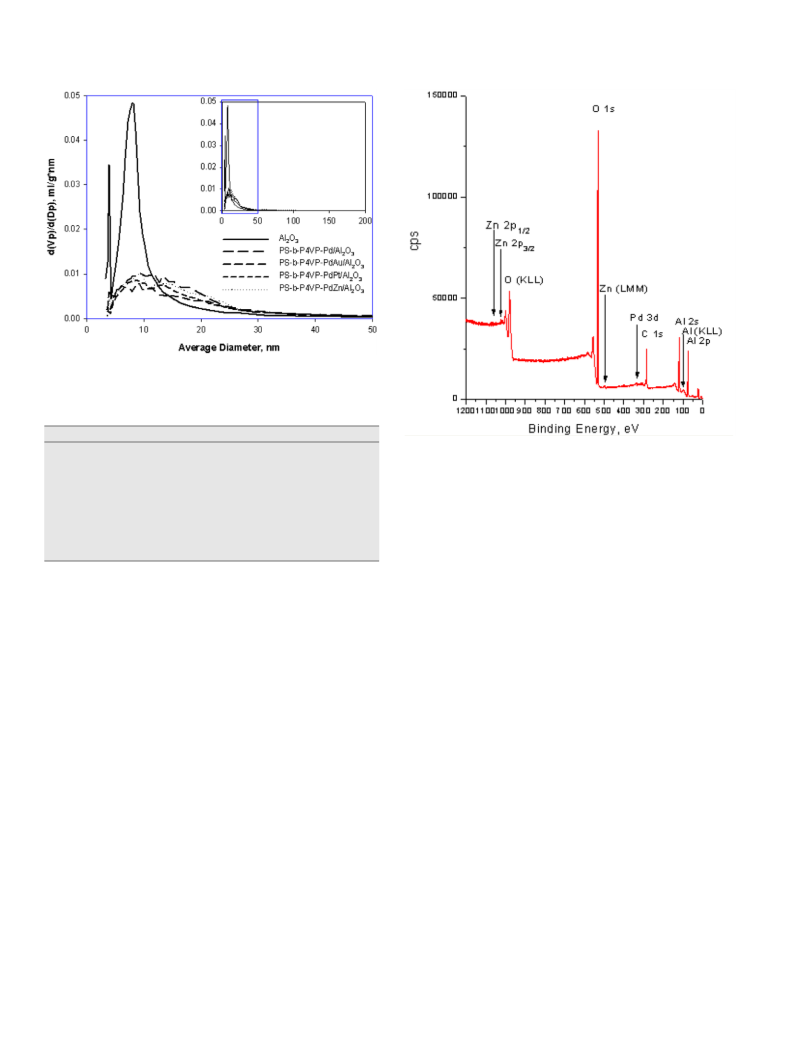
E.M. Sulman et al. / Journal of Catalysis 262 (2009) 150–158
157
Table 6
[3] H. Song, R.M. Rioux, J.D. Hoefelmeyer, R. Komor, K. Niesz, M. Grass, P. Yang,
G.A. Somorjai, J. Am. Chem. Soc. 128 (2006) 3027.
Catalytic properties of the heterogenized catalysts after repeated use.
[4] L.M. Bronstein, D.M. Chernyshov, R. Karlinsey, J.W. Zwanziger, V.G. Matveeva,
E.M. Sulman, G.N. Demidenko, H.-P. Hentze, M. Antonietti, Chem. Mater. 15
(2003) 2623.
[5] P.L.L. Gunter, J.W. Niemantsverdriet, F.H. Ribeiro, G.A. Somorjai, Catal. Rev.-Sci.
Eng. 39 (1997) 77.
[6] G.A. Somorjai, Y.G. Borodko, Catal. Lett. 76 (2001) 1.
[7] A.T. Bell, Science 299 (2003) 1688.
[8] D. Astruc, F. Lu, J.R. Aranzaes, Angew. Chem. Int. Ed. 44 (2005) 7852.
[9] C. Mueller, M.G. Nijkamp, D. Vogt, Eur. J. Inorg. Chem. 20 (2005) 4011.
[10] L.M. Bronstein, in: H.S. Nalwa (Ed.), Encyclopedia of Nanoscience and Nan-
otechnology, APS, Stevenson Ranch, CA, 2005, p. 193.
