EXPERIMENTAL
1
The H NMR spectra were recorded on a Varian Gemini 300 (300 MHz) spectrometer, internal standard
1
3
was TMS. The C NMR spectra were obtained on a Varian Mercury 400 (100 MHz) spectrometer using
J-modulation (APT). The electronic absorption spectra were recorded on a Shimadzu UV 3100 spectrometer.
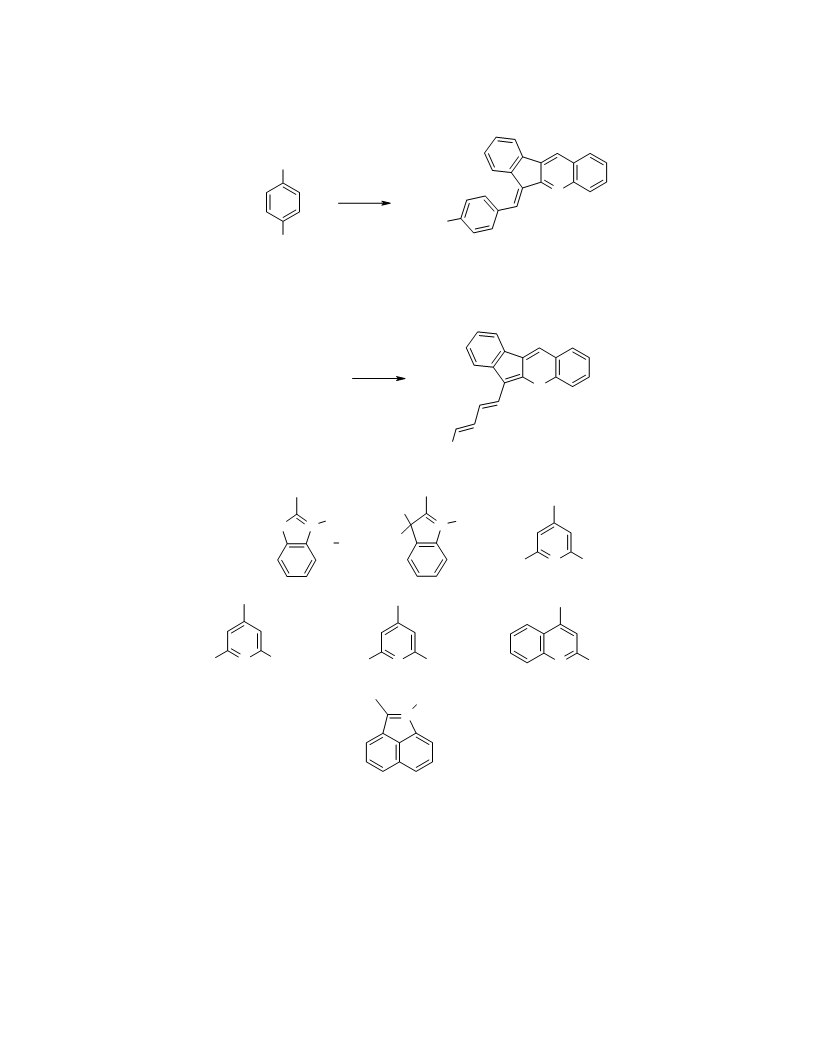
Indeno[2,1-b]chromene (1) was synthesized by the known procedure of [14] from 2-indanol and
1
salicylic aldehyde. H NMR spectrum (DMSO-d ), δ, ppm (J, Hz): 6.33 (1H, s); 7.19 (1H, t, J = 8.4); 7.3-7.6
6
1
3
(
5H, m); 7.78 (1H, d, J = 7.5); 7.96 (1H, d, J = 7.5); 8.22 (1H, s, H-11). C NMR spectrum (DMSO-d ), δ, ppm:
6
9
7.4 (CH); 116.8 (CH); 120.0 (ipso); 120.2 (CH); 121.9 (CH); 122.2 (CH); 124.1 (CH); 124.4 (CH); 126.3
ipso); 129.2 (CH); 129.9 (CH); 130.1 (ipso); 131.4 (CH); 141.5 (ipso); 151.0 (ipso); 152.2 (ipso). Electronic
