10.1002/cctc.202000501
ChemCatChem
FULL PAPER
prepared by the deposition metal nanoparticles from Pd (0.1 and 1.0
NPL) and NiMo targets on the copper TEM grids covered with carbon.
Estimated metal loading in catalysts on granulated Sibunit and Al2O3
supports are presented in Table S2, SM.
[1]
O. G. Ellert, M. V Tsodikov, S. A. Nikolaev, V. M. Novotortsev, Russ.
Chem. Rev. 2014, 83, 718–732.
V. Ponec, Appl. Catal. A Gen. 2001, 222, 31–45.
V. Dal Santo, A. Gallo, A. Naldoni, M. Guidotti, R. Psaro, Catal.
Today 2012, 197, 190–205.
[2]
[3]
[4]
[5]
F. Cai, L. Yang, S. Shan, D. Mott, B. Chen, J. Luo, C.-J. Zhong,
Catalysts 2016, 6, 96.
G. Sharma, A. Kumar, S. Sharma, M. Naushad, R. Prakash Dwivedi,
Z. A. ALOthman, G. T. Mola, J. King Saud Univ. - Sci. 2019, 31,
257–269.
X-ray photoelectron spectra (XPS) were recorded on an Axis Ultra DLD
spectrometer (Kratos analytical, UK) using monochromatic AlKα radiation.
The pass energy of the analyser was 160 eV for survey spectra and 40
eV for high resolution scans. The Kratos charge neutralizer was used for
alumina supported samples and the spectra were charge-corrected to
give Al2p peak a binding energy of 74.7 eV.
[6]
J. Zhang, M. Chaker, D. Ma, J. Colloid Interface Sci. 2017, 489,
138–149.
[7]
[8]
[9]
S. Hong, H. Lee, J. Yeo, S. H. Ko, Nano Today 2016, 11, 547–564.
J. Yu, J. Nan, H. Zeng, Appl. Surf. Sci. 2017, 402, 330–335.
T. N. Rostovshchikova, E. S. Lokteva, E. V. Golubina, K. I.
Maslakov, S. A. Gurevich, D. A. Yavsin, V. M. Kozhevin, in Adv.
Nanomater. Catal. Energy, Elsevier, 2019, pp. 61–97.
E. V. Golubina, E. S. Lokteva, K. I. Maslakov, T. N.
Rostovshchikova, M. I. Shilina, S. A. Gurevich, V. M. Kozhevin, D. A.
Yavsin, Nanotechnologies Russ. 2017, 12, 19–26.
E. S. Lokteva, A. A. Peristyy, N. E. Kavalerskaya, E. V. Golubina, L.
V. Yashina, T. N. Rostovshchikova, S. A. Gurevich, V. M. Kozhevin,
D. A. Yavsin, V. V. Lunin, Pure Appl. Chem. 2012, 84, 495–508.
T. N. Rostovshchikova, M. I. Shilina, E. V. Golubina, E. S. Lokteva, I.
N. Krotova, S. A. Nikolaev, K. I. Maslakov, D. A. Yavsin, Russ.
Chem. Bull. 2015, 64, 812–818.
T. N. Rostovshchikova, V. V. Smirnov, S. A. Gurevich, V. M.
Kozhevin, D. A. Yavsin, S. M. Nevskaya, S. A. Nikolaev, E. S.
Lokteva, Catal. Today 2005, 105, 344–349.
E. V. Golubina, T. N. Rostovshchikova, E. S. Lokteva, K. I.
Maslakov, S. A. Nikolaev, T. B. Egorova, S. A. Gurevich, V. M.
Kozhevin, D. A. Yavsin, A. Y. Yermakov, Pure Appl. Chem. 2018,
90, 1685–1701.
A. A. Bryzhin, I. G. Tarkhanova, K. I. Maslakov, S. A. Nikolaev, S. A.
Gurevich, V. M. Kozhevin, D. A. Yavsin, M. G. Gantman, T. N.
Rostovshchikova, Russ. J. Phys. Chem. A 2019, 93, 1976–1985.
W. Ahmad. Sulfur in Petroleum: Desulfurization Techniques. In T.
Saleh (Ed.) Applying Nanotechnology to the Desulfurization Process
in Petroleum Engineering. Hershey. PA; IGI Global. 2016. pp. 1–52.
