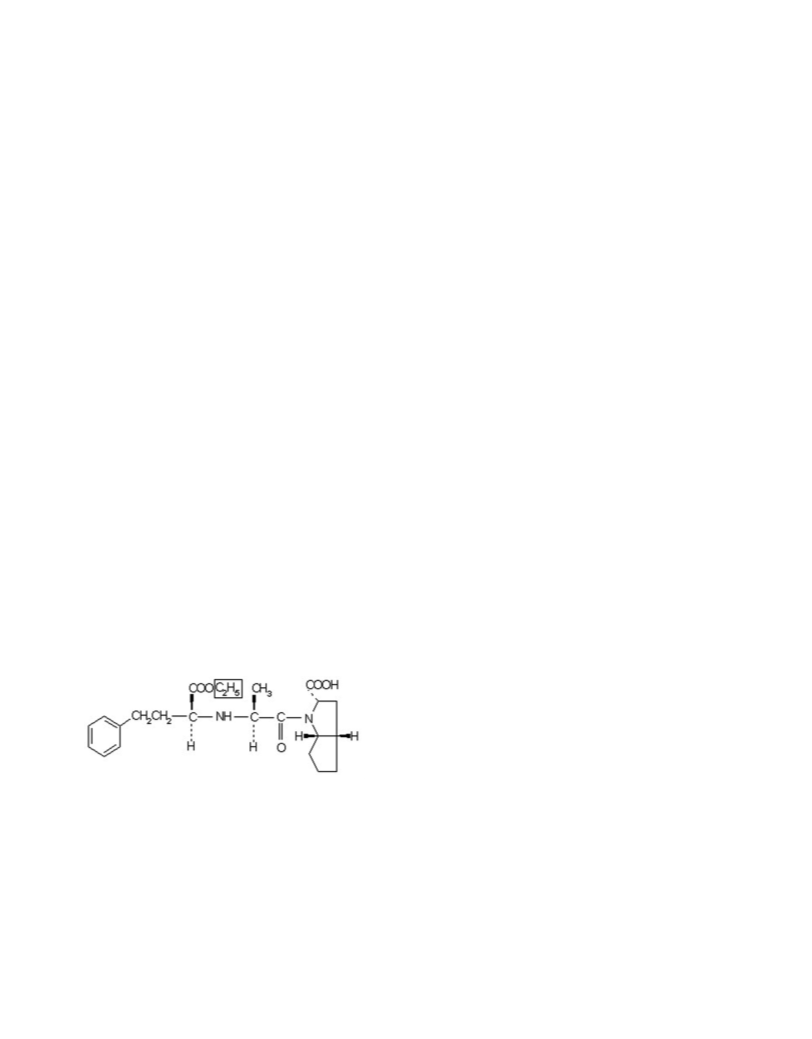
J. Chil. Chem. Soc., 55, Nº 4 (2010)
It was established by chromatographic analysis of all stressed samples
as explained in section sꢅꢄꢃss ꢅꢃsꢅꢁꢀꢋ). The composition, pH and the flow rate
of the mobile phase were modified to optimize separation between ramipril,
degradation products and the internal standard.
were treated as explained in section pꢄꢃpꢉꢄꢉꢅꢁꢇꢀ ꢇf sꢉmpꢊꢃ, and the times of
sonication evaluated were 5, 10 and 15 min. The percentage of ramipril was
found to be 96,6 ± 0,1%, 99,8 ± 0,8% and 99,4% ± 0,3% (mean ± SD, n =
6) for 5, 10 and 15 minutes of sonication respectively, therefore a 10 minutes
sonication time was selected in the proposed method.
(
mꢀꢍꢅꢂꢄ ꢒꢉꢊꢇꢄꢉꢍꢇꢂꢋ
The method was validated according to the ICH guidelines for validation
mꢀꢍꢅꢂꢄ ꢒꢉꢊꢇꢄꢉꢍꢇꢂꢋ
Linearity
The calibration curve of ramipril was linear over the concentration range
of 50.0 to 300.0 mg/mL (6 different concentrations were used and each solution
was injected 5 times). The equation of the standard curve based on the ratio of
the peak heights of ramipril / internal standard to the ramipril concentration
1
3
of analytical procedures . The parameters validated were linearity, precision,
accuracy, selectivity, robustness, detection and quantitation limits, and stability
of standard solution.