Catalyst
Pd content, wt%
Selectivity, %
Activity,
−1
mol LN mol Pd−1 s
Run 2a
Run 5
Run 2
Run 5
Run 2
Run 5
PS-b-P4VP–Pd/Al2O3
PS-b-P4VP–PdAu/Al2O3
PS-b-P4VP–PdPt/Al2O3
PS-b-P4VP–PdZn/Al2O3
0.02
0.02
0.03
0.04
0.02
0.02
0.03
0.04
98.5
99.0
98.0
98.5
99.0
98.0
98.0
99.0
57.9
27.0
23.1
58.7
27.2
23.5
128.3
131.4
a
Run 1 is shown in Table 5.
[11] N.V. Semagina, A.V. Bykov, E.M. Sulman, V.G. Matveeva, S.N. Sidorov, L.V.
Dubrovina, P.M. Valetsky, O.I. Kiselyova, A.R. Khokhlov, B. Stein, L.M. Bronstein,
J. Mol. Catal. A 208 (2004) 273.
[12] M.V. Seregina, L.M. Bronstein, O.A. Platonova, D.M. Chernyshov, P.M. Valetsky,
J. Hartmann, E. Wenz, M. Antonietti, Chem. Mater. 9 (1997) 923.
[13] S. Klingelhoefer, W. Heitz, A. Greiner, S. Oestreich, S. Förster, M. Antonietti, J.
Am. Chem. Soc. 119 (1997) 10116.
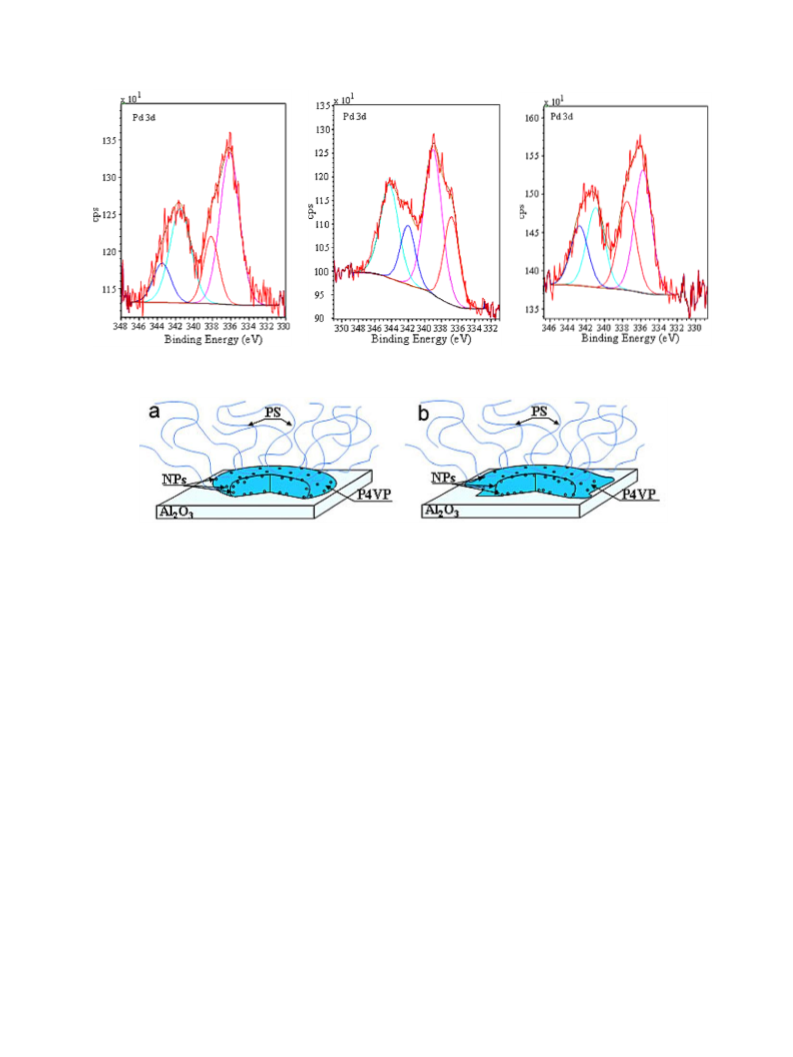
believe that the stronger the acceptor properties of a modifier (to-
ward Pd) leading to a stronger adsorption of NPs on alumina, the
longer the induction period. Note that for the commercial Pd/Al2O3
where Pd is directly adsorbed on alumina, the induction period is
480 s [63].
