6
38
D. Hamani et al. / Journal of Solid State Chemistry 184 (2011) 637–643
IV
VI
structures, the Te and Te atoms lie in the positions typical to
sp-hybridization) is discussed. Subsequent to this point, the above
mentioned anomaly in the Raman spectrum of Cs Te 12 is con-
IV
VI
pure Te O
2
or Te O
3
crystalline lattices, i.e. in the apexes of
2
4
O
IV
essentially anisotropic Te O
4
polyhedra (disphenoids), or in the
coordination octahedra, respectively. In
analyzing such mixed structures, one can see that, on the one
sidered. To reproduce that experimental fact within the framework
of a standard model approximation, a high radial softness of the
VI
centers of isotropic Te O
6
+
electronic shell of the Cs ions was suggested, which is in line with
IV
hand, the Te –O covalent bonds (close to those in the TeO
molecule) are shorter (and thus stronger) than those making
2
its very high experimental polarizability. The concluding remarks
are given in the last section.
VI
up the Te O
6
octahedra which, in such a case, can hardly be
IV
2ꢀ
considered as tellurate anions; on the other hand, no [Te O
3
]
tellurite anions (or related fragments) are present there. Thus, no
objective criteria specifying those oxides as tellurates or tellurites
can be evidenced. So the question arises: which chemical evolu-
2. Experiment and calculations
To synthesize crystalline Cs
rial, TeO , was prepared by decomposing the commercial ortho-
telluric acid H TeO (Aldrich, 99.9%) at 550 1C for 24 h, and heated
in a stoichiometric proportion with Cs CO (Interchim, 99+ %) at
575 1C for 5 h in a platinum crucible under O atmosphere. The
2 4
Te O12 samples, the basic mate-
IV
VI
tion would undergo a Te O
atoms, brought by classical modifiers like Cs
2
–Te O
3
complex oxide when extra O
O, are added to it? In
2
2
6
6
other words, would this compound be transformed into a tellurite
structure or into a tellurate one? At this point, it can be recalled
2
3
2
that in the initial publication on the structure of Cs
2
Te
4
O
12 [1],
synthesized compound was identified from the X-ray diffraction
data (powder X-ray Siemens D5000 diffractometer, Bragg–
this compound was classified as ‘‘tellurite–tellurate’’. However, no
IV
2ꢀ
[
Te O
3
]
tellurite anions can be found in its lattice. To make
Brentano
recorded in the 15–980 cm
y–2y, Cu Ka radiations). The Raman spectrum was
ꢀ
1
more extended comment on this point , we wish to notice that, as
a rule (always to our knowledge), the formation of various forms
of tellurium dioxides is necessarily based on the 5s–5p hybridiza-
range using a Jobin–Yvon spectro-
+
meter (64000 model) equipped with a Kr laser (647.0 nm
exciting line) and a CCD detector operating in a backscattering
geometry. For this, a good signal/noise ratio required two scans
(during 240 s per scan). The sample focalization was done
through a microscope (ꢁ 50), and the diameter of the laser spot
IV
tion inside the Te atoms, which minimizes the electron energy.
2
As a result, 5s lone electron pair centers are displaced from their
former positions coinciding with the atomic centers, and lie aside
IV
those atoms. Consequently, covalent Te –O bonds coming from a
focused on the samples was about 1 mm. Measurements were
IV
given Te atom are always pointed away from its lone pair thus
performed at a low power (o200 mW) to avoid the deterioration
ꢀ
1
causing the coordination polyhedron to be strongly anisotropic.
of the sample. The spectral resolution was about 2.5 cm
at the
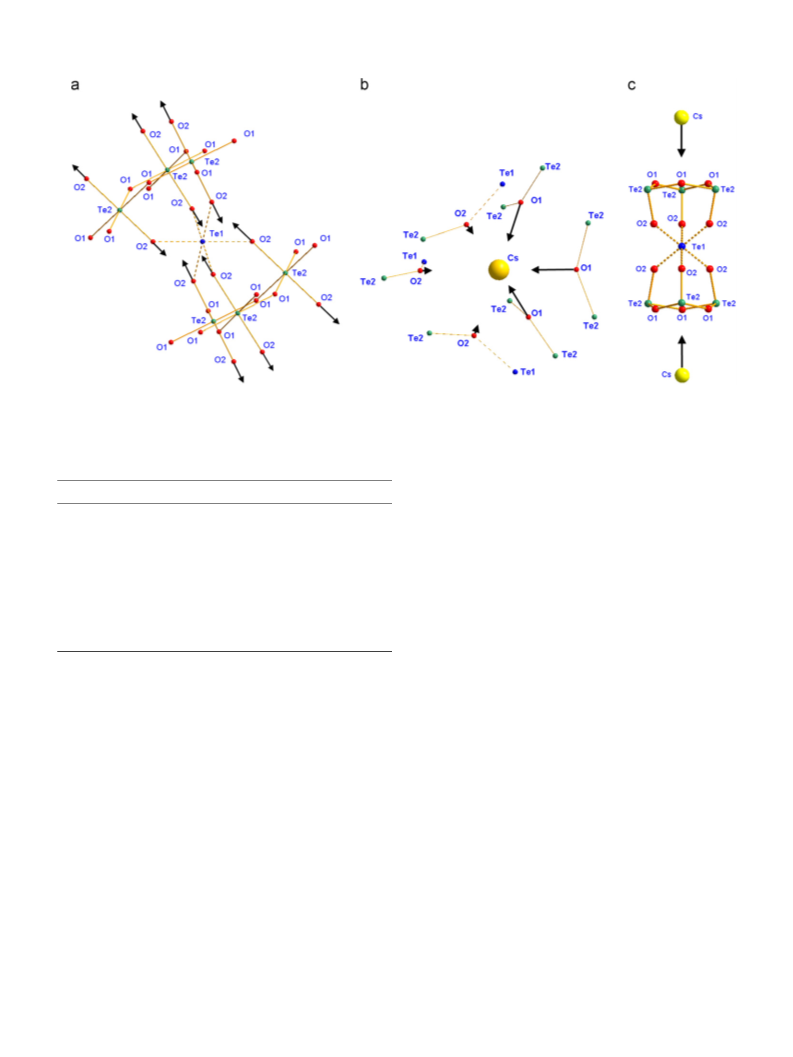
Crystalline Cs
2
Te
4
O
12 is the only exception. In this structure, each
exciting line. The infrared spectrum was scanned on a Fourier
transform spectrophotometer (Nicolet 6700) using the CsI pressed
pellet technique in a range 150–1000 cm . It was recorded in
the absorbance mode. The first-principles calculations were
performed to obtain self-consistent information about the struc-
ture, frequencies, symmetry, eigenvectors and IR intensities (but
IV
Te atom occupies the highest-symmetry (D3d) position, and is
isotropically surrounded by six oxygen atoms distanced from it by
ꢀ
1
˚
2
.104 A, thus forming a highly symmetric and weakly bound
IV
Te O
6
polyhedron existing in no other compound. One further
amazing point can be found in the Raman spectrum of Cs
In its lattice, half of oxygen atoms forms symmetric Te –O–Te
bridges which, geometrically, are similar to the Te –O–Te
bridges in the Te O
2
VI
Te
4
O
12
.
