3462
B.P. Baranwal et al. / Inorganica Chimica Acta 362 (2009) 3461–3464
Table 1
Analytical results for titanium(IV) complexes.
Reactantsa (g, mmol)
Product (colour) (% yield)
Found (calculated) %
Molecular
weight
Ti
Cl
C
H
[Ti(acac)Cl3] (4.82, 19.03) + C15H31COONa (5.32,
19.11)
[Ti(acac)Cl3] (1.35, 5.33) + C15H31COONa (2.98,
10.70)
[Ti(acac)Cl3] (2.70, 10.66) + C15H31COONa (8.96,
32.18)
[Ti(acac)Cl3] (3.38, 13.34) + C17H35COONa (4.1,
13.38)
[Ti(acac)Cl3] (2.34, 9.24) + C17H35COONa (5.68,
18.53)
[Ti(acac)Cl3] (2.6, 10.26) + C17H35COONa (9.48,
30.93)
[Ti(acac)Cl2(OOCC15H31)] (dark yellow)
(85) (1)
[Ti(acac)Cl(OOCC15H31)2] (light yellow)
(88) (2)
[Ti(acac)(OOCC15H31)3] (light yellow)
(82) (3)
[Ti(acac)Cl2(OOCC17H35)] (dark yellow)
(81) (4)
[Ti(acac)Cl(OOCC17H35)2] (light yellow)
(85) (5)
[Ti(acac)(OOCC17H35)3] (light yellow)
(89) (6)
10.10
(10.11)
6.98
(6.91)
5.21
(5.24)
9.46
(9.55)
6.35
14.80
(14.98)
5.05
53.40
(53.29)
64.30
(64.10)
69.65
(69.71)
55.20
(55.10)
65.75
(65.71)
71.00
8.10
487
(473)
679
(693)
931
(913)
486
(501)
735
(749)
984
(8.11)
10.08
(10.05)
11.05
(11.10)
8.40
(8.46)
10.40
(10.38)
11.30
(11.34)
(5.11)
14.00
(14.14)
4.60
(6.39)
4.80
(4.79)
(4.73)
(71.04)
(997)
a
Reflux about 2–4 h.
2.3. Physico-chemical measurements
(antisymmetric) and (
msymOCO) (symmetric) vibrations of the car-
boxylate ions, respectively [20]. The differences between the sym-
Infrared spectra were recorded on a Varian 3100-FTIR Excalibur
series spectrophotometer using KBr discs. Molecular weights were
determined in a semi-micro ebulliometer (Gallenkamp) with a
thermistor sensing device. FAB mass was done on a JEOL SX 102/
DA-6000 mass spectrometer using m-nitrobenzyl alcohol (NBA)
as a matrix. 1H NMR spectra were recorded at 250.17 MHz on a
Bruker DPX 250 NMR spectrometer in CDCl3. Molar conductance
was measured on a century CC-601 digital conductivity meter at
10ꢀ2–10ꢀ3 molar concentrations in DMF. Elemental analyses (C,
H) were done on a Carlo-Erba 1108 elemental analyzer. Magnetic
moment was measured on a Gouy balance using Hg[Co(SCN)4] as
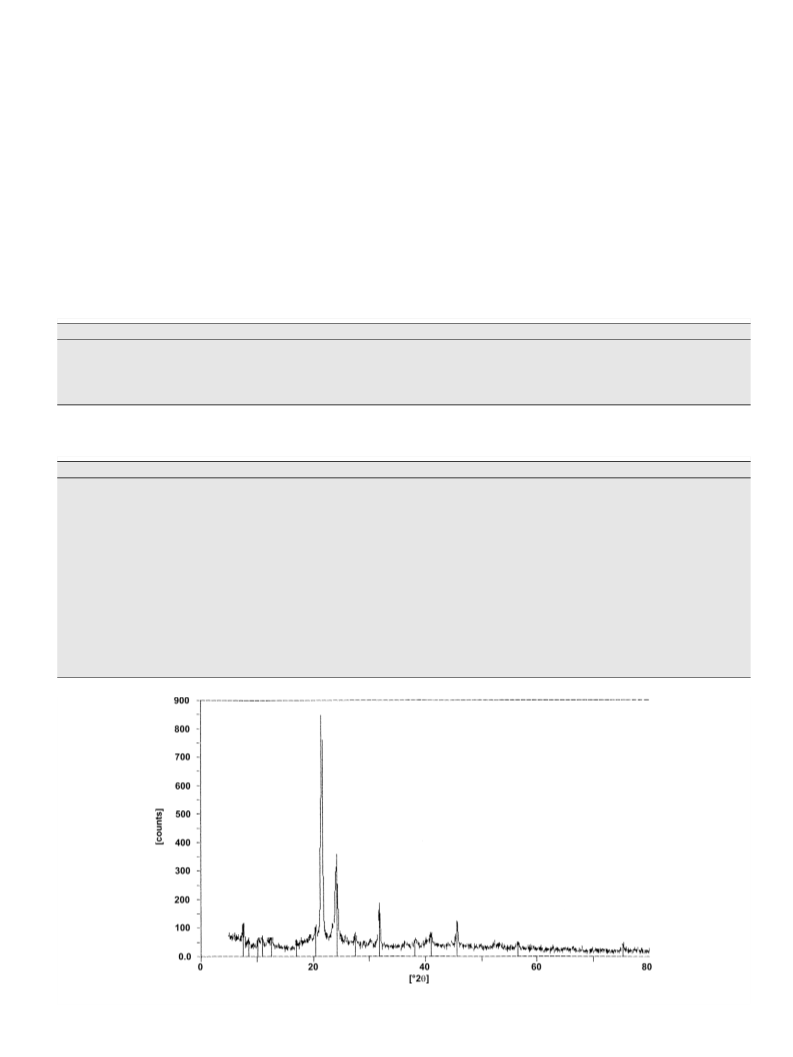
a calibrant. Powder X-ray diffraction data were collected on a PW
1710 diffractometer.
