1
4
S. Oleszek et al. / Thermochimica Acta 527 (2012) 13–21
BFR plastics, then antimony bromide (SbBr ) can be formed as well
purchased from Wako Pure Chemical Industries Ltd. Tetrabromo-
bisphenol A (TBBPA, 97.0%) was obtained from Sigma Aldrich.
Powder mixtures of TBBPA + ZnO (3.34:1, w/w, assuming that
all bromine present in TBBPA releases and reacts with to form
3
[
12].
Trials performed on the recovery of bromine (TAMARA pilot
plant in Germany) during co-combustion of electronic scrap with
municipal solid waste (MSW) [2] indicate that the released bromine
can react with heavy metals present in MSW and form metal bro-
mides. These bromides are volatile and concentrate in the fly ash,
from where bromine can be recovered with an appropriate wet
scrubbing system [13,14]. Although the TAMARA test work focused
on bromine recovery, the reported results also indicate the advan-
tage of bromine for recovery of metals or metal oxides by their
selective bromination and evaporation. Evaluation of bromine par-
titioning and its influence on metal volatilization in those tests is
discussed in details in Ref. [15].
Recent literature [16–23] indicates that such recovery of heavy
metals has been proposed by simultaneous treatment of electric arc
furnace (EAF) dust with polyvinyl chloride (PVC) waste. In that case,
however, chlorine originating from PVC degradation is utilized for
selective chlorination and then volatilization of the formed chloride
metals.
ZnBr ) and SbBr + ZnO (3:1, w/w) were stirred manually. The sam-
2
3
ple weights used for thermal analysis were about 5–8 mg.
2.2. Methods
Thermogravimetric (TG) analysis: Thermal analysis of mixtures
(
TBBPA + ZnO and SbBr + ZnO) and pure compounds (SbBr , ZnO,
3
3
Sb O , ZnBr , and TBBPA) were carried out on a Rigaku Thermo Plus
2
3
2
TG 8120 thermobalance. A sample was placed in an open alumina
pan and heated up to the desired temperature at a constant heating
◦
rate of 10 C/min. Analysis was carried out in a flow of Helium (He)
or a mixture of He + 5 vol% O2 with a constant rate of 150 mL/min
(the applied concentration of oxygen corresponds to that occurring
in industrial rotary kiln treatment of WEEE). TG analysis of the mix-
ture was performed at least in triplicate to assure repeatability of
the obtained results.
Our initial studies on selective bromination–evaporation of
metal oxides involved thermodynamic computations [24] using the
Gibbs energy changes of selected bromination reactions. The results
For mixtures of TBBPA + ZnO and SbBr + ZnO, the inorganic
3
◦
products were characterized in solid residues collected at 320 C
and 250 C, respectively, using EPMA (Jeol, JXA-8920) and XRD
◦
indicated that brominated gases such as HBr and SbBr , formed
3
(Rigaku, Rint 220, Cu-K␣).
during BFRs thermal degradation, could act as bromination agents
for metallic oxides (e.g. ZnO, PbO, CuO, and Fe O ). Moreover,
For the mixture of TBBPA + ZnO and pure TBBPA the degrada-
2
3
tion products remaining in solid residue were collected between
these particular metal oxides have a thermodynamic tendency to
brominate more easily than Sb O . Due to their high volatilities,
◦ ◦
2
50 and 340 C (every 10 C) and extracted with acetone (0.5 mL)
2
3
using an ultrasonic bath. The organic compounds were analyzed
by combined gas chromatography (GC, Rigaku 6890N)/mass spec-
trometry (MS, JEOL, Automass Sun); the chromatogram peaks were
identified by comparison of the fragmentation patterns to those of
the NIST spectral library.
Simultaneous thermogravimetry-mass spectrometry (TG-MS)
analysis: Analysis of pure TBBPA and of a mixture of TBBPA + ZnO
were carried out with TG (TG-TDA 6200, Seiko Instrument, Extar
especially for zinc bromide (ZnBr ), the possible separation of the
2
volatile compounds by bromination in thermal treatment of solid
wastes with BFR plastics can be considered. Results of our previ-
ous experimental investigations [25] on bromination–vaporization
of Zn during thermal treatment of the mixture (TBBPA + ZnO) by
using differential scanning calorimeter and small laboratory fur-
nace (dynamic heating conditions, inert atmosphere) indicated
that the bromination reaction depends mainly on availability of
bromine around ZnO particles (it was conformed by additional
studies on isothermal treatment of mixture TBBPA + ZnO [26])
while the vaporization of the formed ZnBr2 can be restrained by
char which is formed as result of flame retardant degradation. In
fact, there is still lack of information concerning the carbothermic
reduction of ZnO into Zn in presence of the char and vaporization of
the formed Zn. There is still insufficient information on the decom-
position pathway of TBBPA in presence of ZnO, which is a clue to
understand the bromination reaction mechanism.
