293
microwave irradiation is proper contact time. For normal drying
processes, the solvent is heated by conduction and convection,
which typically results in large temperature gradients within the
solvent and catalyst support [10]. For the ultrasonic-assisted tech-
nique, the ultrasound wave enhances good mass transport and
even thermal distribution throughout the impregnation solution
and the pores of the catalyst support using the cavitation phe-
nomenon typical of ultrasonic methods. Other desirable physical
nique can assist catalyst synthesis by modifying the shape of the
supported metal to be quasi-spherical in morphology. Moreover,
the catalyst preparation time can be reduced by this technique
[11–13]. For the ultrasonic-assisted technique, the ultrasound wave
improves mass transport, emulsification, thermal heating, and a
variety of support effects by the phenomenon of cavitation which
involves formation, growth and collapse of bubbles in the liq-
uid medium. Consequently, catalyst particle diameters may be
decreased, surface defects can be introduced on the particle surface,
and the surface area of the support may be increased [14,15]. In the
present work, Au/C catalysts having high Au dispersion have been
synthesized by microwave-assisted and ultrasonic wave-assisted
techniques, and their catalytic activities for acetylene hydrochlo-
rination have been studied and compared to catalysts prepared
by typical incipient wetness methods. Finally, the effects of gold
weight loading and pretreatment options on the catalyst perfor-
mance were also investigated.
SCCM total flow) and a temperature ramp rate of 5 ◦C/min from
25 ◦C to 500 ◦C. Gold weight loadings were determined by atomic
absorption (AAnalyst 400; PerkinElmer) after digestion of the Au/C
catalyst in aqua regia at 110 ◦C for 4 h and then filtering of the solid
catalyst from the solution containing the Au salt. Finally, the BET
surface areas of the supported Au catalysts were determined using
a Micromeritrics ASAP 2020. The measurements were performed
by degassing of the sample at 120 ◦C for 12 h prior to using N2
as the adsorbate at −196 ◦C. The specific surface area was calcu-
lated based on the Brunauer–Emmett–Teller (BET) method while
the pore volume, average pore diameter, and pore size distribution
were calculated using Barrett–Joyner–Halenda (BJH) desorption
branch analysis and t-plot micropore area analysis.
Acetylene hydrochlorination using the supported Au catalysts
was carried out in a fixed bed reactor (Pyrex glass tube, 3/8ꢀꢀ OD).
The C2H2 feed gas was treated to remove acetone inhibitor by pass-
ing through a molecular sieve 5A trap and then added along with
He diluent to a flow of anhydrous HCl (Praxair, 99.999% HCl purity)
before feeding to the reactor. The following reaction protocol was
used for all catalysts. The reactor loaded with fresh catalyst was
dried in flowing helium at 120 ◦C for 30 min to remove residual
H2O and then pre-chlorinated using a flow of He and HCl (1: 1
ratio) at 180 ◦C for 1 h. Acetylene was then added at a desired flow
to give a feed composition of C2H2: HCl: He = 1.0: 1.1: 1.0 at an ini-
tial reaction temperature of 180 ◦C. The exit gas from the reactor
was passed through a temperature-regulated scrubber (15 ◦C) con-
taining 5 M NaOH solution to absorb unreacted HCl. The gaseous
products were analyzed by an on-line gas chromatograph (model
HP5890 with FID detector using a capillary column of Agilent J&W
HP-Plot Poraplot Q column). Acetylene conversion and VCM selec-
tivity were calculated by mass balance methods. The fractional
conversion of C2H2 was determined using the standard formalism
of (C2H2(in) − C2H2(out))/C2H2(in) and VCM selectivity was calculated
from the amount of VCM formed/amount of total products formed.
Since only C2 products were observed, no normalization to VCM
was required.
2. Experimental
A conventional Au/C catalyst was first prepared by incipient
wetness impregnation using concentrated aqua regia as a solvent
[16]. The carbon support (coconut shell-based and supplied by Car-
bokarn) was initially washed with dilute, aqueous HCl (1 M) (Qrec
HCl 37%) to eliminate impurities. The carbon was washed thor-
oughly by distilled water and then filtered and dried overnight at
110 ◦C. A solution of HAuCl4·xH2O (Sigma–Aldrich: 50% gold assay)
in concentrated >aqua regia solvent (mixture of HCl (Qrec HCl 37%):
HNO3 (Qrec HNO3 67%) = 3:1) was added drop-wise to the activated
carbon while stirring about half an hour. Then the impregnated cat-
prepared by ultrasonically-agitating the dissolved HAuCl4 in aqua
regia solution and carbon support in an ultrasonic bath (Crest Ultra-
sonic Model: 275HTAE 42–45 kHz) and maintaining at 45–50 ◦C
for 6 h [17]. The microwave-assisted technique (MW sample) was
applied during the drying stage of the impregnated carbon support
microwave-assisted preparation by using a microwave oven (Sanyo
EMS 1063s 800W compact microwave) for 2 min.