W. N. W. Abdullah, W. A. W. A. Bakar, R. Ali, W. N. A. W. Mokhtar,
M. F. Omar, J. Clean. Prod. 2017, 162, 1455–1464.
Z. Ismagilov, S. Yashnik, M. Kerzhentsev, V. Parmon, A. Bourane, F.
M. Al-Shahrani, A. A. Hajji, O. R. Koseoglu, Catal. Rev. 2011, 53,
199–255.
V. S. Rudnev, I. V. Lukiyanchuk, M. S. Vasilyeva, V. P. Morozova, V.
M. Zelikman, I. G. Tarkhanova, Appl. Surf. Sci. 2017, 422, 1007–
1014.
I. G. Tarkhanova, A. A. Bryzhin, M. G. Gantman, T. P. Yarovaya, I.
V. Lukiyanchuk, P. M. Nedozorov, V. S. Rudnev, Surf. Coatings
Technol. 2019, 362, 132–140.
E. B. Gordon, A. V. Karabulin, V. I. Matyushenko, V. D. Sizov, T. N.
Rostovshchikova, S. A. Nikolaev, E. S. Lokteva, E. V. Golubina, K. I.
Maslakov, I. N. Krotova, et al., High Energy Chem. 2016, 50, 292–
297.
N. E. Kavalerskaya, E. S. Lokteva, T. N. Rostovshchikova, E. V.
Golubina, K. I. Maslakov, Kinet. Catal. 2013, 54, 597–606.
B. K. D. Moulder J.F., Stickle W.F., Sobol P.E., Handbook of X-Ray
Photoelectron Spectroscopy, Perkin-Elmer Corporation Physical
Electronics Division 6509 Flying Cloud Drive Eden Prairie,
Minnesota 55344 United Stales Of America, 1995.
T. Pillo, R. Zimmermann, P. Steiner, S. Hüfner, J. Phys. Condens.
Matter 1997, 9, 3987–3999.
A. N. Mansour, Surf. Sci. Spectra 1994, 3, 231–238.
M. C. Biesinger, B. P. Payne, L. W. M. Lau, A. Gerson, R. S. C.
Smart, Surf. Interface Anal. 2009, 41, 324–332.
L. I. Godina, A. V. Kirilin, A. V. Tokarev, I. L. Simakova, D. Y. Murzin,
Ind. Eng. Chem. Res. 2018, 57, 2050–2067.
R. S. Weatherup, B. C. Bayer, R. Blume, C. Ducati, C. Baehtz, R.
Schlögl, S. Hofmann, Nano Lett. 2011, 11, 4154–4160.
B. C. Bayer, D. A. Bosworth, F. B. Michaelis, R. Blume, G. Habler, R.
Abart, R. S. Weatherup, P. R. Kidambi, J. J. Baumberg, A. Knop-
Gericke, et al., J. Phys. Chem. C 2016, 120, 22571–22584.
A. N. Mansour, Surf. Sci. Spectra 1994, 3, 239–246.
E. V. Golubina, E. S. Lokteva, A. V. Erokhin, A. A. Veligzhanin, Y. V.
Zubavichus, V. A. Likholobov, V. V. Lunin, J. Catal. 2016, 344, 90–
99.
The samples were studied by transmission electron microscopy (TEM)
on a JEOL JEM 2100F/UHR instrument (Japan) equipped with EDS
accessory. Bright field and high-angle annular dark field (HAADF) modes
were used.
[10]
[11]
[12]
[13]
[14]
The catalyst surfaces were examined on a JEOL JSM – 6000 NeoScope
scanning electron microscope with a built-in EX-230 energy dispersive X-
ray analyser. The images were recorded in the secondary electron
imaging (SEI) mode in a high vacuum at an accelerating voltage of 5 kV.
