efficiently in the presence of excess Tiron as this may be needed for
it to be successful in competing for coordination sites around the
manganese atom. The experiment with added H2O2 indicates that
the importance of the Tiron anion is not only due to its efficiency of
in situ H2O2 generation but crucially its ability to utilise the in situ
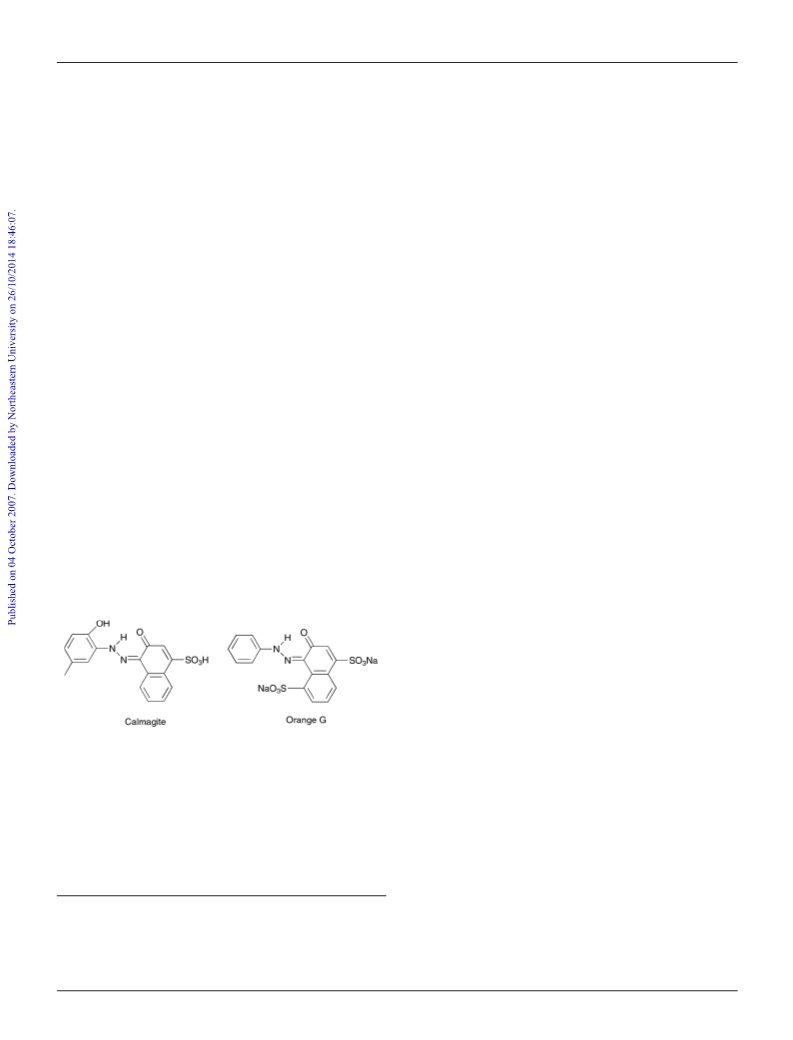
H2O2 generated for the oxidation of Calmagite. The efficacy of de-
protonated Tiron in this may be related to its redox, non-innocent,
properties that enable it to exist as a catechol, semi-quinone or
quinone species while still bound to the manganese centre. The
H2O2 formed presumably oxidises the Tiron-catechol to its semi-
quinone or quinone forms and/or binds to a vacant site on the
Mn atom to form a Mn(III), Mn (IV)–oxo (or possibly Mn(V)–
oxo) species which is then responsible for the oxidation of bound
Calmagite (Scheme 1).
used in relation to this work (with Calmagite) and the work of
Lenoir et al. using FeIII–TAML (with Orange II) has important
implications in terms of the treatment of wastewaters.
Acknowledgements
TSS is supported in this work by the Royal Society of Chemistry
(RSC, UK) Research Fund 2007.
Notes and references
† N-2-Hydroxyethylpiperazine-Nꢀ-3-propanesulfonic acid (EPPS) buffer
(200 mM, 10.0 mL), hydroxylamine hydrochloride (NH3OH+Cl− 1.00 M,
10.0 mL), Tiron (1,2-dihydroxybenzene-3,5-disulfonate, disodium salt
(15.0 mM, 10.0 mL) and sodium hydroxide pellets (semi-conductor grade,
0.40 g, 1.0 mM) were added to de-ionised water (∼20 mL). When all
of the NaOH had dissolved, the pH was adjusted to 8.0 using freshly
prepared (CO2-free) NaOH(aq). To this solution were added Calmagite
(1.00 mM, 10.0 mL) and MnCl2·4H2O (0.0500 mM, 1.00 mL) to produce
a purple coloured solution which was transferred to a volumetric flask
(100.0 mL) and made up with de-ionised water. The pH was checked
again and if necessary adjusted to 8.0 and the solution transferred
to a dreschel bottle equipped with a glass inlet. The dreschel bottle
was placed in a water bath at 20
1
◦C and dioxygen gas at a rate
of 0.6 L min−1 was passed through the rapidly stirred solution. The
absorbance of the solution at 544 nm (e = 10 500 L mol−1 cm−1) was
monitored at regular intervals using a 0.10 cm glass cuvette. As the
Calmagite dye was oxidized it changed colour from a deep purple to a
pale yellow. Where appropriate, aqueous Mn(II) solution was replaced
with [Na]5[MnIII(3,5-(SO3)2Cat)2]·10H2O (0.0500 mM, 1.00 mL), where
H2Cat = 1,2-dihydroxybenzene. [Na]5[MnIII(3,5-(SO3)2Cat)2]·10H2O was
prepared as described previously.12 The oxidation of Orange G (1.00 mM,
10.0 cm3) when used in place of Calmagite was monitored at 478 nm
(e = 11 600 L mol−1 cm−1). Analysis for hydrogen peroxide was achieved
by removing aliquots (0.50 mL) of reaction solution which were added
to titanium(IV) solution (3.00 mL) and the absorbance of the samples
measured at kmax of 407 nm (t = 0 min as blank) using a 0.10 cm glass
cuvette.13
Scheme 1 General mechanism for the catalytic in situ generation of H2O2
and oxidation of Calmagite. CAL = de-protonated Calmagite anion; Cat =
de-protonated Tiron catechol anion and Q = Tiron quinone anion.
