Evidence of an equilibrium between selenides and osmium(VIII) reagents
and selenoxides and osmium(VI) reagents
a
b
b
b
b
Alain Krief,* Alexandra Destree, Virginie Durisotti, Nicolas Moreau, Caroline Smal and
a
Catherine Colaux-Castillo
Laboratoire de Chimie Organique de Synthèse, Département de Chimie, Facultés Universitaires
a
Notre-Dame de la Paix, 61 rue de Bruxelles, B-5000 Namur, Belgium. E-mail: alain.krief@fundp.ac.be;
Fax: +32 8172 4536; Tel: +32 8172 4539
Laboratoire de Travaux Pratiques de Chimie Organique 1 Licence, Département de Chimie, Facultés
b
iere
Universitaires Notre-Dame de la Paix, 61 rue de Bruxelles, B-5000 Namur, Belgium
Received (in Cambridge, UK) 16th October 2001, Accepted 29th January 2002
First published as an Advance Article on the web 15th February 2002
Driving the equilibrium between selenides and osmium(VIII
)
reagents with selenoxides and osmium(VI) by a subsequent
reaction (rearrangement of allyl selenoxides to allyl alcohols
or addition of osmium(VIII) species on CNC double bonds) to
one side, allows the transformation of methyl geranyl
selenides to linalool and of methyl citronellyl selenoxide to
Scheme 3
This equilibrium, if it exists, could be displaced in one or the
other direction by coupling a subsequent reaction such as the
transformation of (i) allyl seleninates to allyl alcohols (Scheme
1) or (ii) olefins to diols (Scheme 2).
We have now found that potassium osmate dihydrate is able
to catalyze the oxygen transfer from some selenoxides to other
selenides favoring thus the equilibrium between two selenides
and the corresponding selenoxides.
6,7-dihydroxy citronellyl selenide.
1
We recently described that AD-mix-b, a commercially availa-
ble mixture containing potassium osmate dihydrate (K
2
O-
sO (OH) , the pre-oxidant), (DHQD) PHAL (the chiral ligand),
2
4
2
potassium carbonate (the base) and potassium ferricyanide (the
co-oxidant) invented by Sharpless,2 reacts with 2-hexenyl
Methyl geranyl selenide 8 and benzyl phenyl selenoxide 6a
were chosen as the two partners with the expectation that the
equilibrium would take place and be drawn to completion by the
cascade allyl selenoxide-[2,3] sigmatropic rearrangement and
seleninate-selenophilic substitution to finally produce linalool
phenyl- and methyl-selenides 1 to produce, the corresponding
allyl alcohols 4 in good yields (Scheme 1). These result from (i)
the selective oxidation, by osmium(VIII) species, of the selenium
atom of allyl selenides leading to the corresponding selenoxides
2
and (ii) rearrangement of the later to allyl alcohols 4 by the
1
0 and benzyl phenyl selenide 11a.
well known cascade [2,3] sigmatropic rearrangement (2 ? 3)
Typically, the reaction was carried out on 8 under conditions
followed by seleninate-selenophilic substitution (Scheme 1).
which mimic the AD-mix reaction shown in Schemes 1 and 2:
[
2 2 4 2
catalytic amounts of K OsO (OH) , (DHQD) PHAL and
potassium carbonate in aqueous t-BuOH] but in the absence of
potassium ferricyanide.1 After 7 h at 20 °C, rac-linalool (d,l)
,2
1
0 was obtained in 70 % yield (Scheme 4).