6
000) combined with a mass selective detector (MSD 5973, Hewlett
Packard) connected by a 5 m steel capillary transfer line (Frontier
Labo, UADTP-5 M, I.D. 0.25 mm).
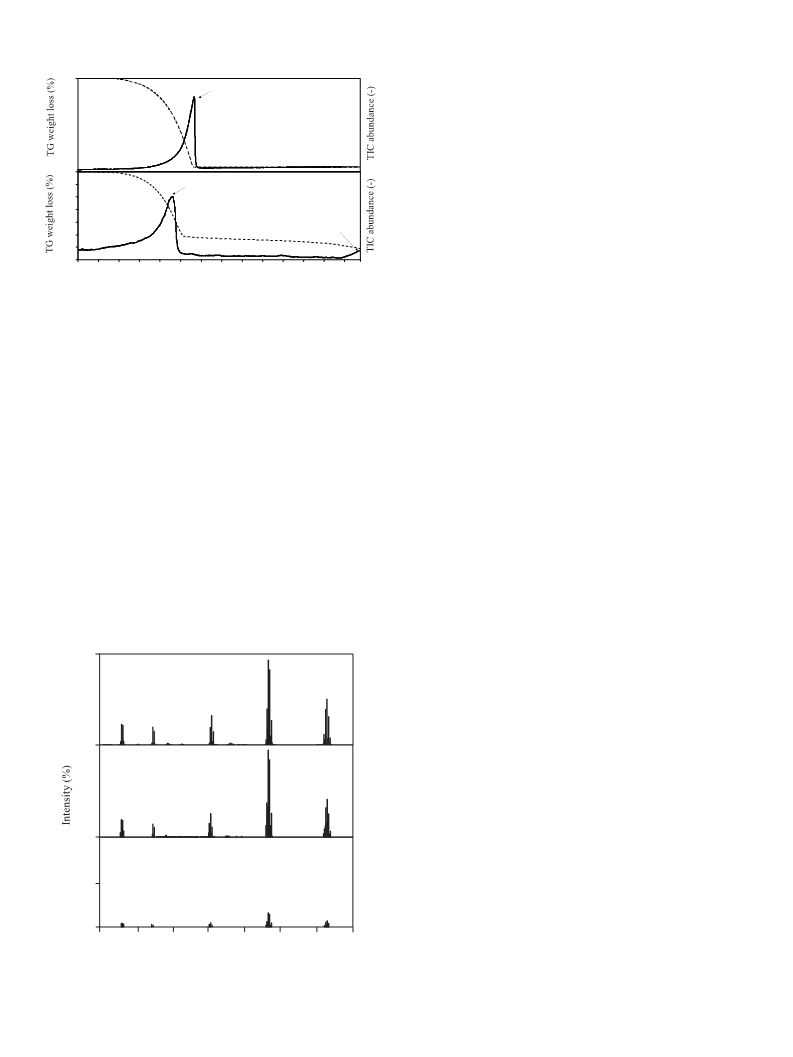
Analysis of pure SbBr3 and of a mixture of SbBr + ZnO were
3
carried out with TG (Rigaku Thermo Plus TG 8120) combined with
a mass spectrometer (JEOL, Automass Sun) connected by capillary
transfer line equipped with an Ultra Alloy-DTM column (0.53 mm
I.D., 0.75 O.D.).
The TGA sample was placed into the alumina pan and heated
◦
(
10 C/min) under a He flow of 150 mL/min. The evolved gases
In this study, thermogravimetric combined with differential
thermal analysis (TG-DTA) were conducted to investigate the
reactivity of ZnO with both HBr, originating from the thermal
were transferred through the capillary transfer line into the mass
◦
detector. The capillary was heated up to 350 C in order to avoid
condensation of degradation products. Note: The MS measure-
decomposition of TBBPA, and solid SbBr , which is supposed to form
3
◦
ments could not be conducted over 400 C due to clogging the
during thermal degradation of BFR plastics containing Sb O syner-
2
3
capillary caused by condensation of the vaporized ZnBr2.
gist. The investigations have been performed in inert and oxidative
atmosphere, and both, the bromination and vaporization pro-
cesses are discussed precisely. Additionally, gaseous products that
evolved during thermal treatment of the mixtures (TBBPA + ZnO
3. Results and discussion
and SbBr + ZnO) were qualified by direct combination of thermo-
3.1. Thermal treatment of TBBPA + ZnO
3
gravimetry with mass spectrometry (TG-MS). The role of ZnO on
degradation pathway of TBBPA was also investigated.
