J.K. Edwards et al. / Journal of Catalysis 236 (2005) 69–79
79
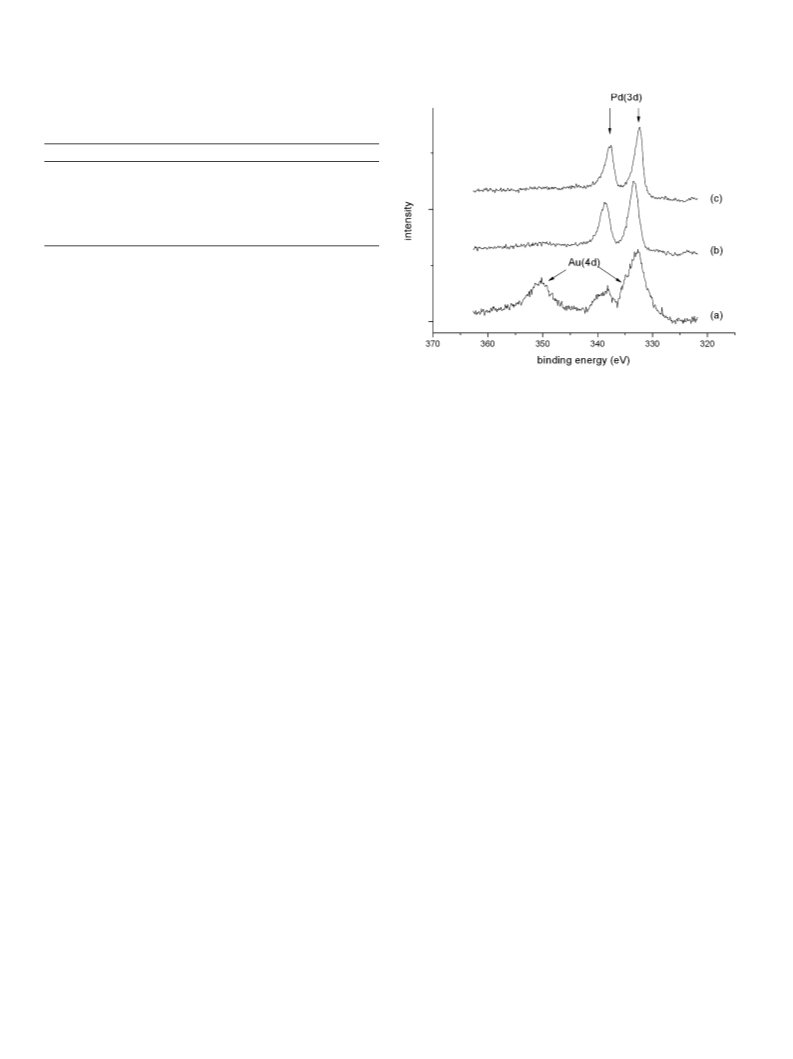
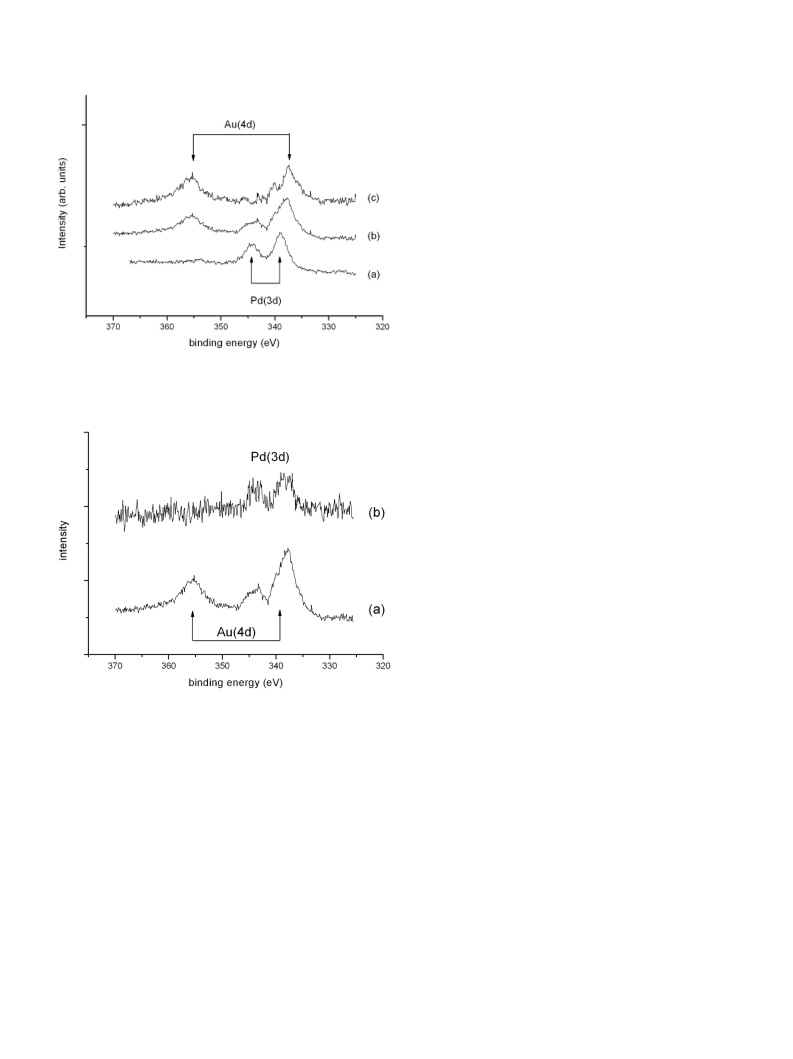
of the observed activity is related to these structures. However,
it is likely that the high activity and high selectivity sites for
H2 oxidation are significantly different from those in the pure
Au/TiO2 catalysts.
[14] H. Sun, J.J. Leonard, H. Shalit, US Patent 4393038 (1981).
[15] L.W. Gosser, J.-A.T. Schwartz, US Patent 4772458 (1988).
[16] L.W. Gosser, US Patent 4889705 (1989).
[17] C. Pralins, J.-P. Schirmann, US Patent 4996039 (1991).
[18] T. Kanada, K. Nagai, T. Nawata, US Patent 5104635 (1992).
[19] J. van Weynbergh, J.-P. Schoebrechts, J.-C. Colery, US Patent 5447706
(1995).
5. Conclusions
[20] S.-E. Park, J.W. Yoo, W.J. Lee, J.-S. Chang, U.K. Park, C.W. Lee, US
Patent 5972305 (1999).
[21] G. Paparatto, R. d’Aloisio, G. De Alberti, P. Furlan, V. Arca, R. Buzzoni,
L. Meda, EP Patent 0978316A1 (1999).
[22] B. Zhou, L.-K. Lee, US Patent 6168775 (2001).
[23] M. Nystrom, J. Wanngangrd, W. Herrmann, US Patent 6210651 (2001).
[24] J.W. Hastie, Chem. Phys. Lett. 17 (1972) 195.
[25] S.E. Park, L. Huang, C.W. Lee, J.-S. Chang, Catal. Today 61 (2000) 117.
[26] E.D. Park, Y.-S. Huang, J.S. Lee, Catal. Commun. 2 (2001) 187.
[27] R. Meiers, U. Dingerdissen, W.F. Hölderich, J. Catal. 176 (1998) 376.
[28] P. Landon, P.J. Collier, A.J. Papworth, C.J. Kiely, G.J. Hutchings, Chem.
Commun. (2002) 2058.