Sꢍꢃꢀss ꢍꢀsꢍꢇꢋꢌ
2
Stress testing was carried out according to the ICH stability testing
was ꢆ = 0.0052x + 0.0404; r = 0.9955.
1
guidance . Ramipril was stressed under various conditions until to facilitate
1
4
approximate 5-20% degradation . For each condition, a blank solution was
prepared and was subjected to stress in the same manner as the drug, also a
control solution of ramipril was prepared, which was stored without the stress
condition.
Precision
The intra-day precision was determined by analysis of three different
preparations in concentrations of 160.0, 200.0 and 240.0 mg/mL on the same
day. The inter-day precision was studied by comparing the assays on three
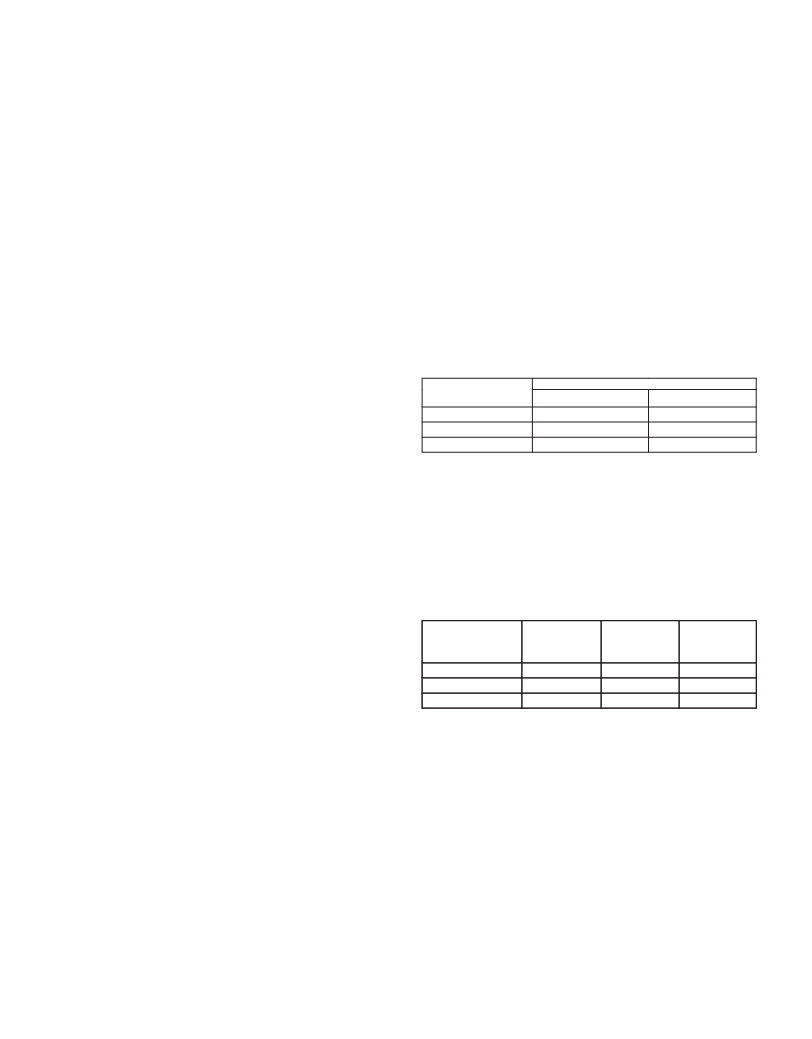
different days. The results are shown in Table 1. The obtained values show a
suitable precision for the analytical method.
a) Hydrolysis acidic, neutral and alkaline:
A solution of ramipril 10 mg/mL was prepared in methanol, then an aliquot
of 2 mL was transferred to a 10 mL volumetric flask and diluted with 0.1 N
HCl, water and 0.1 N NaOH to volume. Samples of 3 mL were kept on a hot
plate at 60 ºC for basic hydrolysis for 5 min, and at 80 ºC for acid and neutral
hydrolysis for 30 h. After which they have been cooled to room temperature,
then the samples were transferred to a 25 mL volumetric flask, neutralized, a
tꢉꢑꢊꢀ 1. Precision determined during method validation.
