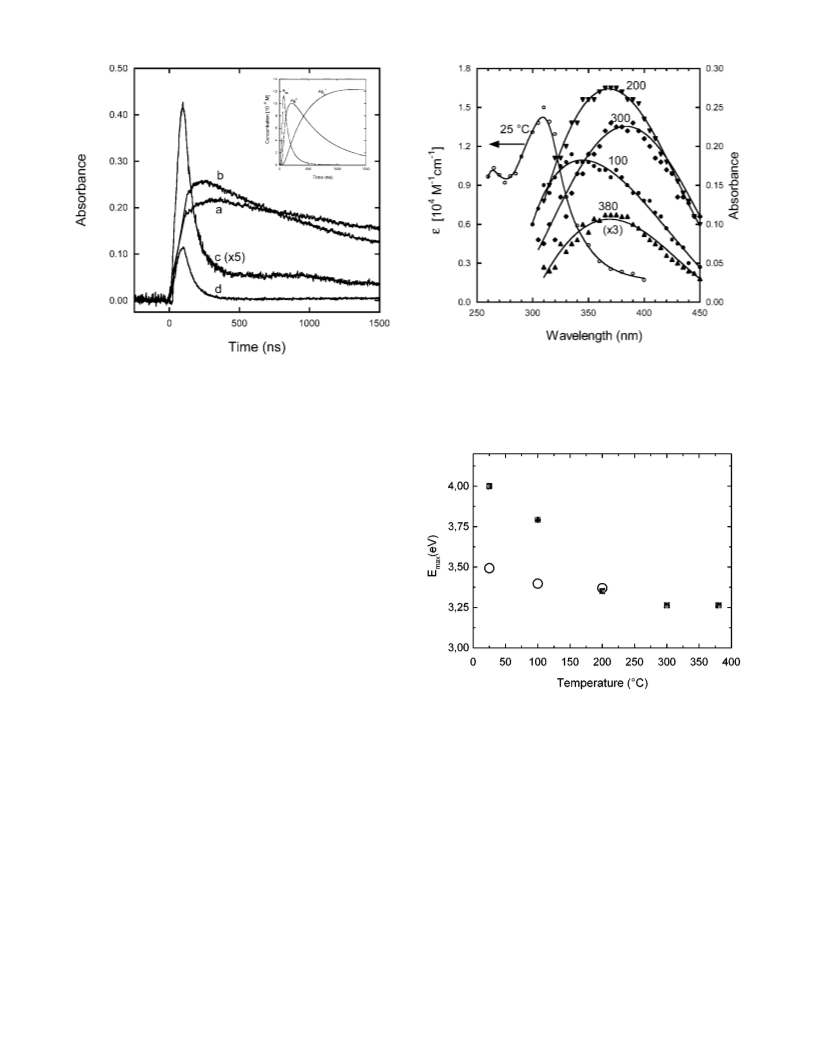
Figure 7 shows the change of the maximum of absorption
band for the silver atom and the charged dimer in water versus
the temperature from 25 to 300 °C. The shift of the absorption
(5) Henglein, A. Ber. Bunsen-Ges. Phys. Chem. 1995, 99, 903.
(6) Belloni, J.; Mostafavi, M. In Metal Clusters in Chemistry; Braun-
stein, P., Oro L. A., Raithby, P. R., Eds.; J. Wiley, VCH: New York, 1999;
pp 1213-1247.
+
spectra of Ag2 is very clear and it is more pronounced than
that of silver atoms. This result is in agreement with the fact
that the solvation energy of neutral silver atom is much less
(less than 0.1 eV) than that of the charged dimers (around 3.5
(7) Baxendale, J. H.; Fielden, E. M.; Keene, J. P.; Ebert, M. In Pulse
Radiolysis; Keene, J. P., Swallow, A., Baxendale, J. H., Eds.; Acadamic
Press: London, 1965; pp 207-220.
(8) Tausch-Treml, R.; Henglein, A.; Lilie, J. Ber. Bunsen-Ges. Phys.
Chem. 1978, 82, 1343.
+
eV). Therefore, the solvent molecules around Ag2 are more
organized and the temperature effect on the solvation shell is
expected to be much pronounced.
(9) Von Pukies, J.; Roebke, W.; Henglein, A. Ber. Bunsen-Ges. Phys.
Chem. 1968, 72, 842.
(10) Mostafavi, M.; Marignier, J. L.; Amblard, J.; Belloni, J. Radiat.
Phys. Chem. 1989, 34, 605.
(11) Janata, E.; Henglein, A.; Ershov, B. G. J. Phys. Chem. 1994, 98,
10888.
(12) Janata, E.; Lilie, J.; Martin, M. Radiat. Phys. Chem. 1994, 43, 353.
(13) Janata, E. Radiat. Phys. Chem. 1994, 44, 449.
(14) Kappoor, S.; Lawless, D.; Kennepohl, P.; Meisel, D.; Serpone, N.
