1324
E. Brunet et al. / Tetrahedron Letters 48 (2007) 1321–1324
2H, C(2)H, C(10)H], 2.52 [m, 1H, C(30)H], 2.33 [d, 1H,
C(400)H], 3.00 [s, 3H, C(6)OCH3], 2.93 [dc, 1H, J = 2.4,
7.2, C(2)H], 2.77 [m, 1H, C(10)H], 2.51 [m, 1H, C(30)H],
2.35 [d, 1H, J = 15.1, C(200)H], 2.21 [s, 6H, C(30)N(CH3)2],
1.91–2.02 [m, 2H, C(4)H, C(14)H], 1.55–1.71 [m, 2H,
C(7)H, C(40)CH], 1.45–1.55 [m, 5H, C(6)CH3, C(14)CH,
C(200)H], 1.35 [d, 3H, J = 7.4, C(10)H], 1.05–1.26 [m, 23H,
C(2)H3, C(4)H3, C(7)H, C(8)H3, C(12)CH3, C(40)H,
C(50)CH3, C(300)CH3, C(500)CH3], 0.83 [t, 3H, J = 7.7,
C(14)CH3), 0.13 [s, 9H, C(400)OSi(CH3)3], 0.07 [s, 9H,
C(20)OSi(CH3)3]; 13C NMR (75 MHz, CDCl3) d (ppm):
176.0, 174.5, 166.2, 159.3, 115.6, 102.3, 96.3, 80.7, 79.5,
79.3, 78.2, 78.1, 77.6, 75.7, 73.2, 73.0, 67.0, 65.1, 65.0,
62.3, 50.4, 49.5, 45.1, 38.8, 37.1, 35.8, 35.7, 29.4, 27.9, 22.1,
21.9, 21.6, 20.9, 19.4, 19.3, 17.3, 16.0, 15.4, 10.5, 9.8, 1.2,
0.8.
J = 15.3, C(200)H], 2.22 [s, 6H, C(30)N(CH3)2], 2.03 (s
ancho, 1H, OH), 1.87–2.01 [m, 2H, C(4)H, C(14)H], 1.57–
1.73 [m, 3H, C(7)H2, C(40)H], 1.50 [m, 1H, C(13)H], 1.45
[dd, 1H, J = 4.9, 15.0, C(200)H], 1.42 [s, 3H, C(6)CH3], 1.31
[d, 1H, J = 7.2, C(10)CH3], 1.25 [s ancho, 1H, C(40)H],
1.22 [s, 3H, C(12)CH3], 1.15 [d, 3H, J = 6.9, C(8)CH3],
1.14 [d, 3H, J = 6.9, C(2)CH3], 1.12 [s, 3H, C(300)CH3],
1.12 [d, 3H, J = 6.9, C(50)CH3], 1.07 [d, 3H, J = 6,
C(4)CH3], 1.05 [d, 3H, J = 6.9, C(500)CH3], 0.87 [t, 3H,
J = 7.1, C(14)CH3], 0.10 [s, 9H, C(400)OSi(CH3)3], 0.09 (s,
9H, C(20)OSi(CH3)3); 13C NMR (75 MHz, CDCl3) d
(ppm): 177.1, 176.0, 165.7, 159.5, 116.3, 102.7, 96.5,
81.8, 80.9, 79.3, 77.2, 77.05, 75.3, 74.5, 73.2, 73.1, 70.2,
67.7, 65.2, 65.0, 44.8, 40.9, 40.3, 38.3, 35.8, 34.3, 30.3, 29.7,
28.0, 27.0, 22.1, 21.7, 21.4, 19.2, 18.6, 16.3, 15.5, 14.9, 10.8,
9.6, 1.0, 0.8.
