1406
SKORIK
the mixture and in the adduct is observed at 198 nm as ylimidazole) imitating the proton donor–chain–
compared to absorption of the initial components acceptor system, a water molecule acts as a bridge to
(
H2Fol and Im).
transfer a proton from protonated methylimidazole to
organic acid anion. In this ionic molecular complex,
N and O atoms are involved in hydrogen bonds. The
The shift of intense bands of bending vibrations of
the ring (1099.9 and 1053.9 cm–1) towards higher freꢀ
quencies (1101.7 and 1056.2 cm–1) and the intact band
of stretching vibrations of the N–H bond of the pyrꢀ
role group (3050–3300 cm–1 [16]) when proceeding
from unbound imidazole Im (3143.5 cm–1) to its conꢀ
pKa values of the molecular complex components
need to be similar to ensure mutual proton exchange.
The presence of proton exchange in conjugate
H2Fol · 3Im · 2H2O can be confirmed by the fact that
its IR spectrum contains absorption bands of both
nonꢀdissociated and dissociated carboxylic groups of
folic acid.
jugate H2Fol · 3Im ·
2H2O (3143.5 cm–1), indicate that
the pyridine nitrogen atom N(3) is involved in binding
to protonated carboxylic groups of folic acid and probꢀ
ably the hydroxyl group. The IR spectrum of the conꢀ
jugate still contains the band of stretching vibrations of
the carboxyl group C=O (1694.4 cm–1) of H2Fol, but
new bands emerge at 1605 and 1404 cm–1, which are
responsible for the asymmetric and symmetric stretchꢀ
ing vibrations of СОО– groups; these bands are also
present in metal folates (1602.5–1604.6 cm–1).
REFERENCES
1. L. O. Shnaidman, Production of Vitamins (Pishchevaya
Promyshlennost, Moscow, 1973) [in Russian].
2. B. L. Hutchings, Patent US 2470490 publ. 1949.
3. E. Kuh and J. Smith, Patent US 2474184 publ. 1949.
4. G. A. Sofronov, M. S. Ukader, I. D. Frizen, et al.,
Patent RU 2132686 (1999).
5 . B . A . K a m e n a n d A . K . S m i t h , A d v. D r u g D e l i v. R e v.
56, 1085 (2004).
6. Lu Veiyu, Liu Min, and Pan Yun. Patent RU 2280650
publ. 2006.
7. T. A. Pavich, A. V. Vorobei, S. M. Arabei, and
K. N. Solov’ev, Zh. Prikl. Spectrosk. 79, 664 (2012).
8. A. N. Eremin, G. K. Zhavnerko, and V. E. Agabekov,
An analysis of thermogravimetry data (Table 1)
shows the conjugate and salts lose water of crystallizaꢀ
tion at the temperature range of 92–142
acid loses water at 165 . Thermal decomposition in
folate anion with complex structure is accompanied by
a set of endotherms (301–419 ) and exotherms
360–620 ). The decomposition of folic acid takes
place in a temperature range of 234–700 and is
accompanied by several exoꢀ and one endotherm at
414 . The conjugate loses imidazole (235 ) with an
exotherm effect and folic acid is decomposed at 405–
700 (with endoterms and exotherms).
°C, while folic
°
C
°
C
(
°С
°
C
Dokl. NAN Belarusi 55 (4), 54 (2011).
°
C
°С
9. A. V. Abakshonok, A. N. Eremin, V. E. Agabekov, and
G. K. Zhavnerko, Ross. Bioterapevt. Zh. 12 (2), 2
(2013).
10. I. A. Milevich, S. A. Vorob’eva, and A. I. Lesnikovich,
Vestn. Bel. Gos. Univ., Ser. 2, No. 1, 30 (2011).
°C
Thus, the combination of analytic, spectral, and
thermogravimetry data confirms the formation of the
folic acid–imidazole conjugate. The synthesized conꢀ
11. J. Pfiffner, S. Binkley, and E. Bloom, and B. O’Dell,
jugate H2Fol · 3Im · 2H2O can be regarded as a molecꢀ
J. Am. Chem. Soc. 69, 1476 (1947).
12. L. V. Korol’chenko, T. Ya. Sakharnaya, E. L. Pidemskii,
et al., Patent RU 739884 publ. 2006.
13. L. V. Simak, T. B. Karpova, E. L. Pidemskii, et al.,
Patent RU 585684 (2006).