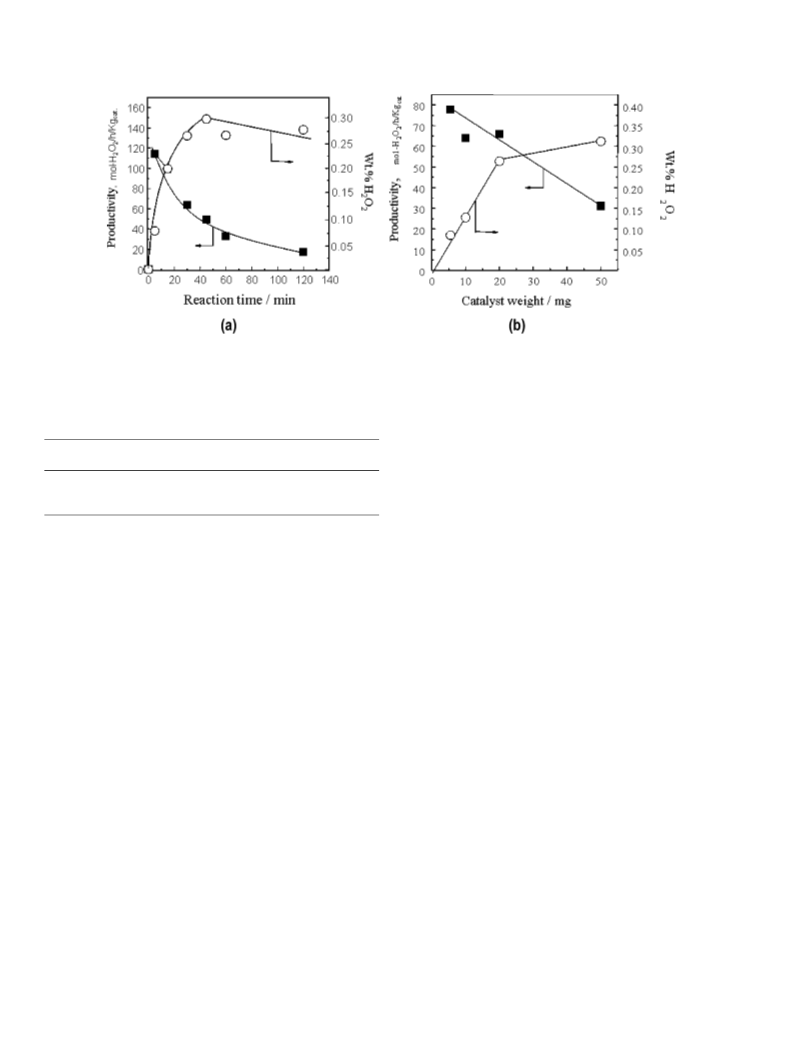
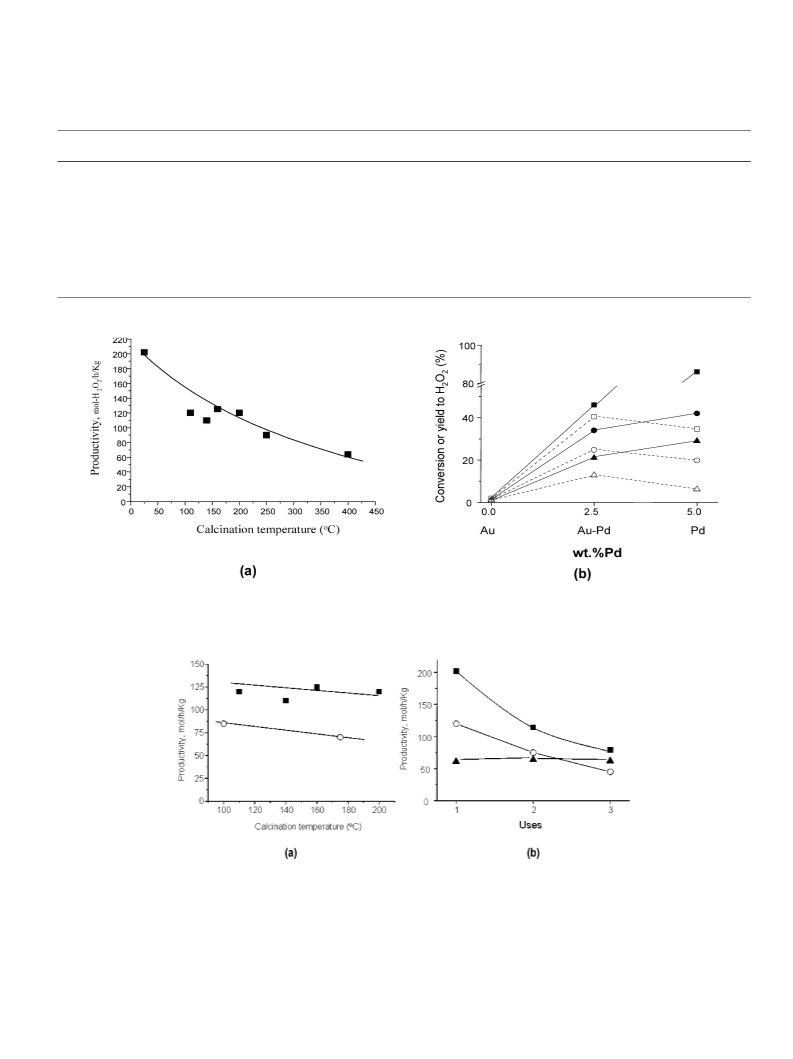
We have shown that calcined Au–Pd/TiO2 materials are
highly effective catalysts for the direct synthesis of H2O2 from
the oxidation of H2, and very high rates and selectivities can
be achieved (114 molH O2 h−1 kgc−a1t and H2O2 selectivity 93%)
2
under appropriate conditions. These catalysts are stable and can
be reused as long as they are calcined at 400 ◦C before initial
use. Pretreatment at lower temperatures leads to an unstable,
but even more active, catalyst. In conclusion, these results pro-
vide the basis for the commercial design of a direct synthesis
process that can be performed outside of the explosive region
and hence can provide safe and efficient synthesis of H2O2.
[29] P. Landon, P.J. Collier, A.J. Papworth, C.J. Kiely, G.J. Hutchings, A. Bur-
rows, Phys. Chem. Chem. Phys. 5 (2003) 1917.
[30] V.R. Choudhary, A.G. Gaikwad, S.D. Sansare, Catal. Lett. 83 (2002) 235.
[31] V.R. Choudhary, A.G. Gaikwad, S.D. Sansare, Catal. Lett. 84 (2002) 81.
[32] R.A. Sheldon, I. Arends, G.-J.T. Brink, A. Dijksman, Acc. Chem. Res. 35