Thus, for a donor modifying metal (or no modifying metal),
deposition of block copolymer micelles on alumina results in the
increase of the catalyst polarity (hydrophilization) and in better ac-
cessibility of catalytically active sites for an amphiphilic substrate
(DHL). We believe that for acceptor modifying metals (Au and Pt),
this phenomenon is offset by stronger adsorption of nanoparticles
on alumina (Scheme 2b), leading to a decrease of the amount of
available catalytic sites, hence leading to a decrease of catalytic ac-
tivity.
[14] T.F. Jaramillo, S.-H. Baeck, B.R. Cuenya, E.W. McFarland, J. Am. Chem. Soc. 125
(2003) 7148.
[15] B. Roldan Cuenya, S.-H. Baeck, T.F. Jaramillo, E.W. McFarland, J. Am. Chem.
Soc. 125 (2003) 12928.
[16] R.S. Underhill, G. Liu, Chem. Mater. 12 (2000) 2082.
[17] Z. Lu, G. Liu, H. Phillips, J.M. Hill, J. Chang, R.A. Kydd, Nano Lett. 1 (2001) 683.
[18] A.B.R. Mayer, J.E. Mark, R.E. Morris, Polym. J. 30 (1998) 197.
[19] A.B.R. Mayer, J.E. Mark, Colloid Polym. Sci. 275 (1997) 333.
[20] R. Nakao, H. Rhee, Y. Uozumi, Org. Lett. 7 (2005) 163.
[21] L.M. Bronstein, D.M. Chernyshov, I.O. Volkov, M.G. Ezernitskaya, P.M. Valetsky,
V.G. Matveeva, E.M. Sulman, J. Catal. 196 (2000) 302.
We also studied stability of the heterogenized catalysts. All the
catalysts demonstrated no activity or selectivity changes at the re-
peated use after five consecutive runs (Table 6). The elemental
analysis data (Table 6) show no loss of active metal either, thus
suggesting exceptional stability of the NPs formed in PS-b-P4VP
and supported on alumina.
[22] J. Grunes, J. Zhu, G.A. Somorjai, Chem. Commun. 18 (2003) 2257.
[23] M. Boudart, J. Mol. Catal. 30 (1985) 27.
[24] D.R. Rolison, Science 299 (2003) 1698.
[25] K. Hayek, R. Kramer, Z. Paal, Appl. Catal. 162 (1997) 1.
[26] E. Sulman, Y. Bodrova, V. Matveeva, N. Semagina, L. Cerveny, V. Kurtc, L. Bron-
stein, O. Platonova, P. Valetsky, Appl. Catal. A 176 (1999) 75.
[27] A. Molnar, A. Sarkany, M. Varga, J. Mol. Catal. A Chem. 173 (2001) 185.
[28] V.V. Rusak, M.I. Zaretskii, A.S. Mozzhukhin, I.V. Usyshkina, L.A. Pushkina, Russ.
J. Appl. Chem. 67 (1994) 1066.
[29] S. Pattnaik, V.R. Subramanyam, M. Bapaji, C.R. Kole, Microbios 89 (1997) 39.
[30] S.J. Tauster, S.C. Fung, R.L. Garten, J. Am. Chem. Soc. 100 (1978) 170.
[31] D.C. Koningsberger, J.H.A. Martens, R. Prins, D.R. Short, D.E. Sayers, J. Phys.
Chem. 90 (1986) 3047.
[32] J.H.A. Martens, R. Prins, D.C. Koningsberger, Catal. Lett. 2 (1989) 211.
[33] P. Kacer, L. Cerveny, Appl. Catal. 229 (2002) 193.
[34] J. Barrault, A. Chafik, P. Gallezot, Appl. Catal. 67 (1991) 257.
[35] P. Ferreira-Aparicio, M. Fernandez-Garcia, A. Guerrero-Ruiz, I. Rodriguez-Ramos,
J. Catal. 190 (2000) 296.
[36] M. Antonietti, J. Conrad, A. Thuenemann, Macromolecules 27 (1994) 6007.
[37] M. Antonietti, S. Foerster, S. Oestreich, J. Hartmann, E. Wenz, Nachr. Chem. Lab.
Tech. 44 (1996) 579.
[38] M. Antonietti, E. Wenz, L. Bronstein, M. Seregina, Adv. Mater. 7 (1995) 1000.
[39] L.M. Bronstein, E.S. Mirzoeva, P.M. Valetsky, S.P. Solodovnikov, R.A. Register, J.
Mater. Chem. 5 (1995) 1197.
[40] T. Mallat, Z. Bodnar, B. Minder, K. Borszeky, A. Baiker, J. Catal. 168 (1997) 183.
