.
Angewandte
Communications
Biosynthesis
Structures of Iridoid Synthase from Cantharanthus roseus with Bound
NAD+, NADPH, or NAD+/10-Oxogeranial: Reaction Mechanisms
Yumei Hu, Weidong Liu, Satish R. Malwal, Yingying Zheng, Xinxin Feng, Tzu-Ping Ko, Chun-
Chi Chen, Zhongxia Xu, Meixia Liu, Xu Han, Jian Gao, Eric Oldfield,* and Rey-Ting Guo*
Abstract: Structures of the iridoid synthase nepetalactol
synthase in the presence of NAD+, NADPH or NAD+/10-
oxogeranial were solved. The 10-oxogeranial substrate binds in
a transoid-O1-C3 conformation and can be reduced by hydride
addition to form the byproduct S-10-oxo-citronellal. Tyr178
Oz is positioned 2.5 from the substrate O1 and provides the
second proton required for reaction. Nepetalactol product
formation requires rotation about C1–C2 to form the cisoid
isomer, leading to formation of the cis-enolate, together with
rotation about C4–C5, which enables cyclization and lactol
production. The structure is similar to that of progesterone-5b-
reductase, with almost identical positioning of NADP, Lys146-
(147), Tyr178(179), and F342(343), but only Tyr178 and
Phe342 appear to be essential for activity. The transoid 10-
oxogeranial structure also serves as a model for b-face hydride
attack in progesterone 5b-reductases and is of general interest
in the context of asymmetric synthesis.
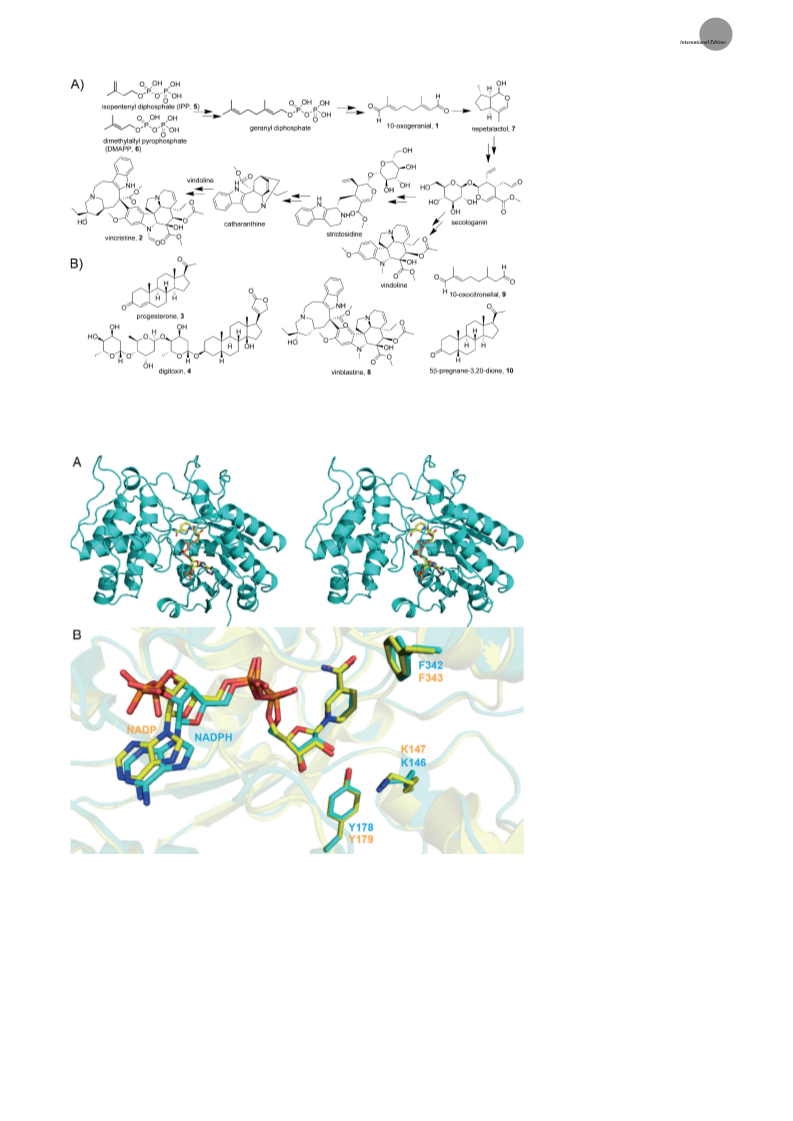
substrates or other ligands. The structures of the iridoid
synthases are of interest because their products are converted
into important natural products, compounds such as the
anticancer drug vincristine (2), as well as being of mechanistic
interest, not least because their amino acid sequences have
high homology to the plant progesterone (3) 5b-reductases
involved in the formation of cardiac glycoside drugs such as
digitoxin (4). A simplified version of the biosynthesis of
vincristine (2) from isopentenyl diphosphate (5) and dime-
thylallyl diphosphate (6) is shown in Scheme 1A; the
structures of other molecules discussed herein are shown in
Scheme 1B.