3. Results and discussion
3.1. Effect of catalyst preparation technique
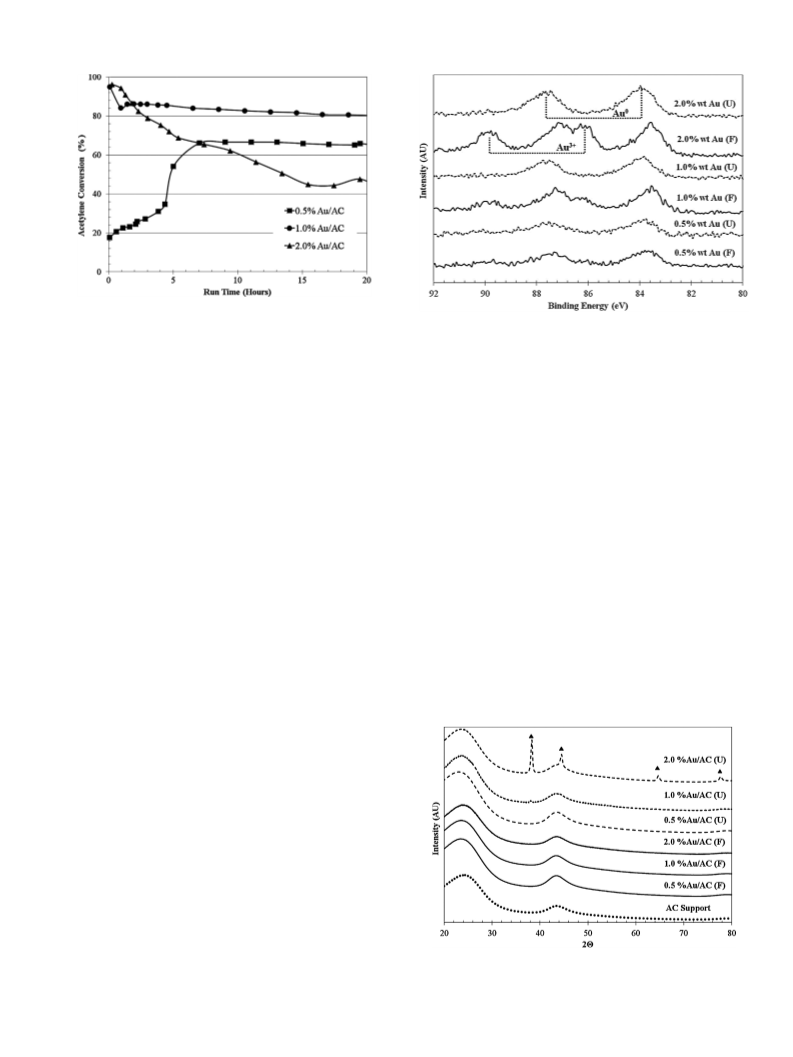
The comparative X-ray diffraction (XRD) patterns of freshly-
prepared (no pretreatments) conventional (CI), ultrasonic-assisted
None of the catalysts shows peaks indicative of any Au0 or gold salt
species, indicating that all Au species are present in either amor-
phous or very small, highly dispersed particles. Thus, Au0 particle
sizes must be <4 nm or most of the Au exists as non-crystalline
Au3+ species [18]. For Au0/C catalysts, the typical peaks of Au0
at 2ꢀ = 38.2, 44.4, 64.7 and 77.7◦, assigned to the diffraction lines
of (1 1 1), (2 0 0), (2 2 0) and (3 1 1) facets of gold, respectively, are
expected [19].
The methods of catalyst preparation also affect the physical
properties of the carbon support. Support analyses data are sum-
marized in Table 1. Impregnation of the Au salts lowers the surface
area of the support, as expected. The effect is most pronounced for
the CI sample. Both MW and UL effects decrease the loss of sur-
face and also maintain the more desirable larger pore diameters of
the carbon micropores. This phenomenon of the UL sample is likely
due to the tiny bubbles formed by the vibrations which collapse and
then re-form from the cavitation phenomena. This occurs when the
expansion speed of bubbles is high enough and the inertial power is
2.2. Catalyst characterization
X-ray diffraction (XRD) was used to investigate the crystalline
characteristics of the catalysts. The XRD spectra of catalysts were
measured by a Rigaku MiniflexII with a Texultra detector using Cu
K␣ radiation over the 2ꢀ range from 20◦ to 80◦. The analysis of
elemental compositions and oxidation states of catalyst surfaces
was determined by X-ray photoelectron spectroscopy (XPS) using
a Kratos Axis Ultra DLD instrument equipped with a monochro-
mated Al K␣ X-ray source and hemispherical analyzer capable of
an energy resolution of 0.5 eV. Temperature-programmed reduc-
tion (TPR) was used for determining the reduction temperature of
Au3+, using a Micromeritics Chemisorb 2920, with 10% H2 in Ar (50

 Wittanadecha
Wittanadecha
 Laosiripojana
Laosiripojana
 Ketcong
Ketcong
 Ningnuek
Ningnuek
 Praserthdam
Praserthdam
 Monnier
Monnier
 Assabumrungrat
Assabumrungrat