1036
R. Bashiri et al. / Inorganica Chimica Acta xxx (2009) xxx–xxx
ultrasonic irradiation (6, 12 and 18 W) were tested but appropriate
2. Experimental
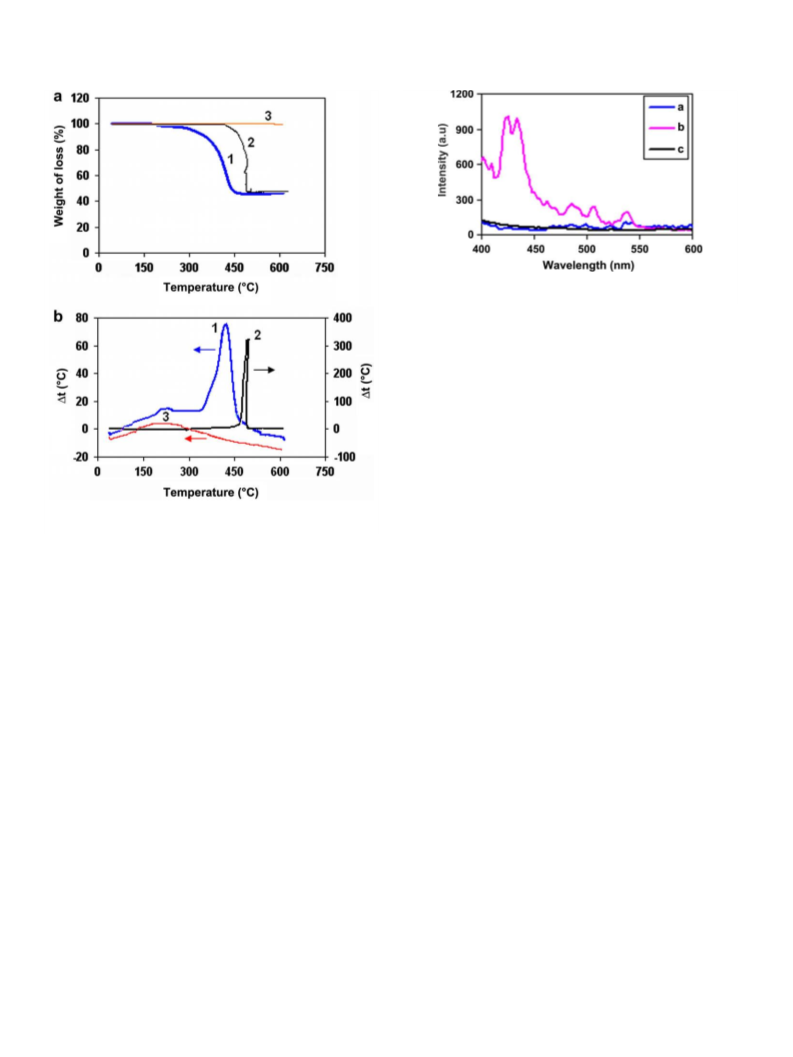
nano-size of compound 1 obtained under the mentioned condi-
tions. In order to obtain silver nanoparticles, precipitate of com-
2.1. Materials and physical techniques
pound
1 was calcinated at 500 °C in a furnace and static
atmosphere of air for 5 h.
All reagents for the synthesis and analysis were commercially
available and used as received. Double distilled water was used
to prepare aqueous solutions. A multiwave ultrasonic generator
(Sonicator_3000; Misonix, Inc., Farmingdale, NY, USA), equipped
2.4. Synthesis of silver nanoparticles by hydrothermal method
To prepare silver nanoparticles by hydrothermal method, pre-
cipitates of compound 1 (after reaction at room temperature with
the same concentration as used in sonochemical method) in 20 ml
double distilled water was posited in a hydrothermal bomb at
150 °C. After 48 h the bomb was immediately cooled to 4 °C and
the obtained precipitates were filtered, washed with double dis-
tilled water and then dried, neither d.p. and nor IR bands was
observed.
with
a converter/transducer and titanium oscillator (horn),
12.5 mm in diameter, operating at 20 kHz with a maximum power
output of 600 W, was used for the ultrasonic irradiation. Microa-
nalyses were carried out using a Heraeus CHN–O– Rapid analyzer.
Melting points were measured on an Electrothermal 9100 appara-
tus and are uncorrected. IR spectra were recorded using Perkin–El-
mer 597 and Nicolet 510P spectrophotometers. The thermal
behavior was measured with an PL-STA 1500 apparatus. The lumi-
nescent properties were investigated with a Shimadzu RF-5000
spectrofluorophotometer. Crystallographic measurements were
made using a Bruker APEX area-detector diffractometer. The inten-
3. Results and discussion
sity data were collected using graphite monochromataed Mo K
a
The Scheme 1 shows the reaction between silver(I) nitrate and
K2SB by three different methods.
radiation. The structure was solved by direct methods and refined
by full-matrix least-squares techniques on F2. Structure solution
and refinement was accomplished using SHELXL-97 program pack-
ages [23]. The molecular structure plot and simulated XRD powder
pattern based on single crystal data were prepared using MERCURY
software [24]. X-ray powder diffraction (XRD) measurements were
performed using a Philips X’pert diffractometer with mono chro-
Single crystal X-ray analysis reveals that compound [Ag(l8
-
SB)2]n (1) crystallizes in an orthorhombic setting with space group
of Fmm2.1 The phenolic oxygen atoms of the SB2À ligand act as bi-
donor bridging atoms where each oxygen atom of the Ph–O groups
link to two different silver(I) ion (Fig. 1a). The structure of com-
pound 1 may also be considered as a new type of coordination
polymer of Ag(I) consisting of one-dimensional linear chains con-
nected via phenolic bridging groups, running parallel to the b axis.