9. CCDC 626463–626465 contains the supplementary crys-
tallographic data for this Letter. These data can be
obtained free of charge from The Cambridge Crystallo-
25
1
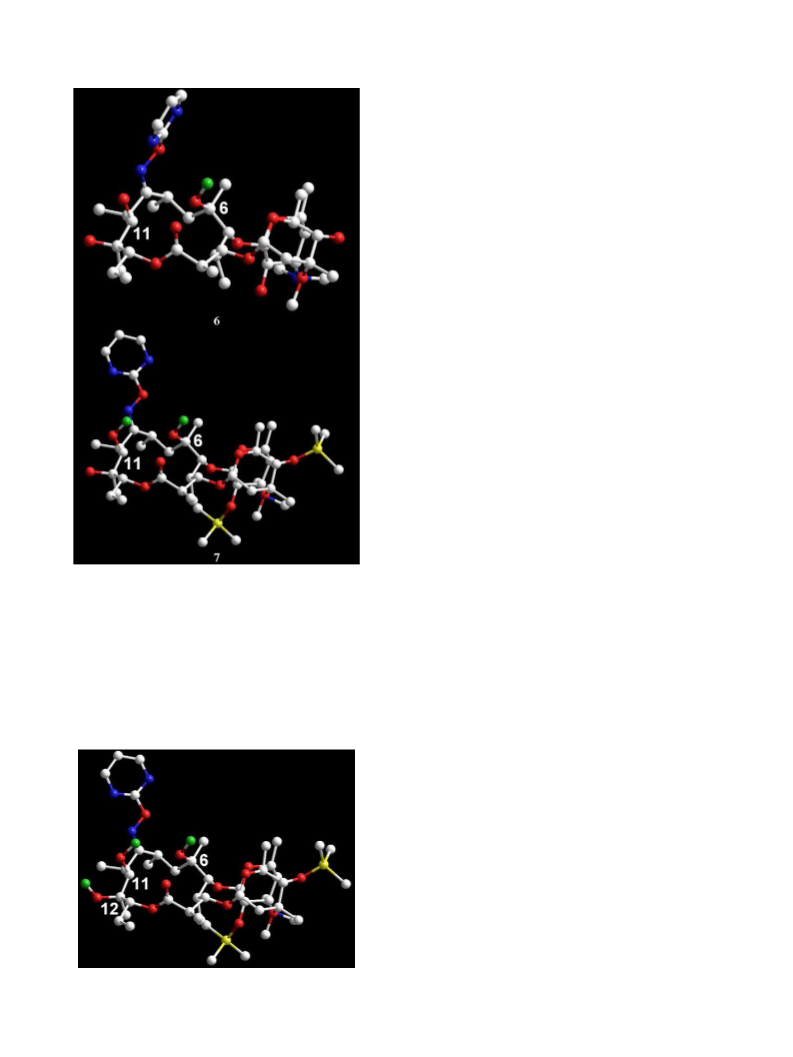
7. Mp 172–174 °C; ½aꢀD ꢁ151.65 (c 0.1, CH2Cl2); H NMR
(500 MHz, CDCl3) (ppm): 8.56 [d, 2H, J = 4.8,
d
C(3Ar)H], 6.97 [t, 1H, J = 4.8, C(4Ar)H], 5.19 [dd, 1H,
J = 11.2, 2.1, C(13)H], 5.09 (s ancho, 1H, OH), 4.84 [d,
1H, J = 5.1, C(100)H], 4.39 [d, 1H, J = 7.6, C(10)H], 4.16
[dc, 1H, J = 6.1, 9.4, C(500)H], 3.92 [m, 1H, C(8)H], 3.84 [s,
1H, C(11)H], 3.70 [d, 1H, J = 10.7, C(3)H], 3.61 [m, 2H,
C(5)H, C(50)H], 3.28 [s, 3H, C(300)OCH3], 3.05–3.23 [m,
2H, C(20)H, C(400)H], 2.86 [dc, 1H, J = 7.3, 9.5, C(2)H],
2.74 [s, 4H, C(6)OCH3, C(10)H], 2.50 [s ancho, 1H,
C(30)H], 2.32 [d, 1H, J = 14.7, C(200)H], 2.20 [s, 6H,
C(30)N(CH3)2], 1.95 [m, 1H, C(14)H], 1.82 [m, 1H, C(4)H],
1.54–1.66 [m, 3H, C(14)H, C(7)H2], 1.43–1.54 [m, 2H,
C(40)H, C(200)CH], 1.40 [s, 3H, C(6)CH3], 1.29 [d, 3H,
J = 6.8, C(10)CH3], 1.26 [m, 1H, C(40)H], 1.23 [s, 3H,
C(300)CH3], 1.20 [s, 3H, C(12)CH3], 1.16 [d, 3H, J = 6.8,
C(2)CH3], 1.14 [d, 3H, J = 6.8, C(50)CH3], 1.12 [d, 3H,
J = 6.8, C(50)CH3], 1.11 [d, 3H, J = 6.8, C(8)CH3], 1.04 [d,
3H, J = 6.8, C(4)CH3], 0.84 [t, 3H, J = 6.9, C(14)CH3],
0.10 (s, 9H, C(400)OSi(CH3)3), 0.08 (s, 9H, C(20)OSi-
(CH3)3). 13C NMR (75 MHz, CDCl3) d (ppm): 176.1,
175.7, 165.9, 159.5, 116.0, 102.5, 96.2, 81.8, 80.8, 79.0,
77.7, 76.7, 75.3, 74.3, 73.3, 73.1, 70.1, 67.1, 65.2, 65.1, 50.8,
49.6, 45.2, 41.0, 39.2, 37.8, 35.8, 34.1, 31.8, 29.4, 27.8, 22.6,
22.2, 20.4, 19.4, 18.6, 16.3, 16.1, 15.1, 10.6, 9.8, 1.03, 0.84.