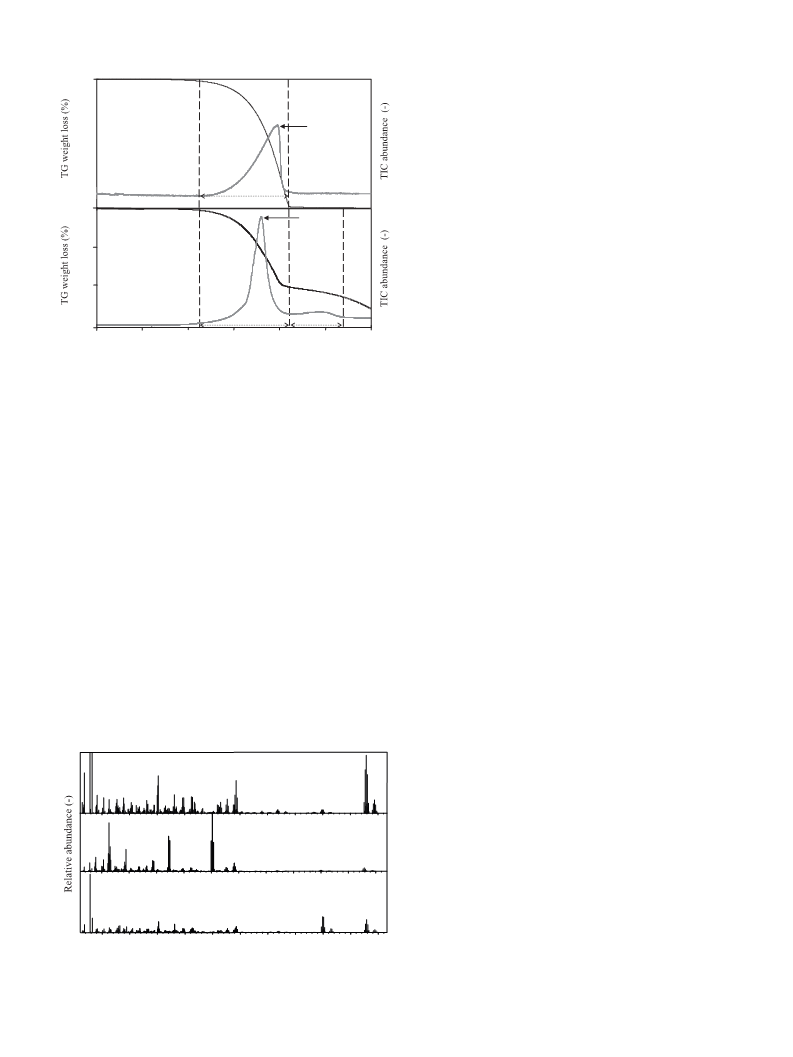
Thermograms obtained in inert (He) and oxidizing (He + 5 vol%
O ) atmospheres for the three pure compounds (TBBPA, ZnBr , and
2
2
ZnO) and for the mixture of TBBPA + ZnO (3.34:1, w/w) are shown
in Fig. 1A and B.
2
. Experimental
In the inert atmosphere, the decomposition and vaporization
of pure TBBPA and also the vaporization of ZnBr2 proceeds in
one step with a 100% loss of mass, while ZnO remains unchanged
2.1. Materials
◦
over the entire temperature range (up to 1300 C) (Fig. 1A). Dur-
Antimony bromide (SbBr ), zinc bromide (ZnBr ), antimony
ing thermal treatment of the mixture (TBBPA + ZnO) mass losses
occur in three distinct stages, at consecutive temperature ranges:
3
2
oxide (Sb O ), and zinc oxide (ZnO), with purities of 99.9% were
2
3

 Oleszek, Sylwia
Oleszek, Sylwia
 Grabda, Mariusz
Grabda, Mariusz
 Shibata, Etsuro
Shibata, Etsuro
 Nakamura, Takashi
Nakamura, Takashi