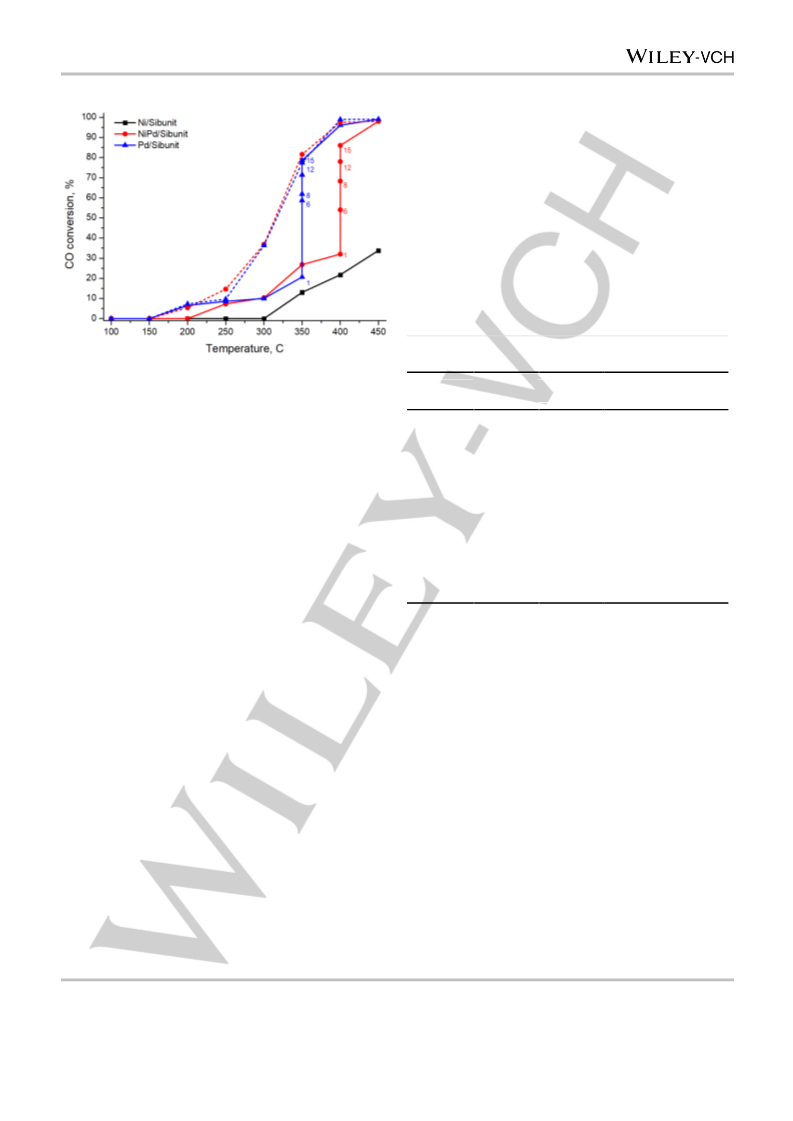
Catalysts were tested in CO oxidation using the pulse microcatalytic
method in a quartz tubular fixed-bed flow-type reactor. 100 mg of catalyst
was placed into the reactor on a Schott filter. Helium at atmospheric
pressure was continuously passed through the reactor at a flow rate of
60 ml/min. The stoichiometric reaction mixture (2 vol.% CO, 1 vol.% O2,
97 vol.% He) was fed into He flow by pulses (10 pulses per hour) using a
six-way valve with a 1 mL gas loop.
[15]
[16]
The concentrations of CO and CO2 in the reaction mixture were
measured by a GC (thermal conductivity detector; packed column
PorapakQ) using the EcoChrom software package for data processing.
The CO conversion was calculated from the GC peak areas of CO and
CO2 using pre-measured calibration curves. The steady-state CO
conversion for each reaction temperature was calculated after 4–20
pulses.
