ISSN 1070-4272, Russian Journal of Applied Chemistry, 2009, Vol. 82, No. 11, pp. 1944−1948. © Pleiades Publishing, Ltd., 2009.
Original Russian Text © O.L. Bersirova, V.S. Kublanovskii, 2009, published in Zhurnal Prikladnoi Khimii, 2009, Vol. 82, No. 11, pp. 1795−1799.
APPLIED ELECTROCHEMISTRY
AND CORROSION PROTECTION OF METALS
Crystalline Roughness as a Morphological Characteristic
of the Surface of Electroplated Silver Coatings
O. L. Bersirova and V. S. Kublanovskii
Vernadskii Institute of General and Inorganic Chemistry, National Academy of Sciences of Ukraine, Kiev, Ukraine
Received February 19, 2009
Abstract—Structure and morphology of electroplated silver coatings deposited from a borate-phosphate-carbonate
electrolyte were studied in relation to the current density and temperature. Average values of the crystalline
roughness were determined.
DOI: 10.1134/S1070427209100081
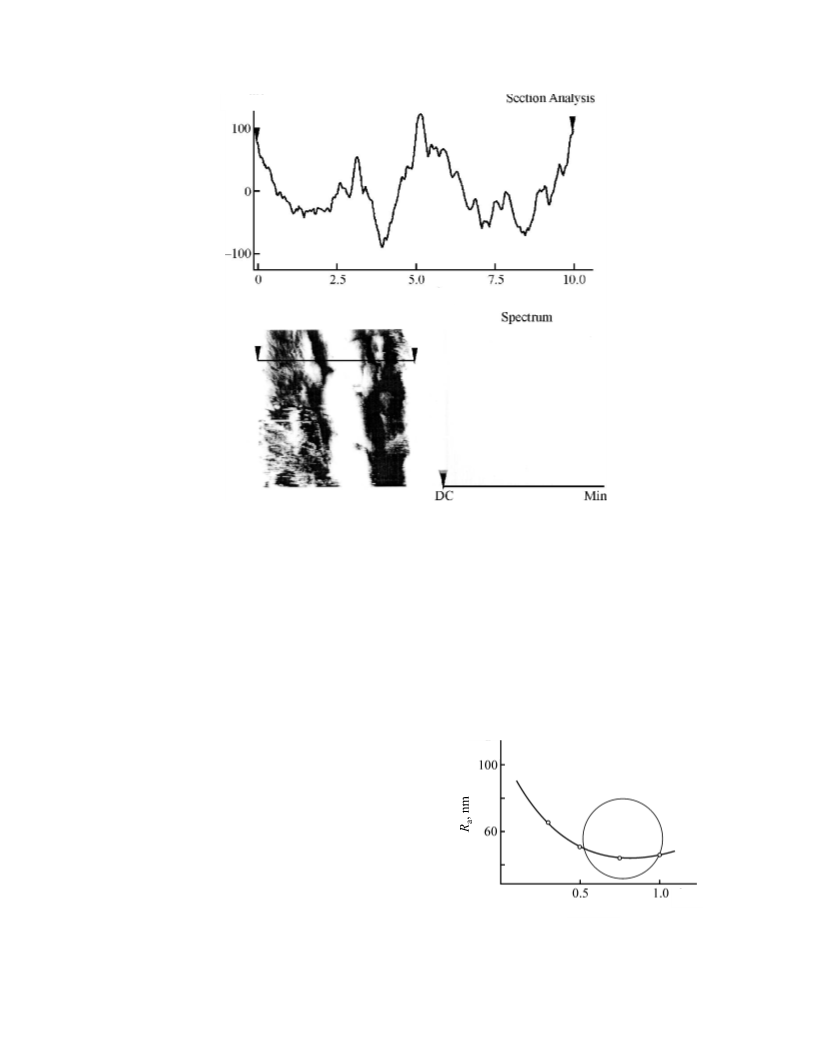
The roughness is evaluated using a number of special
microgeometric criteria, roughness parameters, the main
of which is the average roughness Ra, the arithmetic mean
deviation of the profile from a median inclined straight
line or plane (median line of the profile, median plane),
constructed using the least squares procedure.
To obtain prescribed necessary properties of
electroplated coatings, electroplaters try to select
conditions in which dense, smooth, and finely grained
deposits are obtained. By contrast, powder metallurgy
requires that a loose deposit not adhering to the substrate
should be produced. Apparently, the functionality of
electrolytic deposits is, in the end, controlled by the
structure and morphology, which primarily depend on
electrodeposition conditions: current density and overall
overvoltage of the process [1, 2]. The relationship between
the structure of a coating and its deposition conditions
is of utmost practical importance and has long been the
subject of extensive studies.
The aim of this study was to examine the structure and
morphology of silver coatings deposited from a borate-
phosphate-carbonate (BPC) electrolyte and, in particular,
the surface evenness (roughness) in relation to the process
kinetics.
EXPERIMENTAL
The quality of cathodic metal deposits is commonly
characterized by a set of physicomechanical and structural
properties of the coatings. To physicomechanical
properties are related the hardness, brittleness, adherence to
a substrate, porosity, and other properties. By the structure
is understood the size, shape, and orientation of the crystals
constituting the deposit. In the case of electrodeposited
metals, the geometric, physicomechanical, and structural
characteristics are interrelated. For example, the
roughness, which is a geometric characteristic of the
surface, is closely related to the deposit structure [3]. The
roughness is a quantitative parameter characterizing the
surface morphology [4]. It is a set of surface irregularities
with comparatively small steps, distinguished, e.g., with
a base line or a part of a plane. The surface roughness
can be evaluated both for the whole surface and for
a particular selected profile along a prescribed path.
In experiments, the structure and morphology of
electrolytic silver coatings deposited from a BPC
solution based on a cyanoargenate complex was studied
by scanning electron microscopy (SEM). Electrolyte
composition: (M): 0.75 KAg(CN)2 + 0.82 KH2PO4 +
0.41 H3BO3 + 0.15 K2CO3 + 0.69 KOH. The deposition
was performed at various polarizing current densities
(0.2–7.5 A dm–2) and temperatures of 20 and 60°C.
A polished brass plate with a working area of 2 cm2
served as a substrate. The morphology of the resulting
silver coatings was studied with a LEO-435VP scanning
electron microscope. The coating thickness of 20 μm was
calculated from the current efficiency and electrolysis
duration.
In all cases, the coatings obtained at current densities
of 0.3–1.5Adm–2 on a brass substrate were silvery white,
1944

 Bersirova
Bersirova
 Kublanovskii
Kublanovskii