APPLIED PHYSICS LETTERS 97, 153117 ͑2010͒
A. Hu,1,a͒ J. Y. Guo,2 H. Alarifi,1 G. Patane,3 Y. Zhou,1 G. Compagnini,3 and C. X. Xu2
1Department of Mechanical and Mechatronics Engineering, Centre for Advanced Material Joining,
University of Waterloo, 200 University Avenue West, Waterloo, Ontario N2L 3G1, Canada
2State Key Laboratory of Bioelectronics, Advanced Photonics Center, Southeast University, Nanjing 210096,
People’s Republic of China
3Dipartimento di Scienze Chimiche, Università di Catania, Viale A. Doria 6, Catania 95125, Italy
͑Received 24 August 2010; accepted 25 September 2010; published online 13 October 2010͒
We achieve robust bonding of Cu wires to Cu pads on polyimide with silver nanopaste cured at
373 K. The paste is prepared by simply condensing Ag nanoparticle ͑NP͒ solution via centrifuging.
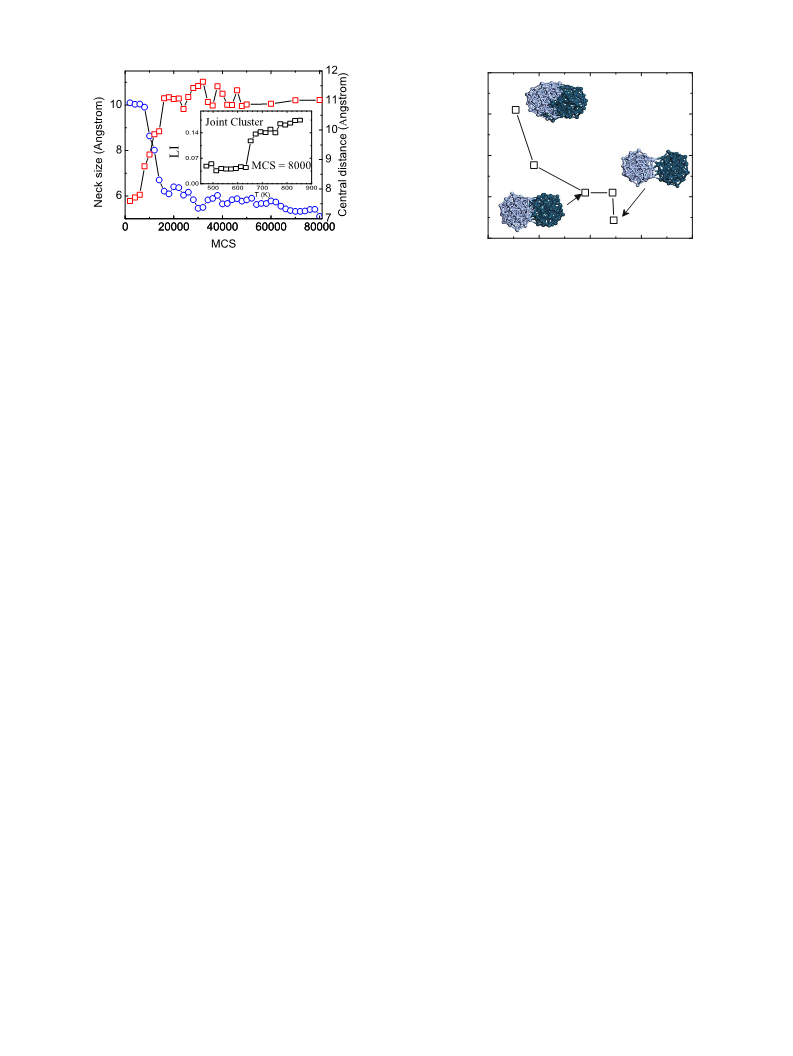
The bonding is formed by solid state sintering of Ag NPs through neck growth and direct metallic
bonding between clean Ag–Cu interfaces. Both experiment and Monte Carlo simulation confirm that
the melting point of joint clusters increases during sintering. This creates improved bonds for use at
an elevated operating temperature using Ag NPs. © 2010 American Institute of Physics.
There is increasing interest in developing low tempera-
ture interconnection processes for flexible electronics, in-
cluding flat-panel displays, organic electronics, and low-cost
These substrates, such as poly͑ethyleneterephthalate͒, poly-
imide, and paper, as well as some electronic components
based on organic groups or nanosized building blocks, can-
not maintain their functions and stabilities at a temperature
range of 473–573 K, which is required for melting lead-free
reflow.1,5–7 On the other hand, higher power chips, e.g., cen-
tral processing units, and automotive electronics near the en-
gine, frequently work at a temperature near 473 K.8,9 An
advanced bonding technology, which allows a low tempera-
ture curing but withstands a higher working temperature, is
thus desired.
Silver nanoparticle ͑NP͒ paste has been developed
through metallo-organic decomposition to avoid unstable
contact resistance and low working temperature of conven-
bonding and curing at low temperatures ranging from 473 to
573 K.3–5,9,11 In this paper, we report a silver paste directly
condensed from silver NP solution through centrifugation.
Robust bonding of Cu wires to Cu pads on polyimide has
been realized at a temperature of 373 K, sufficiently low for
flexible electronics. The experiments show that this low tem-
perature curing paste can work at a higher temperature than
the sintering temperature with enhanced bond strength.