Relative standard deviation (%)
Concentration
mg/mL)
60.00
200.00
40.00
a)
b)
(
intra-day
0.71
inter-day
1.47
1
1
mL of internal standard (3.1 mg/mL) was added, and they were diluted to
1.39
1.84
volume.
2
0.73
0.27
a)
b)
Analyzed on the same day (n=3).
b) Oxidation:
Analyzed on three different days (n=9).
A solution of ramipril 10 mg/mL was prepared in methanol, then an
aliquot of 2 mL was transferred to a 10 mL volumetric flask and diluted with
Accuracy
3
% H O Samples of 3 mL were kept on a hot plate at 80 ºC for 6 h and at room
2 2.
To evaluate the accuracy of the method, recovery test were performed by
adding know amounts of standard of ramipril in the level 80, 100 and 120 %
of the ramipril levels in the tablets (three replicates of each level) to common
tablet excipients (lactose, starch and magnesium stearate). The accuracy of the
assay was determined by comparing the found amount with the added amount.
The results obtained are shown in Table 2. The obtained values confirm the
accuracy of the proposed method.
temperature (25 ± 2ºC) for 7 days in the dark, then they were transferred to a 25
mL volumetric flask, a 1 mL of internal standard (3.1 mg/mL) was added, and
they were diluted to volume.
c)
Thermal degradation:
About 50 mg of ramipril was exposed to dry heat at 70ºC in an oven for
2
1 days. Then a solution of ramipril 200 mg/mL with internal standard 125 mg/
mL was prepared.
tꢉꢑꢊꢀ 2. Recovery percentage of ramipril determined during method
validation.
d)
Photostability:
Sample
Level
(
Added
Amount
(mg)
Found
An aqueous/methanolic solution of ramipril 2 mg/mL and solid drug in 1
mm layer in a petri-plate, were exposed to UV (365 nm) and VIS radiation for
days. Then solutions of ramipril 200 mg/mL with internal standard 125 mg/
Recovery
Amount
b)
(%)
a)
%)
(mg)
8
mL were prepared. Dark controls were run simultaneously.
80
2.00
2.50
3.00
2.00 ± 0.02
2.48 ± 0.05
2.99 ± 0.04
99.85
99.29
99.53
1
00
reSultS and diScuSSion
1
20
a)
b)
Mean ± SD (n= 3).
(Found amount/ Added amount) x 100.
Sꢍꢉꢑꢇꢊꢇꢍꢁ-ꢇꢋꢄꢇꢈꢉꢍꢇꢋꢌ lc ꢆꢀꢍꢅꢂꢄ ꢄꢀꢒꢀꢊꢂꢏꢆꢀꢋꢍ ꢉꢋꢄ ꢂꢏꢍꢇꢆꢇzꢉꢍꢇꢂꢋ
The main target of the stability-indicating chromatographic method is to
get the separation between ramipril and their degradation products. Some of the
reported method for ramipril used gradient and/or a high column temperature,
which requires more sophisticated instrumentation and/or long stabilization
Selectivity
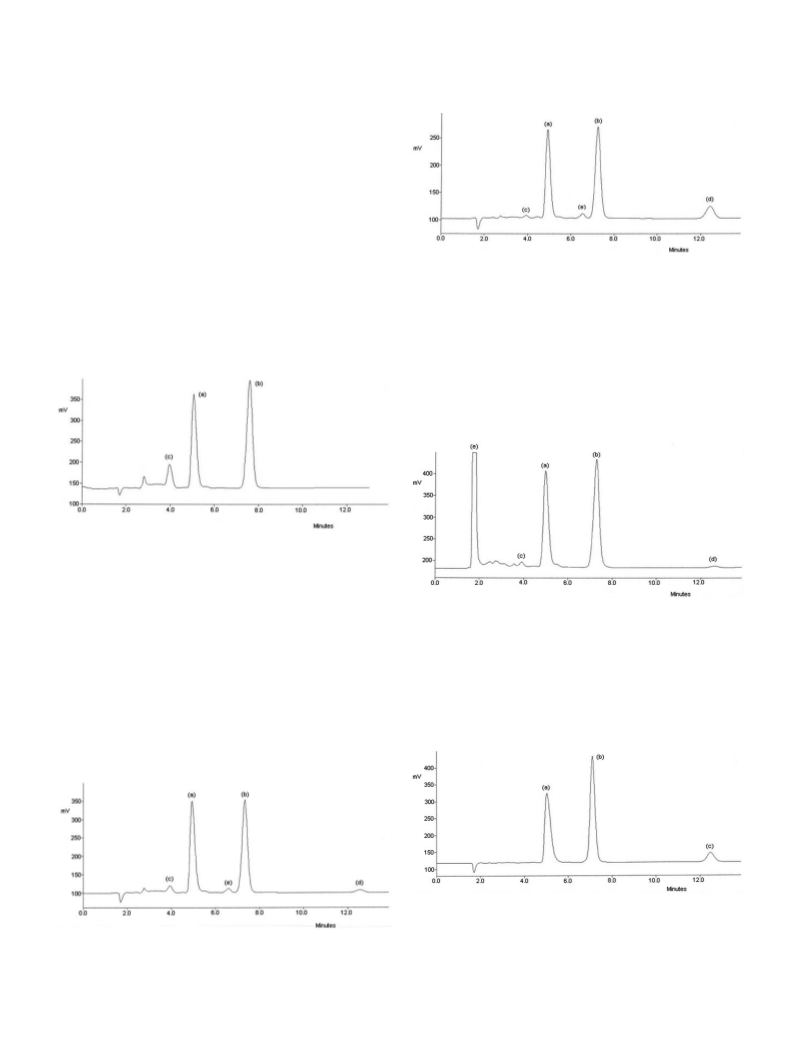
Results from the stress testing studies indicated that the method is selective
