Encapsulation of Ag films on SiO2 by Ti reactions using Ag–Ti alloy/bilayer
structures and an NH3 ambient
T. L. Alford and Daniel Adams
Department of Chemical, Bio, and Materials Engineering, Arizona State University, Tempe,
Arizona 85287-6006
T. Laursen and B. Manfred Ullricha)
Center for Solid State Science, Arizona State University, Tempe, Arizona 85287-1704
͑Received 16 February 1996; accepted for publication 29 March 1996͒
Thin encapsulated silver films have been prepared on oxidized silicon by nitridation of
ϳ200-nm-thick Ag–19 at. % Ti alloy films and Ag͑120 nm͒/Ti͑22 nm͒ at 300–700 °C in an
ammonia ambient. The encapsulation process has been studied in detail by Rutherford
backscattering, and scanning Auger and secondary-ion-mass spectrometry, which showed that
Ti-nitride and Ti-oxide-silicide formation take place at the surface and the Ag–Ti/SiO2 interface,
respectively. Four-point-probe analysis of the alloy films suggests that the resistivity is controlled by
the residual Ti concentration. Resistivity values of ϳ4 ⍀ cm were measured in encapsulated Ag
alloy films with initial low Ti concentrations. The annealed bilayer structure had minimal Ti
accumulations in Ag and the resistivity values were comparable to that of the as-deposited Ag ͑ϳ3
⍀ cm͒. © 1996 American Institute of Physics. ͓S0003-6951͑96͒03923-X͔
The electrical connections in integrated circuits ͑IC͒ are
provided by a metallization scheme, which in the case of
very-large-scale integration ͑VLSI͒ is based on Al-alloy line
structures. As IC technologies move toward ultralarge-scale
integration ͑ULSI͒, Cu- and Ag-based metallization schemes
are being considered for replacing the conventional Al-based
schemes.1 Elemental Cu and Ag have low resistivities and
better electromigration resistance than Al. Such improved
inherent properties have become required specifications for
metallization with feature sizes less than 0.25 m.
Metallization line structures are required to have struc-
tural integrity and good electrical conductivity, and they
must maintain these properties throughout subsequent fabri-
cation and operation of the IC. One concern regarding struc-
tural integrity is the poor adhesion of Cu and Ag to dielectric
materials, such as SiO2. Another potential concern is Ag
agglomeration, which has been observed when annealing
takes place in air.2 Tarnishing of Ag in sulfur environments,3
and the oxidation of Cu will require passivation layers to
ensure structural integrity.4 Reinforcing the structural integ-
rity can be accomplished by interfacial chemical reactions.
Such reactions should limit themselves to the formation of
thin reaction layers that promote adhesion and prevent
atomic transport between the metal and its surroundings ͑i.e.,
provide thin diffusion barriers͒. In cases where adjoining ma-
terials do not display this behavior, a desired interfacial
structure can often be formed by adding appropriate elemen-
tal films or alloying elements to the metal. This approach has
been taken in studies of Al, Mg, Ti, and Cr additions in
connection with Cu metallization.5–7
top surface, whereas, a Ti silicide–oxide bilayer is formed
between the Cu and SiO2. The presence of oxygen in TiN͑O͒
is due to residual oxygen in the NH3 ambient.6,7 This letter
reports on such an ‘‘encapsulation’’ process for Ag, where
the Ti is introduced by deposition of Ag–Ti alloys and bi-
layers. This approach has demonstrated more promise in ob-
taining low-resistivity films than encapsulation of Cu.
The Ag–Ti depositions were prepared on Si wafers with
210-nm-thick thermally grown oxides. The alloy structures
consisting of ϳ200-nm-thick Ag–x% Ti ͑where xϭ6, 10,
18, 19, and 26 at. %͒ films were made by dual electron-beam
deposition. The bilayer structure corresponded to 15 at. %
Ti, and were prepared by sputter deposition of a 22-nm-thick
Ti film followed by 120 nm Ag without breaking vacuum.
The encapsulation process took place at 300–700 °C in a
fused-quartz-tube furnace using a flowing NH3 ambient of
99.99% purity and a flow rate of 2 l/min at atmospheric
pressure. The encapsulated Ag films have been characterized
by depth profiling: Rutherford backscattering spectrometry
͑RBS͒, Auger electron spectroscopy ͑AES͒, and secondary-
ion-mass spectrometry ͑SIMS͒. Glancing-angle x-ray-
diffraction and four-point-probe resistance measurements
were used to determine the films’ structural and electrical
properties.