A 1 mM solution of silver NPs was prepared by reduc-
tion of silver nitride with sodium citrate at a temperature of
about 363 K.12 Condensation was carried out with a centri-
fuge at 4000 rpm for 30 min. The concentration of silver in
the resulting paste was about 0.2 M. These Ag NPs were
deposited on silicon wafers for scanning electron microscopy
͑SEM͒ and x-ray photoelectron spectroscopy ͑XPS͒. The
bonding of Cu wires to Cu pads was conducted with Ag
paste at 5 MPa pressure at different curing temperatures.
Prior to bonding the surfaces of the Cu pads were cleaned in
ultrasonic bath with acetone. Transmission electron micros-
copy ͑TEM͒ has been performed on cross-sectional samples
cut with microtomy.
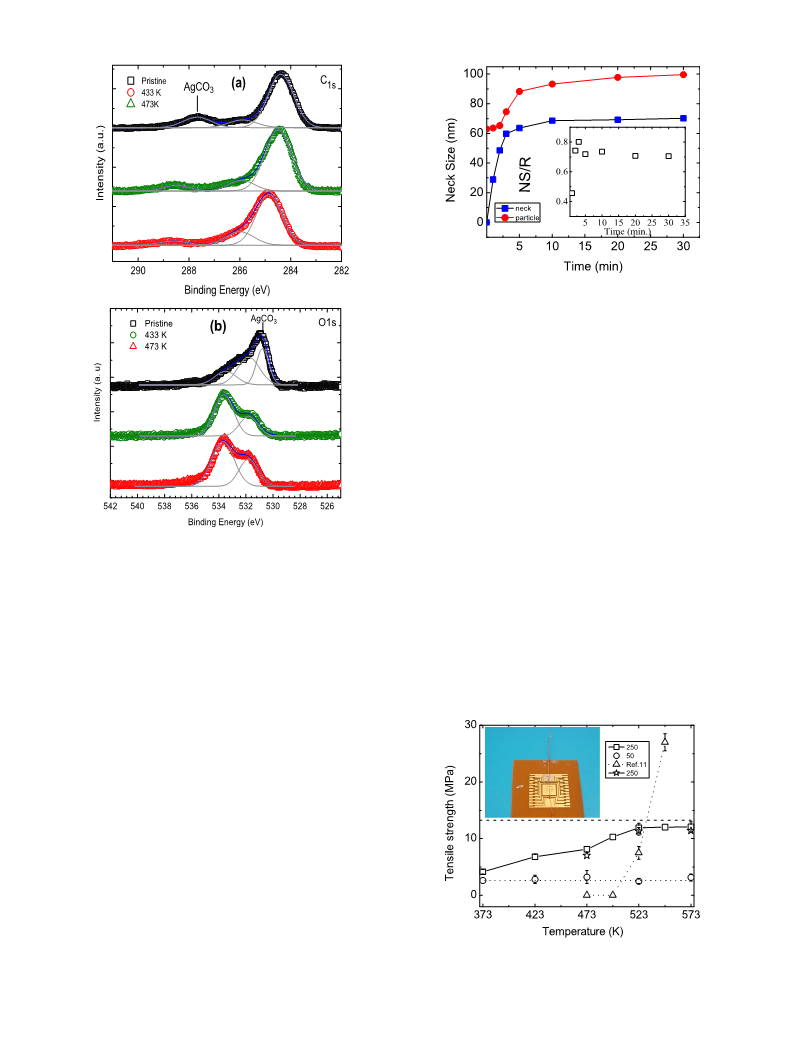
Figure 1͑a͒ shows a SEM micrograph of condensed Ag
NPs. Most of the Ag NPs are spherical. The mean size of the
NPs is about 50 nm. It is clearly visible that Ag NPs are still
well separated after condensation. This phenomenon is clari-
fied in detailed TEM investigation. Figure 1͑b͒ displays a
typical Ag NP deposited on formvar coated TEM copper
grids. An amorphous organic shell with a thickness of 1 nm
is clearly visible. It is reasonable to deduce that this shell
prevents the coalescence of Ag NPs. Shown in Fig. 1͑c͒ is an
SEM image of Ag NPs sintered at 523 K for 3 min. The
bonding is evident by the formation of the necked connec-
tions between Ag NPs. The bridging paths occur between
neighboring Ag NPs, orients arbitrarily, and eventually gen-
erates a three-dimensional ͑3D͒ connection network. These
joints are not a result of melting of the particles because
these NPs are expected to melt at a temperature ranging of
673–873 K based on the size effect,13 far above the present
(a)
(b)
Ag
1nm
200 nm
(c)
Cu (d)
Ag
5nm
100 nm
FIG. 1. ͑a͒ SEM images of condensed Ag NPs. ͑b͒ TEM images of a typical
Ag NP. Two lines indicate the boundaries of organic shells. ͑c͒ SEM image
of Ag NPs sintered at 250 °C for 3 min. ͑d͒ TEM images of the bonding
interfaces between Ag NPs and Cu pads at curing temperature of 523 K.
a͒
Electronic mail: a2hu@unwaterloo.ca.
0003-6951/2010/97͑15͒/153117/3/$30.00
97, 153117-1
© 2010 American Institute of Physics
69.166.47.134 On: Thu, 11 Dec 2014 10:15:53

 Hu
Hu
 Guo
Guo
 Alarifi
Alarifi
 Patane
Patane
 Zhou
Zhou
 Compagnini
Compagnini
 Xu
Xu