(2002) 774.
[33] C.D. Wagner, L.E. Davis, M.V. Zeller, J.A. Taylor, R.M. Raymond, L.H.
Gale, Surf. Interface Anal. 3 (1981) 211.
[34] A. Hilaire, P. Legare, Y. Holl, G. Maire, Surf. Sci. 103 (1981) 125.
[35] A.F. Lee, C.J. Baddeley, C. Hardacre, M.R. Ormerod, R.M. Lambert,
G. Schmid, H.J. West, Phys. Chem. 99 (1995) 6096.
[36] H. Takatani, H. Kago, Y. Kobayashi, F. Hori, R. Oshima, Trans. Mater.
Res. Soc. Japan 28 (2003) 871.
Acknowledgments
This work formed part of the EU AURICAT project (Con-
tract HPRN-CT-2002-00174) and the EPSRC/Johnson Matthey-
funded ATHENA project. The authors thank the World Gold
Council (through the GROW scheme), and Cardiff Univer-
sity (AA Reed studentship) for providing support for J.K.E.,
C.J.K., and A.H. also acknowledge the generous support of the
NSF Materials Research Science and Engineering Center (NSF
DMR-0079996).
[37] Y. Kobayashi, S. Kiao, M. Seto, H. Takatani, M. Nakanishi, T. Oshima,
Hyperfine Interact. 156/157 (2004) 75.
[38] H. Takatani, F. Hori, M. Nakanishi, R. Oshima, Australia J. Chem. 53
(2003) 1025.