[41] D.R. Miller, N.A. Peppas, Macromolecules 20 (1987) 1257.
[42] Y.G. Li, Y.M. Lee, J.F. Porter, J. Mater. Sci. 37 (2002) 1959.
[43] E. Brunet, M.J. de la Mata, O. Juanes, J.C. Rodriguez-Ubis, Angew. Chem. Int.
Ed. 43 (2004) 619.
[44] I.B. Tsvetkova, L.M. Bronstein, S.N. Sidorov, O.L. Lependina, M.G. Sulman, P.M.
Valetsky, B. Stein, L.Z. Nikoshvili, V.G. Matveeva, A.I. Sidorov, B.B. Tikhonov, G.N.
Demidenko, L. Kiwi-Minsker, E.M. Sulman, J. Mol. Catal. A Chem. 276 (2007)
116.
4. Conclusions
In this paper we demonstrated that deposition of NP containing
micellar catalysts on alumina causes no changes in the NP mor-
phology, but leads to different catalytic behavior depending on the
NP structure. In the absence of the acceptor metal-modifier (for
Pd and PdZn NPs), the key modifying factor is the increase of the
polarity of the system upon heterogenization. This leads to the in-
crease of the amount of catalytic sites and to the increase of the
catalytic activity. When a metal-modifier is the acceptor (for PdAu
and PdPt NPs), this leads to the electron deficiency of the surface
Pd atoms and most likely their stronger adsorption on alumina,
thus impeding access to some catalytic centers. This also results in
the emergence of an induction period, the length of which is con-
sistent with the acceptor ability of a modifying metal. Comparison
of the catalysts studied in this work with the commercial Pd/Al2O3
shows that the activity of the heterogenized micellar catalysts con-
taining mono- and bimetallic nanoparticles is much higher than
that of the commercial catalyst owing to lower activation energy
and/or higher amount of catalytic sites.
[45] A.J. Bard (Ed.), Encyclopedia of Electrochemistry of Elements, Dekker, New
York/Basel, 1976.
[46] J.R. Bowser, Inorganic Chemistry, Brooks-Cole Publishing Co., Pacific Grove, CA,
1993, p. 805.
Acknowledgments
[47] C. Wagner, W. Riggs, L. Davis, G. Mullenberg, Handbook of X-Ray Photoelectron
Spectroscopy, Perkin-Elmer Corporation, Minnesota, 1978.
[48] B.J. Tielsch, J.E. Fulghum, Surf. Interface Anal. 24 (1996) 422.
[49] B.H. Davis, in: G. Ertl, H. Knozinger, J. Weitkamp (Eds.), Handbook of Heteroge-
neous Catalysis, VCH, Weinheim, 1997, p. 13.
This work has been supported, in part, by the NATO Science for
Peace Program (grant SfP-981438) and, in part, by Russian Founda-
tion for Basic Research (grant 04-03-32928-a). We also thank Prof.
Dr. M. Antonietti for the PS-b-P4VP sample and Dr. J. Hartmann for
the TEM images of micellar samples.
[50] J.M. Thomas, W.J. Thomas, Introduction to the Principles of Heterogeneous
Catalysis, Academic Press, New York, 1967, p. 544.
[51] P. Gallezot, in: G. Ertl, H. Knozinger, J. Weitkamp (Eds.), Handbook of Hetero-
geneous Catalysis, VCH, Weinheim, 1997, p. 2209.
References
[52] B. Delmon, in: G. Ertl, H. Knozinger, J. Weitkamp (Eds.), Handbook of Hetero-
geneous Catalysis, VCH, Weinheim, 1997, p. 264.
[53] J.R. Anderson, Structure of Metallic Catalysts, Academic Press, London/New
York/San Francisco, 1975, p. 462.
[1] G.A. Somorjai, A.M. Contreras, M. Montano, R.M. Rioux, Proc. Natl. Acad.
Sci. 103 (2006) 10577.
[2] C.R. Henry, Surf. Sci. Rep. 31 (1998) 231.

 Sulman, Esther M.
Sulman, Esther M.
 Matveeva, Valentina G.
Matveeva, Valentina G.
 Sulman, Mikhail G.
Sulman, Mikhail G.
 Demidenko, Galina N.
Demidenko, Galina N.
 Valetsky, Pyotr M.
Valetsky, Pyotr M.
 Stein, Barry
Stein, Barry
 Mates, Tom
Mates, Tom
 Bronstein, Lyudmila M.
Bronstein, Lyudmila M.