We expressed and purified an iridoid synthase, Canthar-
anthus roseus (1R, 4aS, 7S, 7aR) nepetalactol (7) synthase,
from the corresponding chemically synthesized gene (Gene-
bank accession number: K7WDL7.1). C. roseus is the
Madagascar periwinkle and produces the anticancer indole
terpenes vincristine (2) and vinblastine (8) from iridoids.
Attempts to crystallize full-length protein were unsuccessful,
so we attempted crystallization of two N-terminus-truncated
variants, DN13 and DN25. DN25 crystals were obtained
(details of protein expression, purification, and crystallization
are given in the Supporting Information). The activity of the
DN25 construct was (within experimental error) the same as
that of the wild-type protein (Figure S1 in the Supporting
Information). We then co-crystallized this IRIS with either
NAD+, NADPH, or NAD+ + 10-oxogeranial and solved the
structures. Full data acquisition and refinement details are
given in Table S1 in the Supporting Information.
We first obtained the structures of IRIS with either
NADPH or NAD+ bound. A stereo view of the NADPH
structure is shown in Figure 1A; the NAD+ structure is
virtually identical (a 0.21 root mean square deviation,
rmsd).The protein fold is most similar to that of progesterone
5b-reductase from Digitalis lanata, a plant that produces
cardiac glycosides such as 4. There is a 1.0 Ca rmsd over 351
residues (using the PDBe Fold Server[5]). An alignment of the
active sites of the IRIS protein (+ NADPH, PDB ID: 5DBF)
and the D. lanata 5b-reductase (+ NADP+, PDB ID code
2V6G[6]) is shown in Figure 1B. The two active-site structures
are very similar and interestingly, the Tyr179 shown by site-
directed mutagenesis to be essential for progesterone 5b-
reductase activity[6] occupies the same position in both the
IRIS (Tyr178) and progesterone 5b-reductase structures, as
do the NADP cofactors and the Lys146 (Lys147 in progester-
one 5b-reductase) residues, which have been proposed to be
involved in progesterone 5b-reductase catalysis. There are,
therefore, great similarities between the IRIS and progester-
one 5b-reductase structures, with the Tyr Oz being in close
T
erpenes and related isoprenoids are the largest class of
small-molecule natural products on earth, and the most
abundant by mass.[1] Most are made by enzymes that are
represented by a relatively small number of folds,[2] primarily
head-to-tail and head-to-head trans-prenyltransferases, cis-
prenyltransferases, and a range of cyclases with one, two, or
three domains. In most cases, the reactions proceed via the
formation of carbocationic transition states/reactive inter-
mediates. However, in recent work, an alternative route to the
formation of terpenes—one involving reductive cyclization—
was reported for iridoid biosynthesis.[3] Iridoids are mono-
terpenes that are produced from (C10) 10-oxogeranial (1) by
NAD(P)H-dependent reduction, followed by a cyclization
step that involves either a Diels–Alder cycloaddition or
a Michael addition.[3a,4] There are no X-ray structures
available for any iridoid synthases (IRISs), with or without
[*] Y. Hu,[+] Prof. W. Liu,[+] Prof. Y. Zheng, Prof. C.-C. Chen, Z. Xu, M. Liu,
X. Han, J. Gao, Prof. R.-T. Guo
Tianjin Institute of Industrial Biotechnology
Chinese Academy of Sciences, Tianjin 300308 (China)
E-mail: guo_rt@tib.cas.cn
Dr. S. R. Malwal, Dr. X. Feng, Prof. E. Oldfield
Department of Chemistry, University of Illinois
Urbana, IL 61801 (USA)
E-mail: eo@chad.scs.uiuc.edu
Dr. T.-P. Ko
Institute of Biological Chemistry, Academia Sinica
Taipei 11529 (Taiwan)
[+] These authors contributed equally to this work.
Supporting information for this article is available on the WWW
15478
ꢀ 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2015, 54, 15478 –15482

 Hu, Yumei
Hu, Yumei
 Liu, Weidong
Liu, Weidong
 Malwal, Satish R.
Malwal, Satish R.
 Zheng, Yingying
Zheng, Yingying
 Feng, Xinxin
Feng, Xinxin
 Ko, Tzu-Ping
Ko, Tzu-Ping
 Chen, Chun-Chi
Chen, Chun-Chi
 Xu, Zhongxia
Xu, Zhongxia
 Liu, Meixia
Liu, Meixia
 Han, Xu
Han, Xu
 Gao, Jian
Gao, Jian
 Oldfield, Eric
Oldfield, Eric
 Guo, Rey-Ting
Guo, Rey-Ting