We have also confirmed that methyl geranyl selenide 8 (i) is
recovered unchanged if the same reaction is performed under
the conditions reported just above, but in the absence of
potassium osmate dihydrate, and (ii) is much more slowly
oxidized when dibutyl selenoxide 6b is used in place of benzyl
phenyl selenoxide 6a (Scheme 4). This suggests that each type
of selenoxide has a different oxidative power and that dialkyl
selenoxides are the least efficient of the series. We have, in fact,
already described that dialkyl selenoxides are less efficient
oxidants than alkyl benzyl, alkyl phenyl, benzyl phenyl or
diphenyl-selenoxides in the AD reaction (SeOAD) using
selenoxides as co-oxidants.3
Scheme 1
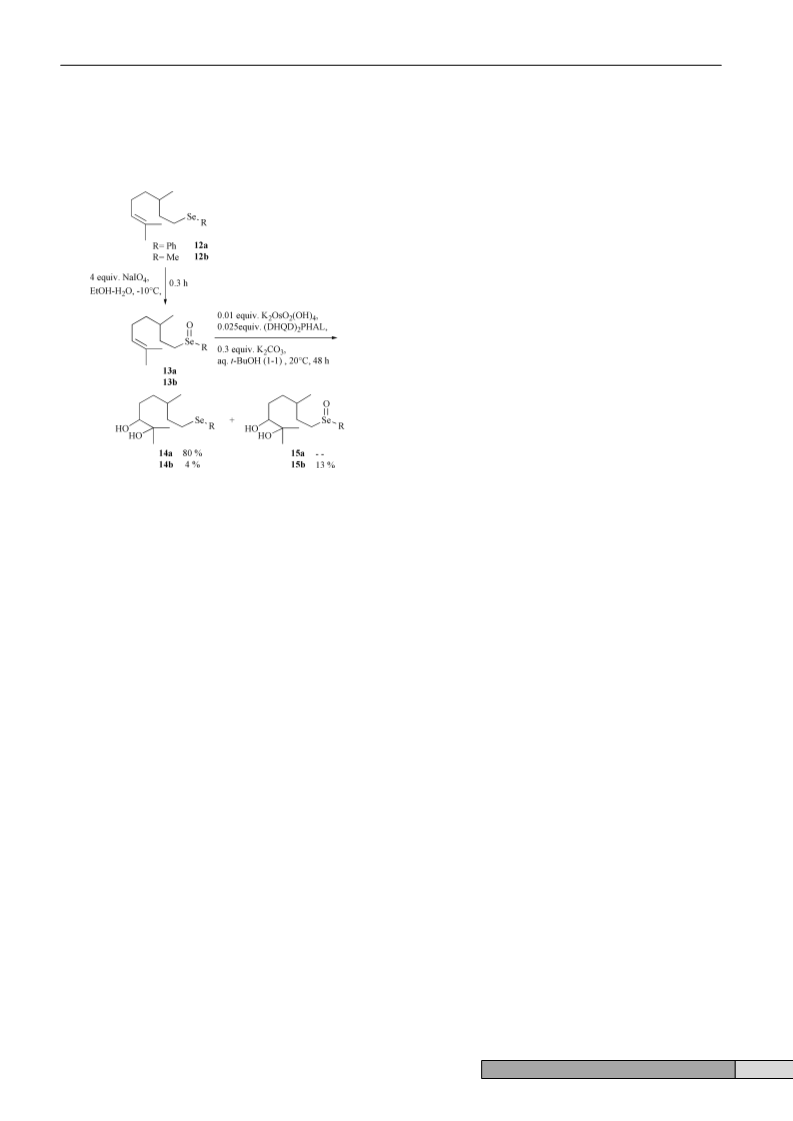
We also found that potassium osmate dihydrate is able to
catalyze, in the presence of water, the transformation of
unsaturated selenoxides to the corresponding dihydroxyalkyl
We also recently described that selenoxides could ad-
vantageously replace potassium ferricyanide, in AD-mix mon-
itored dihydroxylation of olefins (Scheme 2).1
Scheme 2
These results, which seem at first contradictory, can be
explained by an equilibrium involving four different species
(
Scheme 3).
Scheme 4
5
58
CHEM. COMMUN., 2002, 558–559
This journal is © The Royal Society of Chemistry 2002

 Krief, Alain
Krief, Alain
 Destree, Alexandra
Destree, Alexandra
 Durisotti, Virginie
Durisotti, Virginie
 Moreau, Nicolas
Moreau, Nicolas
 Smal, Caroline
Smal, Caroline
 Colaux-Castillo, Catherine
Colaux-Castillo, Catherine