VI
not Raman intensities) of the zone center vibrations of Cs
as well as about the Te–O interatomic overlap population in
various TeO - and TeO -based compounds using the same ab
2 4 12
Te O ,
VI
VI
VI
VI
3
lattice. Since the Raman spectrum of Te O
3
2
3
sym
is absolutely dominated by the
n
TeVI–O–TeVI vibrational mode lying
[3], it can be thought that the homolog of that
mode should be readily revealed in the Raman spectrum of
initio routine. The computational technique was based on the
density functional theory (DFT) using the Becke’s three-parameter
hybrid nonlocal exchange functional [5] combined with the
Lee–Yang–Parr gradient-corrected correlation functional B3LYP [6]
through the CRYSTAL06 software [7,8]. This method has already
been extensively used and proved to be reliable for description of
crystalline oxide compounds with regard to structural and energy
characteristics [9,10]. The lattice geometry was preliminary
optimized, which was particularly important here for the calcula-
tions of vibrational frequencies as CRYSTAL06 uses the analytical
ꢀ
1
near 330 cm
ꢀ
1
Cs
2
Te
4
O
12. However, no strong bands below 600 cm
are seen
in that spectrum (see below), thus wrongly indicating a ‘‘bridge-
less’’ character of that compound. We wish to underline that, as a
rule, the Raman spectroscopy is an efficient instrument for
studying structural organization of ionic-covalent oxide crystals,
glasses or ceramic compositions, which is of special importance
when direct (X-ray diffraction) measurements are not possible or
informative. Therefore, the surprising situation in the spectrum of
first derivative of the total energy to construct the
G-point mass-
Cs
2
Te
4
O
12 may be generally instructive for the Raman spectro-
weighted Hessian matrix. Crystal frequencies were computed by
diagonalising this matrix. The atomic centers have been charac-
terized by all electrons basis sets, namely, 976631-311G* for Te,
6-31d1 for O [11] and 9766331-311G for Cs [12]. The k-point
sampling was chosen to be 32 points in the irreducible part of the
Brillouin zone. The LADY program (recent version of CRYME [13])
was additionally used to perform an empiric lattice-dynamical
model treatment allowing us to calculate the Raman intensities of
the vibrations via the Bond Polarizability Model (BPM) [14] in
which Raman intensities are classically described as related to the
polarizability variations of the vibrating system. The lattice
potential function was described by a Valence Force Field (VFF)
approximation whose parameters were taken from [15]. As it is
implied in BPM, the polarizability of our compound was consid-
ered as the sum of the Te–O ionic–covalent chemical bond
polarizabilities. The bonds were characterized by the three para-
meters, namely, longitudinal and transversal bond polarizability
scopy practice and methodology. So, when analyzing the struc-
tural organization and vibrational properties (i.e. considering a
2 4
classic ‘‘structure-spectra’’ problem) of the Cs Te O12 lattice, one
would be faced with surprising particularities in either case, and
their understanding can be of fundamental crystal chemistry
interest as well as of practical importance. In this paper, we wished
to clarify the origin of the both above mentioned ‘‘anomalies’’ by
analyzing the experimental evidence jointly with the ab initio and
lattice-dynamical model calculation data on the structure, vibra-
tional spectra and electronic organization of the Cs
comparing them with those obtained for TeO and
The paper is organized as follows. The details of experimental and
computational procedures are exposed in the next section, after
which the experimental and theoretical calculation results are
presented. The sections which follow them contain their analysis
2 4
Te O12 lattice, in
3
a
-TeO lattices.
2
IV
and discussion. First the chemical factor causing the Te atoms
to keep their 5s atomic orbitals intact (i.e. without undergoing the
0
0
derivatives (
a
l
and
t
a , respectively), complemented by parameter

 Hamani, David
Hamani, David
 Mirgorodsky, Andrei
Mirgorodsky, Andrei
 Masson, Olivier
Masson, Olivier
 Merle-Mejean, Therese
Merle-Mejean, Therese
 Colas, Maggy
Colas, Maggy
 Smirnov, Mikhael
Smirnov, Mikhael
 Thomas, Philippe
Thomas, Philippe