The Ag atoms in this compound are involved in an g2 interaction
with two Cph atoms of neighboring molecules (Fig. 1b). Thus, the
Ag atoms in compound 1 are linked to two carbon atoms of phenyl
moieties of SB2À with distances Ag–C2i (i: x, y + 1/2, z + 1/2) = Ag–
C2iv (iv: Àx + 1/2, Ày, z + 1/2) of 2.426(5) Å. Hence, the AgI coordi-
nation sphere is augmented to four and rather than exhibiting a
AgO2 coordination sphere, the compound can be considered to
have a bihapto (O2C2Ag) center with coordination number of four
(Fig. 1a). Ag–C bond lengths in other compounds with similar g2
Ag–C interactions range from 2.40 to 2.70 Å [25–28], for example
in the [Ag(benzene)ClO4] are 2.496 and 2.634 Å [29]. Some other
Ag(I) polycyclic aromatic polymeric complexes containing of Ag–
C(sp2) bond report mean Ag–arene distances of 2.82–3.37 Å [30–
35]. The dihedral angle formed by Ag–C2–C3 and C3–C2–C1 is
105.9(2)° and also the dihedral angle of C2i(i: x, Ày + 1, z)–C1–
C2–C3 is 5.3(8)°, indicating that the silver atoms interact with
matized Cu Ka radiation. The crystallite sizes of selected samples
were estimated using the Paul Scherrer method. The samples were
characterized with a scanning electron microscope with gold
coating.
2.2. Synthesis of [Ag2(
l
8-SB)]n (1) and preparation of its single crystals
8-SB)]n, 1 mmol (0.250 g)
To isolate the single crystals of [Ag2(
l
of 4-[(4-hydroxyphenyl)sulfonyl]-1-benzenol (H2SB) was dissolved
in 20 ml acetonitrile and was heated and stirred with solution of
2 mmol (0.114 g) KOH in 5 ml H2O for an hour. After deprotonation
of the ligand in this process, the solution was cooled to room tem-
perature and then a solution of 2 mmol (0.340 g) AgNO3 in 5 ml
acetonitrile was added to the mixture and was stirred for an hour.
After filtering it was allowed to stand in darkness at room temper-
ature to evaporate for several days to obtain suitable crystals. The
crystals were washed with acetone and air dried, d.p. >370 °C,
Yield: 0.255 g (55%), (Anal. Calc. for C12H8Ag2O4S: C, 31.03; H,
1.72. Found: C, 30.01; H, 1.79%). IR (selected bands; in cmÀ1):
552 s, 696 w, 723 w, 835 m, 1094 s, 1138 s, 1217 m, 1317 s,
1400 s, 1565 s, 2930 w, 3050 w.
the
p electrons of the aromatic rings. The individual polymeric
chains are almost parallel to each other and further bridged via
the Ag–C bonds, resulting in a three-dimensional framework as
shown in Fig. 1b. The carbon atom that connected to the –SO2
group is disordered over two positions in a 1:1 ratio, depending
on the location of the C@C double bond, and so is the adjacent
SO2 group. The following restraints were applied for the disordered
atoms: SIMU and DELU restraints for C1 to C4 and S1, O2 and O3.
ISOR restraints for C3, C4 (standard deviation 0.01) and S1, O2 and
O3 (standard deviation 0.002). The S@O distances were restraint to
be the same within a standard deviation of 0.02. Each SB2À anion in
compound 1 acts as a eight donating bridging ligand via two O-
2.3. Synthesis of [Ag2(l8-SB)]n (1) nanopowders by sonochemical
method
To prepare the nanopowders of [Ag2(l8-SB)]n by sonochemical
method, a high-density ultrasonic probe immersed directly into
the solution of K2SB (40 ml, 0.0125 M) in double distilled water,
then into this solution, a proper volume of AgNO3 aqueous solution
(10 ml, 0.1 M) was added in drop wise manner. The solution was
ultrasonically irradiated with the power of 18 W for an hour. The
obtained precipitates were filtered, subsequently washed with
double distilled water and then dried, d.p. = 272 °C (Anal. Calc. for
C12H8Ag2O4S: C, 31.03; H, 1.72. Found: C, 30.25; H, 1.67%). IR
(selected bands; in cmÀ1): 549 s, 694 w, 723 w, 835 m, 1091 s,
1131 s, 1220 m, 1312 s, 1402 s, 1566 s, 2931 w, 3052 w. Certainly
we should mention that different concentration of metal and
ligand solution (0.02, 0.05 and 0.08 M) with different power of
and four C-atoms, connecting eight AgI ions in a
l-8,6 mode (Fig.
1a). The phenolic groups of the SB2À ligand act as bridging group
1
The intensity data were collected within the range 2.26 6 h 6 28.25° for a crystal
of dimension 0.36 Â 0.36 Â 0.22 mm3. Crystal data: formula, C12H8Ag2O4S; Mr, 463.98
g/mol; orthorhombic system, space group Fmm2; a = 17.9958(18), b = 7.0921(7),
c = 9.2580(9) Å, V = 1181.6(2) Å3; Dcalc = 2.608 Mg/m3 (Z = 4); F(0 0 0) = 888;
R(wR) = 0.0346 (0.1005) for 821 reflections, with I > 2r(I); R(wR) = 0.0349 (0.1026)
for all data.

 Bashiri, Robabeh
Bashiri, Robabeh
 Akhbari, Kamran
Akhbari, Kamran
 Morsali, Ali
Morsali, Ali