[39] C. Kan, W. Cai, C. Li, L. Zhang, H.J. Hofmeister, J. Phys. D 36 (2003)
1609.
[40] T. Nakagawa, H. Nitani, S. Tanabe, K. Okitsu, S. Seino, Y. Mizukoshi,
T.A. Yamamoto, Ultrason. Sonochem. 12 (2004) 249.
Supporting information
Supporting information associated with this article can be
[41] P. Buffat, Thin Solid Films 32 (1970) 283.
[42] D. Dissanayake, J. Lunsford, J. Catal. 206 (2002) 173.
[43] D. Dissanayake, J. Lunsford, J. Catal. 214 (2003) 113.
[44] M. Valden, X. Lai, D.W. Goodman, Science 281 (1998) 1647.
[45] D.C. Maeir, D.W. Goodman, J. Am. Chem. Soc. 126 (2004) 1892.
[46] V.A. Bondzie, S.C. Parker, C.T. Campbell, Catal. Lett. 63 (1999) 143.
[47] N. Lopez, J.K. Nørskov, J. Am. Chem. Soc. 124 (2002) 11262.
[48] G.C. Bond, D.T. Thompson, Gold Bull. 33 (2000) 41.
[49] Q. Fu, H. Saltsburg, M. Flytzani-Stephanopoulos, Science 301 (2003) 935.
[50] J. Guzman, B.C. Gates, J. Phys. Chem. B 106 (2002) 7659.
[51] M.S. Chen, D.W. Goodman, Science 306 (2004) 252.
[52] M. Haruta, Gold Bull. 37 (2004) 27.
[53] S. Carrettin, P. McMorn, P. Johnston, K. Griffin, G.J. Hutchings, Chem.
Commun. (2002) 696.
[54] S. Carrettin, P. McMorn, P. Johnston, K. Griffin, G.J. Hutchings, C.J.
Kiely, J. Phys. Chem. Chem. Phys. 5 (2003) 1329.
[55] P. Landon, J. Ferguson, B.E. Solsona, T. Garcia, A.F. Carley, A.A. Herz-
ing, C.J. Kiely, S.E. Golunski, G.J. Hutchings, Chem. Commun. (2005)
3385–3387.
References
[1] R.A. Sheldon, Stud. Surf. Sci. Catal. 66 (1991) 573.
[2] M. Besson, P. Gallezot, Catal. Today 57 (2000) 127.
[3] D. Enders, L. Wortmann, R. Peters, Acc. Chem. Res. 33 (2000) 157.
[4] A.R.J. Vaino, Org. Chem. 65 (2000) 4210.
[5] S. Lee, P.L. Fuchs, J. Am. Chem. Soc. 124 (2002) 13978.
[6] P. Wadhwani, M. Mukherjee, D. Bandyopadhyay, J. Am. Chem. Soc. 123
(2001) 12430.
[7] P.B. Walsh, Tappi J. 74 (1991) 81.
[8] M. Taramasso, G. Peego, B. Notari, US Patent 4410501 (1983).
[9] H.T. Hess, in: I. Kroschwitz, M. Howe-Grant (Eds.), Kirk–Othmer En-
cyclopaedia of Chemical Engineering, vol. 13, Wiley, New York, 1995,
p. 961.
[10] H. Henkel, W. Weber, US Patent 1108752 (1914).
[11] G.A. Cook, US Patent 2368640 (1945).
[12] Y. Izumi, H. Miyazaki, S. Kawahara, US Patent 4009252 (1977).
[13] Y. Izumi, H. Miyazaki, S. Kawahara, US Patent 4279883 (1981).

 Edwards, Jennifer K.
Edwards, Jennifer K.
 Solsona, Benjamin E.
Solsona, Benjamin E.
 Landon, Philip
Landon, Philip
 Carley, Albert F.
Carley, Albert F.
 Herzing, Andrew
Herzing, Andrew
 Kiely, Christopher J.
Kiely, Christopher J.
 Hutchings, Graham J.
Hutchings, Graham J.