Langmuir 1994, 10, 3018.
(15) Belloni, J.; Delcourt, M. O.; Marignier, J. L.; Amblard, J. In
Radiation Chemistry; Hedwig, P., Nyikos, L., Schiller, R., Eds.; Akade´miai
Kia¨do: Budapest, 1987; p 89.
(16) Belloni, J.; Khatouri, J.; Mostafavi, M.; Amblard, J. In Ultrafast
reaction Dynamics and SolVent Effects; Rossky, P. J., Gauduel, Y., Eds.;
American Institute of Physics: Woodbury, NY, 1994; p 527.
(17) Dorfman, L. M.; You, F. Y. In Electrons in Fluids; Jortner, J.,
Kestner, N. R., Eds.; Springer: Berlin, 1973; p 447.
(18) Kevan, L. J. Phys. Chem. 1981, 85, 1828.
(19) Basco, N.; Vidyarthi, S. K.; Walker, D. C. Can. J. Chem. 1973,
51, 2497.
(20) Fargataziz, Cordier, P.; Perkey, L. M. Radiat. Res. 1976, 68, 23.
(21) Dimitrijevic, Nada M.; Bartels, D. M.; Johnah, C. D.; Takahashi,
K.; Rajh, T. J. Phys. Chem. A 2001, 105, 954.
Conclusion
The rate constant for the reduction of silver ions by hydrated
electrons is fitted to the Arrhenius relationship (from 25 to 300
°C at 200 atm) and the apparent activation energy is found to
be 17.8 kJ mol-1. The measurements of transient absorption
spectrum show that the maximum of the absorption spectra of
silver atom is shifted from 355 to 370 nm when the temperature
is increased from 25 to 200 °C. The shift of the absorption
+
maximum is more pronounced for the charged dimer, Ag2
.
The maximum of the absorption band located at 315 nm at room
temperature is shifted to 380 nm at 300 °C in water. We note
that the shift of the maximum of the absorption band of hydrated
electron for which the hydration free energy is 1.6 eV is also
very significant.22,30
Our observations over a wide range of temperature confirm
+
clearly that the absorption spectra of Ag2 presents CTTS
characteristics but that of silver atom does not depend so
significantly to the temperature.31 A molecular dynamics
simulation is in progress to help in understanding of the
orientation of water molecules around the atom and the charged
dimer with increasing temperatures.
(22) Wu, G.; Katsumura, Y.; Muroya, Y.; Li, X.; Terada, Y. Chem. Phys.
Lett. 2000, 325, 531.
(23) Buxton, G. V.; Stuart, C. R. J. Chem. Soc., Faraday Trans. 1995,
91, 279.
(24) Wu, G.; Katsumura, Y.; Muroya, Y.; Li, X.; Terada, Y. Radiat.
Phys. Chem. 2001, 60, 395.
(25) Mills, R. J. Phys. Chem. 1973, 77, 685.
(26) Harris, K. R.; Woolf, L. A. J. Chem. Soc., Faraday Trans. I. 1980,
76, 377.
(27) Krynicki, K.; Green, C. D.; Sawyer, D. W. Faraday Discuss. 1980,
66, 199.
(28) Elliot A. J. Rate Constants and G-Values for the Simulation of the
Radiolysis of Light Water over the Range 0-300 °C. Reports Nos. AECL-
11073, COG-94-167; Atomic Energy of Canada Ltd.: Ontario, Canada, 1994;
p 2.
Acknowledgment. This work was supported by Japan
Society for the Promotion of Science under the contract
JSPS-RFTF 98P00901. The authors warmly thank Tomomi
Morioka, graduate student, for the help in several experiments.
The authors also thank Dr. Kenji Takahashi, Hokkaido Uni-
versity, for the design of the new cell. This work was also partly
supported by the innovative basic research program in the field
of high temperature engineering using the HTTR conducted by
Japan Atomic Energy Research Institute.
(29) Buxton, G. V.; Mackenzie, S. R. J. Chem. Soc., Faraday Trans.
1992, 88, 2833.
(30) Christensen, H.; Sehested, K. J. Phys. Chem. 1986, 90, 186.
(31) According to a personal communication made by our colleague
Dr. P. Archirel, the recent SCF calculations shows that the solvation of the
external electron of silver atom is very improbable then the silver atom
spectra in water does not present a real CTTS character. That could explain
why we find that the shift of the absorption spectra versus temperature is
not very important.
References and Notes
(1) Baxendale, J. H.; Busi, F. The Study of fast Processes and transient
Species by Electron Pulse Radiolysis, NATO ASI Series 86; D. Reidel
Pubishing Co.: Dordrecht, 1982.

 Mostafavi, Mehran
Mostafavi, Mehran
 Lin, Mingzhang
Lin, Mingzhang
 Wu, Guozhong
Wu, Guozhong
 Katsumura, Yosuke
Katsumura, Yosuke
 Muroya, Yusa
Muroya, Yusa