14. R. Dawson, D. Elliott, W. Elliott, and K. Jones, Data
for Biochemical Research (Clarendon, Oxford, 1986).
15. U. Radzhobov, Doctorate Dissertation in Chemistry
(Dushanbe, 2011).
16. I. M. Sharipov, Candidate Dissertation in Pharmaceuꢀ
tical Science (Samara, 2014).
17. V. P. Parini, Usp. Khim. 31, 822 (1982).
18. A. N. Isaev, Ross. Khim. Zh. 51 (5), 34 (2007).
Translated by D. Terpilovskaya
ular chargeꢀtransfer complex (CTC) (CTCs of azoles
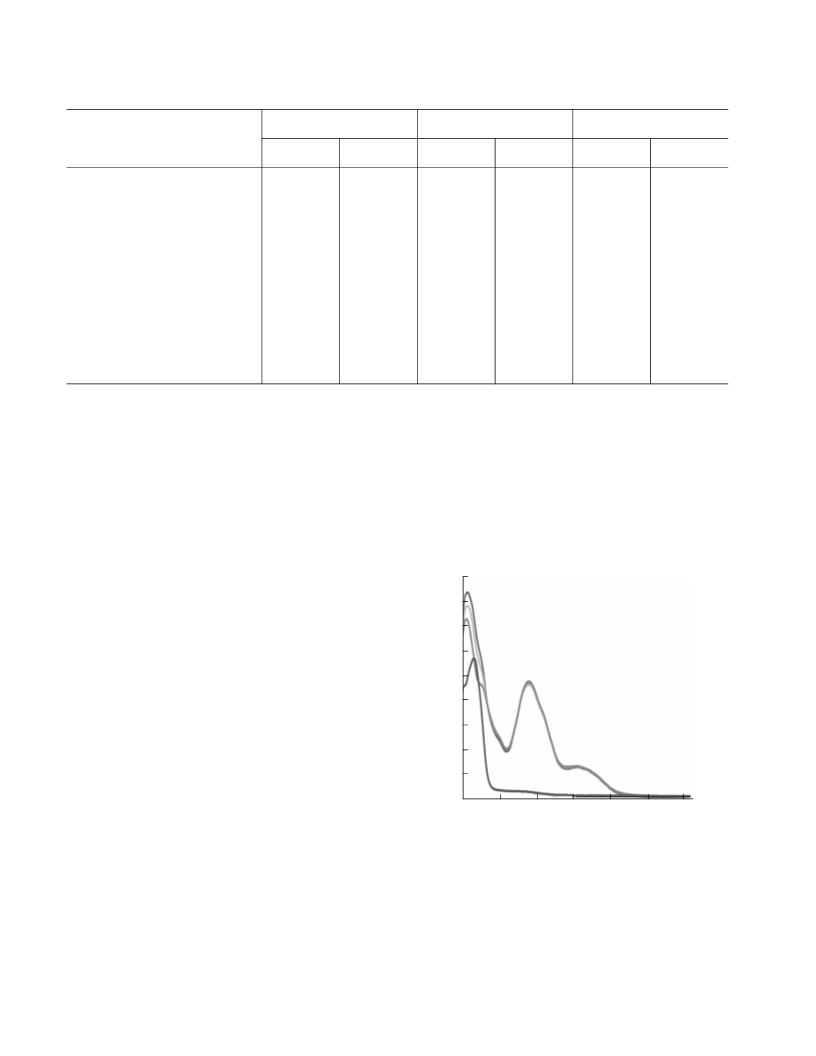
have been poorly studied). The charge transfer band
for this complex shifts from the UV region to the visiꢀ
ble spectral region (figure); the solution is yellow. It is
almost only aromatic compounds that act as donors in
organic CTCs [17]. According to the modern views,
molecular complexes can contain ions, free radicals,
ion radicals, and excited molecules. Molecular comꢀ
plexes also include hydrogenꢀbonded complexes.
Isaev [18] performed quantumꢀchemical simulaꢀ
tion of proton transfer in some molecular systems
similar to the system discussed in this study. In the
hydrogenꢀbonded molecular complex MeIm–
+
…
–
…
H
H2O CH3COO (MeIm–H+ is protonated methꢀ
RUSSIAN JOURNAL OF INORGANIC CHEMISTRY Vol. 60 No. 11 2015

 Skorik
Skorik