Communication
ChemComm
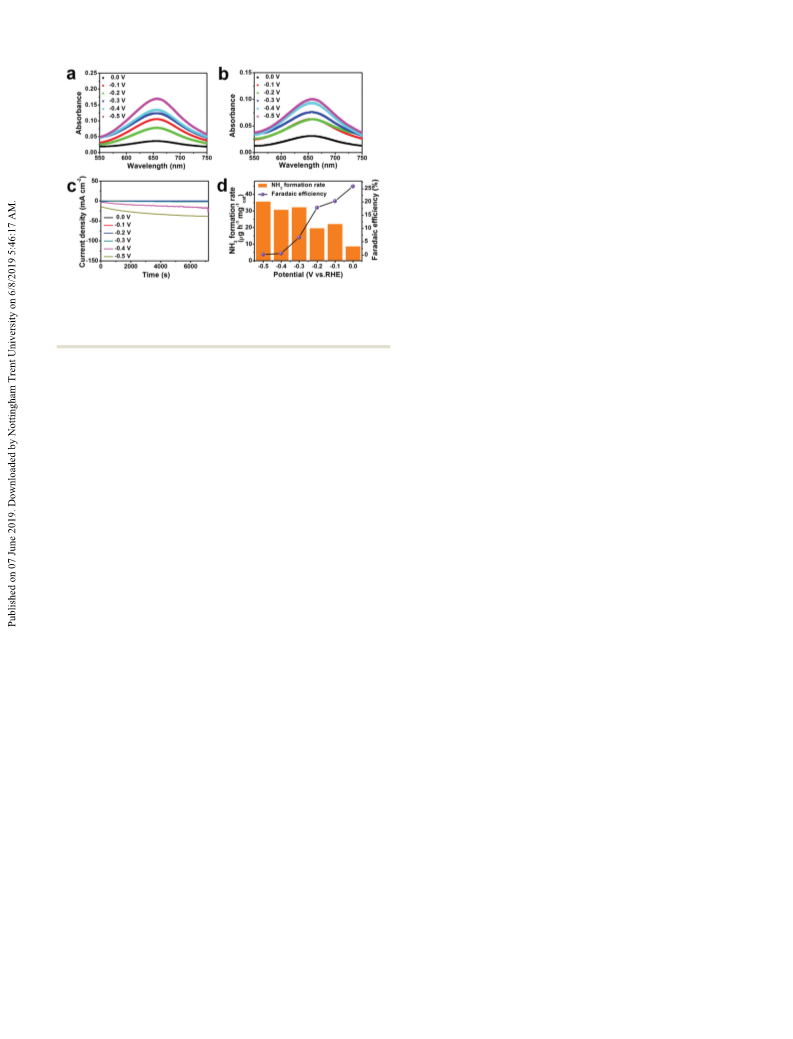
OV-rich a-Fe2O3 demonstrated excellent electroactivity for a N2
reduction reaction in an alkaline electrolyte with a NH3 formation
rate of 32.13 mg hÀ1 mgcatÀ1 and a faradaic efficiency of 6.63% at
À0.3 V as well as a good stability, outperforming all of the reported
iron oxide catalysts and most of the state-of-the-art catalysts. Our
task-specific ionic liquid strategy toward OV-rich metal oxides is
therefore of great interest for the rational design of advanced N2
fixation electrocatalysts.
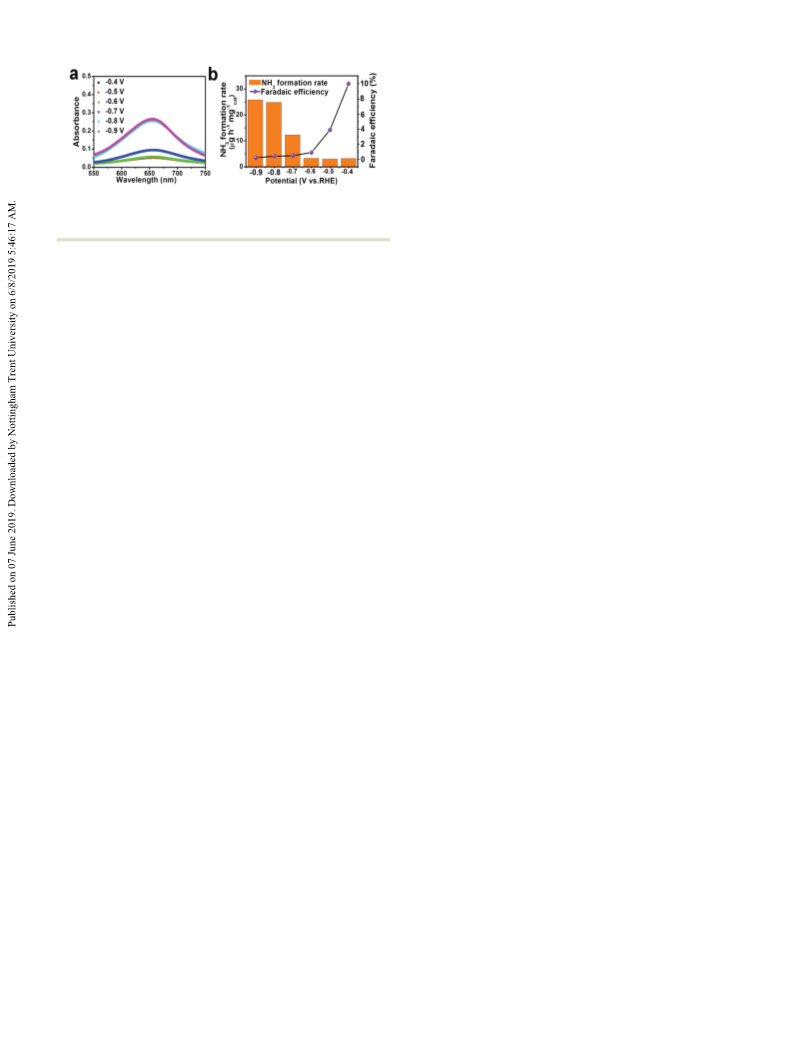
Fig. 4 Fe2O3–IL in 0.1 M Na2SO4 under the potentials from À0.4 V to
À0.9 V for 2 h: UV-Vis absorption spectra of NH3 (a); NH3 formation rates
and FEs (b).
