8
S. M. IVANOV ET AL.
Table 2. The antibacterial and antifungal activities of compounds 5a and 5b.
The inhibition of growth (%)
ꢃ
Bacteria
Fungi
Cryptococcus
Staphylococcus
aureus
Escherichia
coli
Klebsiella
pneumonia
Pseudomonas
aeruginosa
Acinetobacter
baumannii
Candida
albicans
Compound
neoformans var. Grubii
Concentration
5a
5b
13.84
–3.3
–1.71
2.01
5.48
18.13
10.72
18.32
22.39
–3.36
2.09
5.55
–11.62
1.75
32 lg/mL
32 lg/mL
ꢃThe growth rates for all bacteria and fungi has a variation of –/þ 10%, which is within the reported normal distribution of bacterial/fungal growth.
C13H22N4Se. Calculated, m/z: 315.1083. Anal. Calcd for 4.6.1. 3-tert-Butyl-8-methylselanylpyrazolo[5,1-c][1, 2,
C13H22N4Se: C, 49.84; H, 7.08; N, 17.88. Found: C, 49.82;
H, 7.15; N, 17.75.
4]triazin-4(1H)-one 10
M.p. 205–215 ꢀC (sublimation). IR (KBr) ꢀmax, cmꢄ1: 3223,
3151 (NH), 3073, 3037, 2970, 2932, 2909, 2821 (CH), 1697,
1675 (C ¼ O), 1602, 1561, 1527, 1479, 1458, 1422, 1392,
1362, 1340, 1282, 1272, 1197, 1159, 1138, 1075, 1022, 982,
4.5.2. 3-tert-Butyl-1,4-dihydropyrazolo[5,1-c][1, 2, 4]
triazine 8
1
938, 922, 838, 768, 683, 636, 620, 574, 527, 509. H NMR
M.p. 140–150 ꢀC (sublimation). IR (KBr) ꢀmax, cmꢄ1: 3272,
3229, 3176, 3131 (NH), 3088, 3012, 2974, 2937, 2903, 2869
(CH), 1576, 1561 (C ¼ N), 1479, 1467, 1420, 1393, 1364,
1353, 1281, 1259, 1231, 1211, 1164, 1099, 1065, 1050, 1017,
997, 970, 939, 897, 875, 860, 836, 785, 729, 694, 663, 626,
(300 MHz, CDCl3), d, ppm (J, Hz): 1.46 (9H, s, C(CH3)3);
2
2.16 (1H, s, JSe,H ¼ 11.8, SeCH3), 7.98 (1H, s, C(7)–H);
10.55 (1H, s, NH). 13C NMR (126 MHz, CDCl3), d, ppm (J,
1
Hz): 10.39 (SeCH3, JSe,C ¼ 60.5); 28.01 (C(CH3)3); 37.30
1
(C(CH3)3); 81.87 (C(8), JSe,C ¼ 127.9); 144.18, 146.77,
148.34 (C(3), C(4), C(8a)); 149.82 (C(7)–H). HRMS: Found,
m/z: 309.0229 [M þ Na]þ. C10H14N4OSe. Calculated, m/z:
309.0225. Anal. Calcd for C10H14N4OSe: C, 42.11; H, 4.95;
N, 19.64. Found: C, 42.05; H, 4.98; N, 19.72.
1
598, 567, 501, 480, 434. H NMR (300 MHz, DMSO-d6), d,
ppm (J, Hz): 1.15 (9H, s, C(CH3)3); 4.70 (2H, s, C(4)H2);
3
3
5.43 (1H, d, JH,H ¼ 1.9, C(8)–H), 7.29 (1H, d, JH,H ¼ 1.9,
C(7)–H); 10.10 (1H, s, NH). 13C NMR (APT, 126 MHz,
DMSO-d6), d, ppm: 27.80 (C(CH3)3); 36.84 (C(CH3)3); 42.56
(C(4)H2); 84.66 (C(8)–H); 138.46, 146.87 (C(3), C(8a));
139.24 (C(7)–H). HRMS: Found, m/z: 179.1297 [M þ H]þ.
C9H14N4. Calculated, m/z: 179.1291. Anal. Calcd for
C9H14N4: C, 60.65; H, 7.92; N, 31.43. Found: C, 60.53; H,
7.95; N, 31.49.