10. Kawashima, Y.; Morimoto, S.; Matsunaga, T.; Kashi-
mura, M.; Adachi, T.; Watanabe, Y.; Hatayama, K.;
Hirono, S.; Moriguchi, I. Chem. Pharm. Bull. 1990, 38,
1485; Duran, D.; Aviyente, V.; Baysal, C. J. Comput. Mol.
Design 2004, 18, 145.
25
11. Mp 181–183 °C; ½aꢀD ꢁ103.61 (c 0.104, CH2Cl2); 1H
NMR (500 MHz, CDCl3) d (ppm): 8.55 [d, 2H, J = 4.9,
C(3Ar)H], 6.93 [t, 1H, J = 4.9, C(4Ar)H], 5.52 [dd, 1H,
J = 1.7, 11.6, C(13)H], 4.87 [d, 1H, J = 4.8, C(100)H], 4.38
[d, 1H, J = 6.9, C(10)H], 4.23 [dc, 1H, J = 9.2, 6.1,
C(500)H3], 3.44–3.83 [m, 6H, C(3)H, C(8)H, C(50)H,
C(11)OCH3], 3.40 [s, 3H, C(12)OCH3], 3.29 [s, 3H,
C(300)OCH3], 2.97–3.23 [m, 7H, C(5)H, C(10)H, C(20)H,
C(400)H, C(6)OCH3], 2.92 [m, 1H, C(2)H], 2.50 [m, 1H,
C(30)H], 2.35 [d, 1H, J = 14.7, C(200)H], 2.20 [s, 6H,
C(30)N(CH3)2] 1.96 [m, 1H, C(4)H], 1.80 [m, 1H, C(14)H],
1.49–1.71 [m, 4H, C(7)H, C(14)H, C(40)H, C(200)H], 1.46,
(s, 3H, C(6)H3], 1.37 [d, 4H, J = 7.7, C(7)H, C(10)CH3],
1.21–1.31 [m, 4H, C(40)H, C(500)CH3], 1.22 [s, 3H,
C(12)CH3], 1.19, [d, 3H, J = 6.3, C(2)CH3], 1.18 [d, 3H,
J = 7.2, C(8)CH3], 1.15 [s, 3H, C(300)CH3], 1.14 [d, 3H,
J = 5.3, C(50)CH3], 1.07 [d, 3H, J = 7.2, C(4)CH3], 0.86 [t,
3H, J = 7.4, C(14)CH3], 0.13 [s, 9H, C(400)OSi(CH3)3),
0.01 [s, 9H, C(20)OSi(CH3)3]; 13C NMR (75 MHz, CDCl3)
d (ppm): 175.6, 174.9, 165.8, 159.0, 115.3, 102.4, 96.4, 80.9,
80.5, 79.4, 78.3, 75.8, 72.7, 66.8, 64.9, 61.2, 53.3, 50.0, 49.3,
44.9, 40.7, 38.8, 37.2, 35.6, 29.3, 28.2, 21.8, 21.6, 19.4, 19.0,
15.8, 14.3, 10.1, 9.6, 0.6, 0.5.
25
8. Mp 190–192 °C; ½aꢀD ꢁ125.17 (c 0.102, CH2Cl2); 1H
NMR (500 MHz, CDCl3) d (ppm): 8.56 [d, 2H, J = 4.8,
C(3Ar)H], 6.94 [t, 1H, J = 4.8, C(4Ar)H], 5.00 [dd, 1H,
J = 2.3, 11.0, C(13)H], 4.88 [d, 1H, J = 5.0, C(100)H], 4.43
[d, 1H, J = 7.2, C(10)H], 4.21 (dc, 1H, J = 6.3, 9.5,
C(500)H], 3.98 [m, 1H, C(8)H], 3.86 [s, 3H, C(11)OCH3],
3.66–3.73 [m, 2H, C(3)H, C(11)H], 3.59–3.64 [m, 2H,
C(50)H, C(12)OH], 3.30 [s, 3H, C(300)OCH3], 3.21 [m, 1H,
C(5)H], 3.15 [m, 1H, C(20)H], 3.13 [d, 1H, J = 9.4,
12. Abuga, K. O.; Chepkwony, H. K.; Roets, E.; Hoogmar-
tens, J. J. Sep. Sci. 2001, 24, 849; Nightingale, C. H. Clin.
Drug Invest. 2000, 19, 293.

 Brunet, Ernesto
Brunet, Ernesto
 Mu?oz, Dulce María
Mu?oz, Dulce María
 Parra, Francisco
Parra, Francisco
 Mantecón, Susana
Mantecón, Susana
 Juanes, Olga
Juanes, Olga
 Rodríguez-Ubis, Juan Carlos
Rodríguez-Ubis, Juan Carlos
 Carmen Cruzado, Ma
Carmen Cruzado, Ma
 Asensio, Ramón
Asensio, Ramón