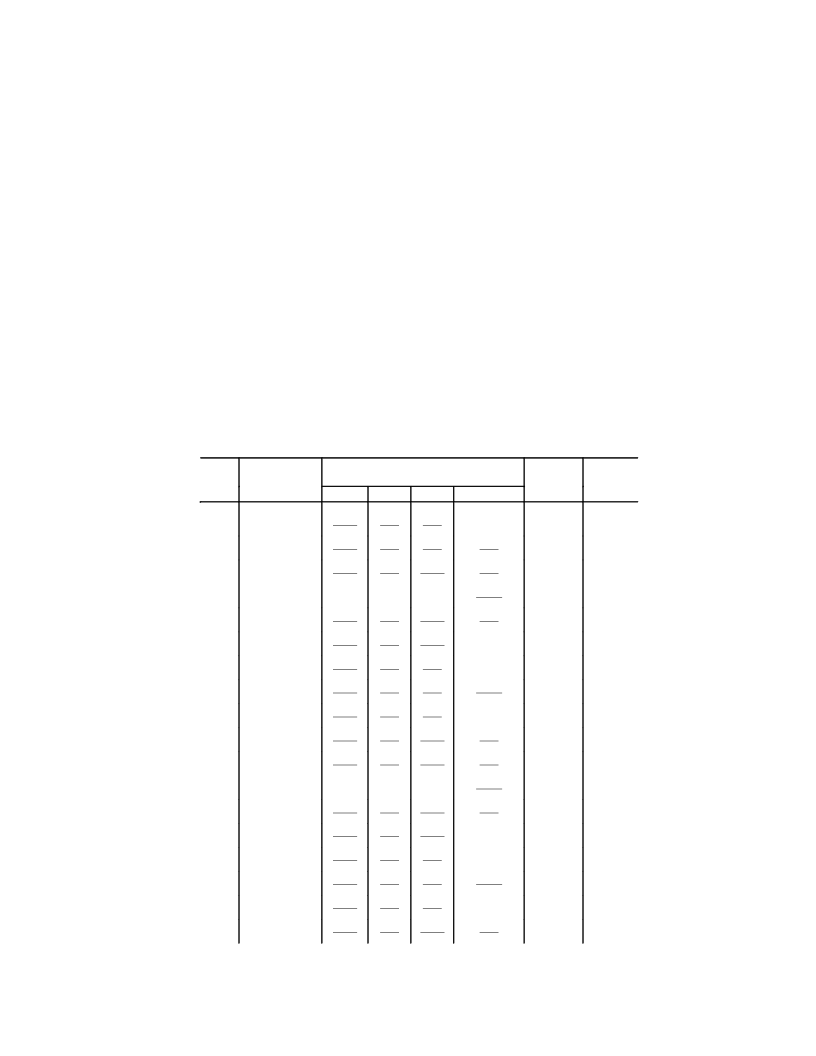
absorption spectrum (MeCN), λmax, nm (log ε): 466 (3.34), 337 (4.22).
H-Indeno[2,1-b]chromenium Trifluoroacetate (3). Indenochromene 1 (0.218 g, 1 mmol) was
(
6
dissolved in trifluoroacetic acid (15 ml) at 20°C. The product was not isolated, using the reaction mixture for
1
plotting spectra. H NMR spectrum (CF COOD), δ, ppm (J, Hz): 7.35 (2H, m); 7.41 (1H, t, J = 6.6); 7.79 (2H,
3
1
3
m); 8.06 (2H, m); 8.12 (1H, d, J = 8.4); 9.25 (1H, s, H-11). C NMR spectrum (CF COOD), δ, ppm: 110.0
3
(
CH); 113.0 (CH); 116.0 (ipso); 116.2 (CH); 120.2 (CH); 121.9 (CH); 122.2 (CH); 123.1 (CH); 123.7 (ipso);
1
26.7 (ipso); 127.0 (ipso); 130.0 (CH); 136.1 (CH); 147.9 (ipso); 183.1 (ipso). Electronic absorption spectrum
(
MeCN + excess HClO ), λmax, nm (log ε): 415 (4.09).