metric and antisymmetric stretches,
D
[
m
asymOCO ꢀ
msymOCO]
were found in the range 100–122 cmꢀ1 which indicated bidentate
chelating nature of carboxylate ligands [21]. In the infrared spectra
of the complexes, no band was found in 1626–1700 cmꢀ1 region
indicating the CO group of acetylacetone was not free in the com-
plexes. Two splitted new bands observed at 1615–1590 cmꢀ1
mC'O
and 1530–1515 cmꢀ1
mC'C indicated the chelating nature of ace-
tylacetonate ion in the complexes [22]. The bands around 670 and
470 cmꢀ1 could be assigned to ring deformation mTi–O and mTi–O
vibrations, respectively [21].
3.2. Hydrolytic stability of the complexes
Complex [Ti(acac)Cl3] is extremely moisture sensitive and
hydrolyses easily. The final product of hydrolysis is TiO2 [18]. How-
ever, complexes [Ti(acac)(Pal)3] and [Ti(acac)(Stea)3] exhibit a high
hydrolytic stability and are air stable for a longer time. This stabil-
ity was tested by dissolving the complexes in benzene and 0.01%
water is added. After stirring the contents for 12 h, no precipitate
was appeared even after keeping the contents open in to air for
2 months. In contrast, the complexes [Ti(acac)Cl2(Pal)] and [Ti
(acac)Cl2(Stea)] were precipitated while tested under same condi-
tions. Complexes [Ti(acac)Cl(Pal)2] and [Ti(acac)Cl(Stea)2] showed
slow precipitation after 7 days under the same conditions. This
clearly indicates that the stepwise removal of chloride ion by high-
er fatty acid anions is directly related to the hydrolytic stability of
the complexes. When all the chloride ions are removed, the com-
plexes become highly stable to hydrolysis. In other way we can
say that the substitution of chloride ions by oxygen donor long-
chain fatty acid anions is directly proportional to the hydrolytic
stability of the complexes. Therefore, the hydrolytic stability order
of the complexes was concluded as:
3. Results and discussion
Some mixed-ligand ternary and quaternary complexes of tita-
nium(IV) were synthesized by the stepwise substitutions of chlo-
ride ions of [Ti(acac)Cl3] by straight chain carboxylic acid anions:
Benzene
½TiðacacÞCl ꢁ þ nRCOONa
½TiðacacÞCl3ꢀnðOOCRÞ ꢁ þ nNaCl
!
3
n
Reflux=100 ꢂC
ð1Þ
where R = C15H31, Pal or C17H35, Stea; n = 1–3.
Benzene was chosen a solvent in these substitutions because
during the reaction there was formation of sodium chloride which
was insoluble in benzene in which the titanium(IV) complexes
were soluble. This made the separation easy. After substitution of
chloride ions, the colour of the complexes became lighter and the
complexes [Ti(acac)(OOCR)3] were of light yellow colour. Molar
conductance of the complexes at 10ꢀ2–10ꢀ3 molar concentrations
in DMF was obtained in the range 11–19
X
ꢀ1 cm2 molꢀ1 and the
½TiðacacÞCl2ðOOCRÞꢁ < ½TiðacacÞClðOOCRÞ2ꢁ < ½TiðacacÞðOOCRÞ3ꢁ
results indicated them to be non-electrolytes [19]. Magnetic mo-
ments of all the complexes at room temperature indicated them
to be diamagnetic, ascertaining titanium is present in 4+ oxidation
state with no unpaired electron.
1
2
3
3.3. 1H NMR spectra
In the 1H NMR spectrum of [Ti(acac)Cl2(OOCC15H31)] a triplet
appeared at d = 0.89 ppm (3H) corresponded to methyl protons of
fatty acid while a singlet corresponding to 26H of the 13-CH2
groups was observed at d = 1.26 ppm interpreted for palmitate
ion [–OOCCH2(CH2)13CH3] of the complex. A peak at d = 1.64 ppm
3.1. Infrared spectra
In infrared spectra of all the complexes O–H stretching vibra-
tions of carboxylic acid were found absent near 3400 cmꢀ1. The
bands at 1710 (CO stretching) and at 935 cmꢀ1 (OH deformation)
of free carboxylic acids were also absent in the spectra of the com-
plexes. Two strong bands were found in the range 1562–1530 cmꢀ1
(2H) indicated the
a-CH2 protons of palmitate ion. A singlet at
d = 2.06 ppm (6H) indicated the methyl protons of acac while the
methine proton of acac was observed as a singlet at d = 5.51 ppm
and 1440–1430 cmꢀ1 which could be assigned due to (
masymOCO)

 Baranwal, Balram P.
Baranwal, Balram P.
 Fatma, Talat
Fatma, Talat
 Singh, Alok K.
Singh, Alok K.
 Varma, Anand
Varma, Anand