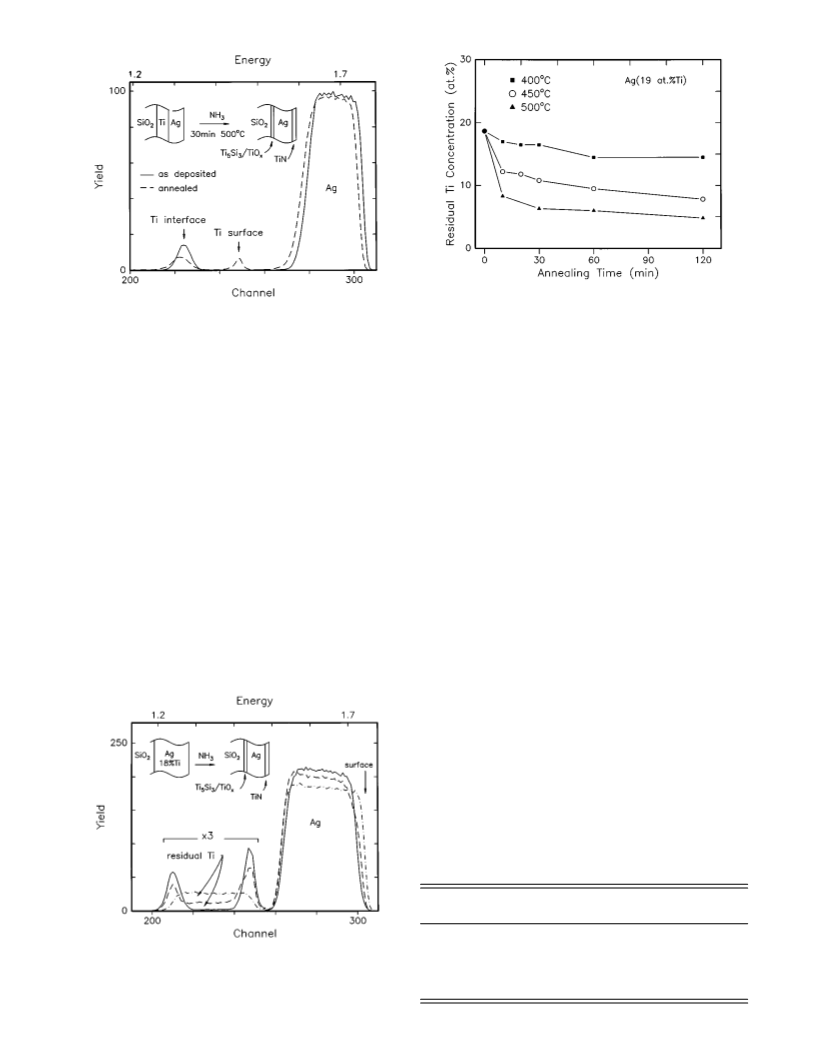
The encapsulation of Ag proceeded in a similar way to
that of Cu. After annealing in an NH3 ambient for 30 min,
the surface changed to have a golden color indicative of TiN
formation. The results of the RBS characterization are shown
in Fig. 1 and 2. Only the Ti and Ag backscattered signals are
displayed, and the Ti signal in Fig. 1 shows clearly that the
500 °C anneal causes Ti to segregate at the surface ͑see peak
labeled ‘‘Ti surface’’͒. The RBS spectra in Fig. 2, where the
actual Ti signal has been enhanced by a factor of 3, show
that Ti in the alloy segregates to the surface and interface
upon annealing. AES and SIMS profiling show evidence for
TiN͑O͒ formation at the surface and Ti-oxide-silicide bilayer
formation at the interface. Based on the RBS, the surface
layers on the alloy correspond to 17 ͑450 °C anneal͒ and 23
Simultaneous preparation of both sides of a Cu film has
been accomplished by exposing the Cu–Ti bilayer or alloy
surface to an NH3 ambient.6,7 At reaction temperatures be-
tween 350 and 600 °C, Ti nitride, TiN͑O͒, is formed on the
a͒
Permanent address: Materials Science and Engineering Department, Cor-
nell University, Ithaca, NY 14853.
Appl. Phys. Lett. 68 (23), 3 June 1996
0003-6951/96/68(23)/3251/3/$10.00
© 1996 American Institute of Physics
3251

 Alford
Alford
 Adams, Daniel
Adams, Daniel
 Laursen
Laursen
 Manfred Ullrich
Manfred Ullrich