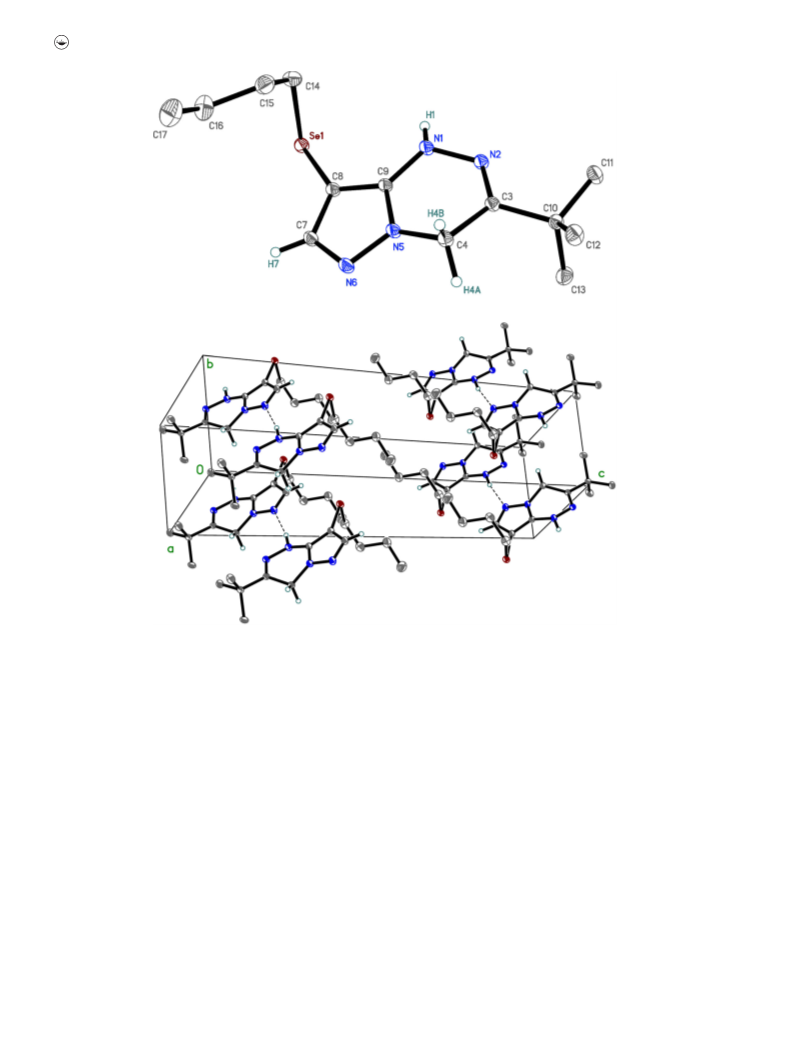
4.6.2. 8,8’-Diselanediylbis(3-tert-butylpyrazolo[5,1-c][1, 2,
4]triazin-4(1H)-one) 11
M.p. 260–263 ꢀC (decomp.). IR (KBr) ꢀmax, cmꢄ1: 3219,
3137 (NH), 2968, 2935, 2907, 2819 (CH), 1718, 1702, 1686
(C ¼ O), 1595, 1561, 1523, 1477, 1458, 1393, 1363, 1331,
1277, 1197, 1160, 1133, 1019, 982, 940, 921, 890, 839, 766,
1
731, 678, 633, 620, 571, 508. H NMR (500 MHz, DMSO-
d6), d, ppm: 1.41 (18H, s, 2C(CH3)3); 7.96 (2H, s, 2C(7)–H);
13.77 (2H, s, 2 NH). 13C NMR (126 MHz, DMSO-d6), d,
ppm (J, Hz): 27.82 (C(CH3)3); 36.73 (C(CH3)3); 82.40 (C(8),
1JSe,C ¼ 155.1); 145.19, 145.37, 148.20, 149.77 (C(3), C(4),
C(7), C(8a)). 77Se NMR (1H–77Se HMBC, 600 MHz, DMSO-
d6), d, ppm: 365.88 (C(8)–Se). HRMS: Found, m/z: 565.0092
[M þ Na]þ. C18H22N8O2Se2. Calculated, m/z: 565.0092. Anal.
Calcd for C18H22N8O2Se2: C, 40.01; H, 4.10; N, 20.74.
Found: C, 39.92; H, 4.18; N, 20.67.
4.6. Synthesis of compounds 9–11 from the reaction
with methyllithium
To the prepared solution of compound 3 (0.74 mmol, MeLi
was used in the previous step), freshly powdered elemental
Se (85 mg, 1.08 mmol) was added in one portion. The result-
ing mixture was stirred vigorously at –97j for 30 min.
Then, the cooling bath was removed and the temperature
was slowly raised to 0 ꢀC in a period of 30 min. Next, crys-
talline KH2PO4 (1 g, 7.35 mmol) was added in one portion,
and a pre-cooled (0 ꢀC) water (3 mL) was added dropwise
with stirring over 2 min. Finally, EtOAc (30 mL) and water
(20 mL) were added. The resulting mixture was stirred vig-
orously at 0ꢀ–5 ꢀC for 5 min. The organic phase was sepa-
rated, and the mother liquor was further extracted with
EtOAc (3 ꢆ 25 mL). The combined organic phases were
dried with anhydrous MgSO4 and filtered. The solvents were
removed in vacuo to give a residue, which was purified by
flash chromatography (eluent EtOAc:hexane 1:5–1:2) to give
compounds 9, white powder (80 mg, 0.39 mmol, 53%), 10,
4.7. Synthesis of compounds 10 and 12 from the
reaction with tert-butyllithium
To the prepared solution of compound 3 (0.74 mmol, t-BuLi
was used in the previous step), freshly powdered elemental S
(50 mg, 1.56 mmol, for the synthesis of 12) or Se (0.12 g,
1.52 mmol, for the synthesis of 10) were added in one por-
tion. The resulting mixtures were stirred vigorously at
–97 ꢀ for 30 min. Then, the cooling bath was removed and
the temperature was slowly raised to –50 ꢀC in a period of
white powder (65 mg, 0.23 mmol, 31%), and 11, yellow crys- 15 min. Next, MeI (0.2 mL, 3.21 mmol, for the synthesis of
tals (20 mg, 0.037 mmol, 10%).
10) or PhCH2Cl (0.5 mL, 4.35 mmol, for the synthesis of 12)
were added in one portion. The resulting reaction mixtures
were stirred for 20 min (for 10) or 1 h (for 12). Next,
Spectral data and m.p. for compound 9 coincided with
those described in literature.[21]

 Ivanov, Sergey M.
Ivanov, Sergey M.
 Minyaev, Mikhail E.
Minyaev, Mikhail E.
 Mironovich, Lyudmila M.
Mironovich, Lyudmila M.