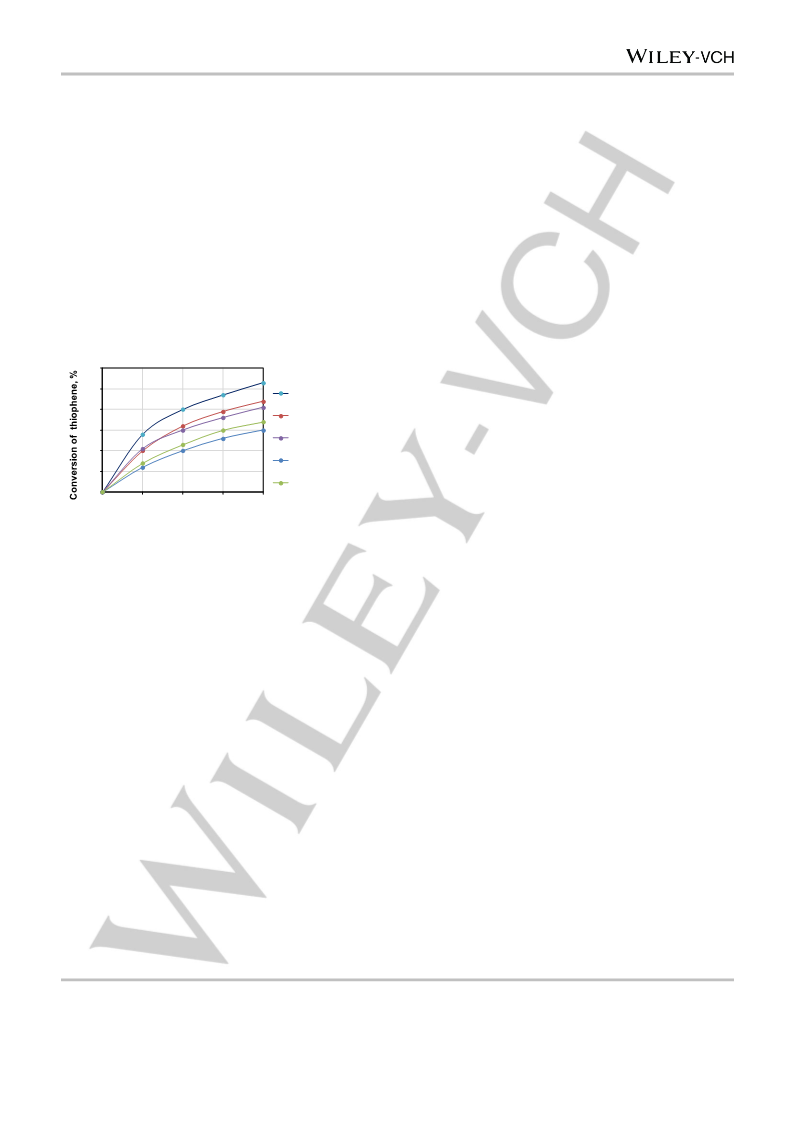
Ni, Mo, and W catalysts were tested as described in[15]. The model
mixture (10 ml of 1 wt.% thiophene solution in isooctane, 0.1 g of the
catalyst and 0.4 ml of 50% H2O2) was placed in a thermostated reactor
equipped with a stirrer. The process was carried out at 60 °C and
samples of the organic phase were periodically collected for GC-FID
analysis on a Kristall 2000М chromatograph (Chromatec, Russia) with a
capillary column (Zebron ZB-1, 30m×0.32mm i.d.×0. 5 μm). After a
standard 4-hour test, the liquid phase was drained and a new portion of
the reagents was fed in the reactor. This cycle was repeated 5 times.
[17]
[18]
[19]
[20]
[21]
[22]
[23]
[24]
Acknowledgements
[25]
[26]
This work was supported by the Russian Foundation for Basic
Research (project 19–33–90024). The authors acknowledge
support from Lomonosov Moscow State University Program of
Development for providing access to the XPS and TEM facilities.
[27]
[28]
[29]
Keywords: laser electrodispersion, nanoparticles, bimetallic
catalysts, heterogeneous catalysis, oxidation, carbon monoxide,
thiophene
[30]
[31]
[32]
D. Teschner, J. Borsodi, A. Wootsch, Z. Revay, M. Havecker, A.
Knop-Gericke, S. D. Jackson, R. Schlogl, Science (80-. ). 2008, 320,
86–89.
This article is protected by copyright. All rights reserved.

 Bryzhin
Bryzhin
 Golubina
Golubina
 Maslakov
Maslakov
 Lokteva
Lokteva
 Tarkhanova
Tarkhanova
 Gurevich
Gurevich
 Yavsin
Yavsin
 Rostovshchikova
Rostovshchikova