Similar mechanisms have been proposed by both Oakes et al.1
and Lenoir et al.6 in the oxidation of azo dyes by peroxides
catalysed by Mn(II) and Fe(III)–TAML complexes respectively.
Conclusions
This study provides, to the best of our knowledge, the first example
of a system that can, not only catalytically generate hydrogen
peroxide in situ from dioxygen in aqueous solution under mild
conditions, but also utilize this hydrogen peroxide to perform
a model oxidation reaction using the same catalytic system
under the same ambient conditions. The mixed manganese(II)
de-protonated Calmagite/Tiron complex is able to efficiently
utilise the in situ H2O2 generated for the rapid decomposition
of Calmagite and this only occurs slowly in the absence of Tiron.
The importance of de-protonated Tiron as a co-ligand for efficient
Calmagite destruction in this work is an interesting, if perhaps
not un-surprising result, given the non-innocent redox properties
of catechols.11 Future studies, including the use of EPR, will
help provide further information on the nature of the oxidising
species in this system. The generation of hydrogen peroxide with
Calmagite provides the first example of a non-catechol molecule
that can generate hydrogen peroxide from the catalytic reduction
of dioxygen using hydroxylamine and this provides an important
additional insight into the mechanism of hydrogen peroxide
generation. The current system provides an interesting example
of the ability of manganese–catecholate complexes to effectively
activate small molecules to perform useful chemical work which
has importance in the modelling of natural systems. The tailoring
of the decomposition of specific dyes with the catalytic system
1 J. Oakes, P. Gratton and I. Weil, J. Chem. Soc., Dalton Trans., 1997,
3805.
2 T. S. Sheriff, J. Chem. Soc., Dalton Trans., 1992, 1051.
3 G. Strukel, Catalytic Oxidations With Hydrogen Peroxide As Oxidant,
Kluwer Academic Publishers, Dordrecht, 1993; R. Hage, J. E. Iburg,
J. Kerschner, J. H. Koek, E. L. M. Lempers, R. J. Martens, U. S.
Racherla, S. W. Russell, T. Swarthoff, M. R. P. Vanvliet, J. B. Warnaar,
L. Vanderwolf and B. Krijnen, Nature, 1994, 369, 637; R. Noyori, M.
Aoki and K. Sato, Chem. Commun., 2003, 1977; D. Chandra and A.
Bhaumik, Ind. Eng. Chem. Res., 2006, 45, 4879.
4 Pollution, Prevention and Abatement Handbook, World Bank Group,
Washington, DC, 1998.
5 H. Zollinger, Color Chemistry: Synthesis, Properties and Applications
of Organic Dyes and Pigments, Wiley, Weinheim, 3rd edn, 2003.
6 D. M. Gould, W. P. Griffiths and M. Spiro, J. Mol. Catal. A: Chem.,
2001, 175, 289; P. Verma, P. Baldrian and F. Nerud, Chemosphere, 2003,
50, 975; N. Chahbane, D.-L. Popescu, D. A. Mitchell, A. Chanda, D.
Lenoir, A. D. Ryabov, K.-W. Scramm and T. J. Collins, Green Chem.,
2007, 9, 49.
7 R. Hage and A. Lienke, Angew. Chem., Int. Ed., 2006, 45, 206.
8 J. M. Campos-Martin, G. Blanco-Brieva and J. L. Fierro, Angew.
Chem., Int. Ed., 2006, 45, 6962.
9 N. J. Milne, J. Surfactants Deterg., 1998, 1, 253.
10 A. Gelasco, S. Bensiek and V. L. Pecoraro, Inorg. Chem., 1988, 37, 3301.
11 C. G. Pierpont and C. W. Lange, Prog. Inorg. Chem., 1994, 41, 331.
12 T. S. Sheriff, P. Carr and B. Piggott, Inorg. Chim. Acta, 2003, 348, 115.
13 A. C. Egerton, A. J. Everett, G. J. Minkoff, S. Rudrakanchana and K. C.
Salooja, Anal. Chim. Acta, 1954, 10, 422; P. A. Clapp, D. F. Evans and
T. S. Sheriff, Anal. Chim. Acta, 1989, 218, 331.
5122 | Dalton Trans., 2007, 5119–5122
This journal is
The Royal Society of Chemistry 2007
©

 Sheriff, Tippu S.
Sheriff, Tippu S.
 Cope, Steven
Cope, Steven
 Ekwegh, Mmaezi
Ekwegh, Mmaezi