This work was supported by the National Natural Science
Foundation of China (Grant No. 21673128).
Conflicts of interest
activation of N2. The as-prepared Fe2O3–IL possessed abundant OVs,
being beneficial to the NRR. Therefore, the integration of OVs into
Fe2O3–IL resulted in the superior electrochemical NRR activity.
We further performed NRR of Fe2O3–IL in neutral 0.1 M
Na2SO4 solution (Fig. 4). Fig. S21 and S22 (ESI†) gave the
calibration curves of NH3 and N2H4ÁH2O. Both the NH3 for-
mation rates and the corresponding FEs of Fe2O3–IL under
various potentials in Na2SO4 solution are shown in Table S3
(ESI†). From À0.4 V to À0.6 V (Fig. 4 and Fig. S23, ESI†), the
There are no conflicts to declare.
References
1 C. J. M. Van Der Ham, M. T. M. Koper and D. G. H. Hetterscheid,
Chem. Soc. Rev., 2014, 43, 5183–5191.
2 K. Honkala, A. Hellman, I. N. Remediakis, A. Logadottir, A. Carlsson,
S. Dahl, C. H. Christensen and J. K. Nørskov, Science, 2005, 307, 555–558.
3 C. Na and G. Zheng, Nano Res., 2018, 11, 2992–3008.
4 X. Cui, C. Tang and Q. Zhang, Adv. Energy Mater., 2018, 8, 1800369.
5 D. Bao, Q. Zhang, F. Meng, H. Zhong, M. Shi, Y. Zhang, J. Yan,
Q. Jiang and X. Zhang, Adv. Mater., 2017, 29, 1604799.
6 M. T. Nguyen, N. Seriani and R. Gebauer, Phys. Chem. Chem. Phys.,
2014, 17, 14317–14322.
7 Y. Abghoui, A. L. Garden, J. G. Howalt, T. Vegge and E. Skulason,
ACS Catal., 2016, 6, 635–646.
8 K. Kugler, M. Luhn, J. A. Schramm, K. Rahimi and M. Wessling,
Phys. Chem. Chem. Phys., 2015, 17, 3768–3782.
9 R. Zhang, Y. Zhang, X. Ren, G. Cui, A. M. Asiri, B. Zheng and X. Sun,
ACS Sustainable Chem. Eng., 2018, 6, 9545–9549.
10 X. Yang, J. Nash, J. Anibal, M. Dunwell, S. Kattel, E. Stavitski, K. Attenkofer,
J. G. Chen, Y. Yan and B. Xu, J. Am. Chem. Soc., 2018, 140, 13387–13391.
11 X. Zhang, R. Kong, H. Du, L. Xia and F. Qu, Chem. Commun., 2018,
54, 5323–5325.
À1
NH3 formation rates basically stayed around 3.00 mg hÀ1 mgcat
(2.45 Â 10À11 mol sÀ1 cmÀ2). Meanwhile, the NH3 formation rates
had a sharp increase from À0.70 V, and offered the formation rate
of 12.30 mg hÀ1 mgcat (1.00 Â 10À10 mol sÀ1 cmÀ2). At À0.8 V,
À1
the formation rate of NH3 (24.81 mg hÀ1 mgcat & 2.02 Â
À1
10À10 mol sÀ1 cmÀ2) was approximately 8-fold that of À0.6 V
(3.32 mg hÀ1 mgcat & 2.71 Â 10À11 mol sÀ1 cmÀ2). Further
À1
negatively shifting to À0.9 V, the NH3 formation rate continuously
À1
increased to 25.77 mg hÀ1 mgcat (2.11 Â 10À10 mol sÀ1 cmÀ2).