4
Indeno[2,1-b]chromene-6-carbaldehyde (4) was obtained by the method of [13] from indenochromene
1
1
(4.36 g, 20 mmol) and POCl (3.98 g, 26 mmol) in DMF (30 ml). H NMR spectrum (DMSO-D ), δ, ppm
3
6
(
J, Hz): 7.33 (1H, t, J = 7.5); 7.47 (1H, t, J = 7.5); 7.55 (1H, m); 7.80 (2H, m); 7.98 (1H, d, J = 8.1); 8.09 (2H, t,
J = 9.0); 8.77 (1H, s); 10.37 (1H, s).
-(Indeno[2,1-b]chromen-6-yl)-2-propenal (5). A solution of POCl (3.98 g, 26 mmol) in MeCN
3
3
(
10 ml) was added dropwise with stirring to a solution of 3-dimethylamino-2-propenal (2.57 g, 26 mmol) in
MeCN (70 ml) cooled in an icebath. The mixture obtained was stirred for 15 min, then indenochromene 1
4.36 g, 20 mmol) was added to it. The reaction mixture was stirred for 2 h, then left to stand for 24 h at room
(
temperature. The MeCN was evaporated in vacuum and the residue rubbed under cold 10% NaOH solution. The
solid was filtered off, washed several times with water, and recrystallized from MeCN. Product 5 (5.22 g, 96%)
1
3
3
was obtained. H NMR spectrum (DMSO-d ), δ, ppm (J, Hz): 6.94 (1H, dd, J = 15.6, J = 7.8); 7.32 (1H, t,
6
J = 6.3); 7.49 (2H, m); 7.65-7.85 (3H, m); 7.85-8.07 (3H, m); 8.54 (1H, s); 9.68 (1H, d, J = 7.8).
6
-{3-(Indeno[2,1-b]chromen-6-yl)-2-propenylidene}-6H-indeno[2,1-b]chromenium Perchlorate (6).
A mixture of aldehyde 5 (0.272 g, 1 mmol) and indenochromene 1 (0.218 g, 1 mmol) in acetic anhydride (10 ml)
was heated to boiling, then cooled, 70% HClO (0.1 ml) was added, and the mixture boiled for 5 min. The solid
4
precipitated after cooling was filtered off, washed with ether, and recrystallized from 2-nitropropane.
6
-[1-(4-Dimethylaminophenyl)methinyl]-6H-indeno[2,1-b]chromenium Perchlorate (8) was
obtained analogously to perchlorate 6 from indenochromene 1 (0.436 g, 2 mmol) and 4-dimethylamino-
benzaldehyde (0.298 g, 2 mmol), and was recrystallized from 2-nitropropane.
Cyanines 10a-g, 11a-g (General Method). A mixture of aldehyde 4 or 5 (1 mmol) and salts 9a-g
(
1 mmol) in acetic anhydride (10 ml) was boiled under reflux for 5 min. After cooling, solid products 10 or 11
were precipitated, and were filtered off, washed with ether, and recrystallized from 2-nitropropane
REFERENCES
1
2
3
.
.
.
H.-J. Timpe and A. V. El'tsov, Adv. Heterocycl. Chem., 33, 185 (1983).
Yu. N. Porshnev, V. A. Churkina, and M. I. Cherkashin, Usp. Khim., 56, 95 (1987).
A. Graness, J. Kleinschmidt, W. Triebel, A. Olszowski, and H. J. Timpe, J. Prakt. Chem., 323, 716
(
1981).
8
4

 Krotko
Krotko
 Fedotov
Fedotov
 Tolmachev
Tolmachev