towards ramipril, degradation products and internal standard as shown in Fig. 2
to 6. Cloxacillin was selected as internal standard due to its suitable retention,
sharp peak and similar physicochemical properties to ramipril. The selectivity
was also evaluated by observing any interference from excipients used in the
tablets; therefore a sample of each commercial product was analyzed, showing
no peaks that interfered with ramipril, degradation products or internal
standard. These results proved the selectivity of the proposed method.
5, 7, 8
times
. In the present study different proportions of methanol (50, 55,
6
0, 65 %) and change in pH (2.2, 2.4, 2.6) were evaluated before the final
chromatographic conditions were selected. Also acetonitrile was tested instead
of methanol, but peak shapes were not satisfactory. From these studies it was
determined that the pH of the mobile phase is very important in order to avoid
peak tailing. Optimal separation of ramipril, degradation products and internal
standard, with sharp peaks was achieved with a methanol – tetrahydrofuran –
phosphate buffer (pH 2.4; 0.01M) (55:5:40, v/v/v) mobile phase under isocratic
conditions at room temperature. The evaluated system suitability parameters
were: rs 4,7 and 2,2 (between ramipril / internal standard and ramipril /
ramiprilat); ꢉ 1,5 and 1,3 (between ramipril / internal standard and ramipril /
ramiprilat); n 2600 and t 1,1 (for ramipril). Fig. 2 to 6 shows chromatograms
of stressed samples and internal standard, which prove the stability-indicating
capability of the assay.
Robustness
To determine robustness of the method, variation of flow rate from 0.8
to 1.2 and the amount of organic solvent in the mobile phase from 53 to 57
were realized and resolution between ramipril and internal standard were
evaluated. At all conditions the resolution was found to be greater than 4 which
proved the robustness of the method.
%
Detection and quantitation limits
Sꢉꢆꢏꢊꢀ ꢏꢃꢀꢏꢉꢃꢉꢍꢇꢂꢋ sꢍꢎꢄꢇꢀs
A study was performed in order to determine the optimum sonication time
for complete leaching of the ramipril from tablets. Ramipril tablets 2,5 mg
The DL and QL were calculated by using the equations: DL = 3.3 x s/ S;
QL = 10 x s/ S, where s is the standard deviation of the response and S is the
slope of the calibration curve. DL was 0.24 mg/mL and QL was 0.74 mg/mL.
4
51

 De Diego
De Diego
 Godoy
Godoy
 Mennickent
Mennickent
 Olivares
Olivares
 Godoy
Godoy