However, the FEs decreased dramatically from À0.40 V to À0.90 V
due to the competition of the HER reaction at more negative
potential. Although the catalytic performance of Fe2O3–IL was lower
12 A. L. Garden, Y. Abghoui and E. Skulason, Alternative Catalytic
Materials: Carbides, Nitrides, Phosphides and Amorphous Boron Alloys,
RSC Publishing, 2018, 133–163.
in the neutral solution than that in alkaline electrolyte, it outper- 13 L. Zhang, X. Ji, X. Ren, Y. Luo, X. Shi, A. M. Asiri, B. Zheng and
X. Sun, ACS Sustainable Chem. Eng., 2018, 6, 9550–9554.
formed many other catalysts under similar conditions (Table S2,
ESI†). Considering the reactivity and selectivity, À0.8 V was the
´
¨
14 A. B. Hoskuldsson, Y. Abghoui, A. B. Gunnarsdottir and E. Skulason,
ACS Sustainable Chem. Eng., 2017, 5, 10327–10333.
optimal potential for electrochemical NRR with Fe2O3–IL in a neutral 15 L. Huang, J. Wu, P. Han, A. M. Al-Enizi, T. M. Almutairi, L. Zhang
and G. Zheng, Small Methods, 2018, 2, 1800386.
electrolyte. For comparison, the formation rate of NH3 catalyzed by
16 S. Licht, B. Cui, B. Wang, F. F. Li, J. Lau and S. Liu, Science, 2015, 45,
Fe2O3–IL was 1.7 times that of Fe2O3–H2O (15.07 mg hÀ1 mgcat
À1
637–640.
from UV-Vis spectrum) at À0.8 V (Fig. S24, ESI†). Similar to alkaline 17 S. Chen, S. Perathoner, C. Ampelli, C. Mebrahtu and D. Su, Angew.
Chem., Int. Ed., 2017, 56, 2699–2703.
18 X. Xiang, Z. Wang, X. Shi, M. Fan and X. Sun, ChemCatChem, 2018,
electrolytes, the hydrazine content was negligible (Fig. S25, ESI†).
The recycling experiment and longer chronoamperometric test
10, 4530–4535.
demonstrated the highly stable capability of Fe2O3–IL in neutral 19 P. Chen, Y. Tong, C. Wu and Y. Xie, Acc. Chem. Res., 2018, 51, 2857–2866.
20 M. Rioult, D. Stanescu, E. Fonda, A. Barbier and H. Magnan, J. Phys.
Chem. C, 2016, 120, 7482–7490.
21 L. Zhuang, J. Yi, T. He, A. Du, X. Yan, G. Lei, Z. Zhu and X. Yao, Nano
electrolyte (Fig. S26 and S27, ESI†). As a result, Fe2O3–IL was also of
great interest for electrochemical NRR in a neutral electrolyte. In
addition, Fe2O3–IL could catalyze NRR in 0.001 M HCl electrolyte.
The results showed that it also had comparable catalytic perfor-
mance in an acid (Fig. S28–S31 and Table S4, ESI†).
In summary, a task-specific ionic liquid strategy was, for the
first time, proposed for the one-step synthesis of OV-rich
a-Fe2O3 nanocubes toward electrocatalytic N2 fixation to NH3.
Res., 2018, 11, 3509–3518.
22 T. Li, H. Feng, Y. Wang, C. Wang, W. Zhu, L. Yuan and G. Zhou,
J. Cryst. Growth, 2018, 498, 10–16.
23 K. S. Egorova, E. G. Gordeev and V. P. Ananikov, Chem. Rev., 2017,
117, 7132–7189.
24 C. Zhang, B. Xin, Z. Xi, B. Zhang, Z. Li, H. Zhang, Z. Li and J. Hao,
ACS Sustainable Chem. Eng., 2018, 6, 1468–1477.
25 N. Bicak, J. Mol. Liq., 2005, 116, 15–18.
n-Octylammonium formate IL, as both the reaction medium 26 J. Lian, X. Duan, J. Ma, P. Peng, T. Kim and W. Zheng, ACS Nano,
2009, 3, 3749–3761.
27 J. Lee and S. Han, Phys. Chem. Chem. Phys., 2013, 15, 18906–18914.
28 L. Hu, A. Khaniya, J. Wang, G. Chen, W. E. Kaden and X. Feng, ACS
and structure-directing template, resulted in the formation of
small a-Fe2O3 nanocubes while the reductant formic anion
in situ triggered rich OVs in a-Fe2O3 nanocubes. The as-prepared
Catal., 2018, 8, 9312–9319.
Chem. Commun.
This journal is ©The Royal Society of Chemistry 2019

 Zhang, Chenyun
Zhang, Chenyun
 Liu, Shuai
Liu, Shuai
 Chen, Tingting
Chen, Tingting
 Li, Zhonghao
Li, Zhonghao
 Hao, Jingcheng
Hao, Jingcheng