SHORT PAPER
29
Can N-Alkyl- and N-Arylimidazoles be Prepared Directly from Alcohols and
Phenols with N,N’-Carbonyldiimidazole?
Can
N-Alkyl-
and
N
-Ary
a
limidazole
l
s
be Pr
e
pared
e
D
irectlyfro
r
m
A
lcohols an
F
dPhenols
w
it
i
h
N
,
N
’-
s
C
arbonyl
c
diimidazole
h
?
er*
Ciba Specialty Chemicals, R&D Segment Coating Effects, WRO 1059.505, P.O.Box, CH-4002 Basel, Switzerland
Fax +41(61)6362115; E-mail: walter.fischer@cibasc.com
Received 27 August 2001; revised 4 October 2001
alents of CDI in dichloromethane at reflux for 5 hours. We
Abstract: The report that N-alkyl- and N-arylimidazoles can be
prepared directly by reactions of N,N’-carbonyldiimidazole (CDI)
with alcohols or phenols was shown to be erroneous. Under the de-
scribed conditions only (N-alkoxy-carbonyl)- and (N-aryloxy-car-
bonyl)-imidazoles (carbamates) were obtained.
isolated as single product the corresponding carbamate,
(2,6-dimethylphenoxy)-carbonyl-1-imidazole (B), in 91%
yield, as a colourless oil that solidified on standing, mp
63–64 °C (in a patent2 2,6-dimethylphenyl-1-imidazole,
structure A, prepared by an umambiguous method, is
mentioned with mp 82–82.5 °C; Lit.1 mentions 38 to be a
viscous oil).
Key words: alcohols, phenols, imidazoles, carbonyl diazole re-
agents, heterocycles
1
The H NMR-spectrum (300 MHz, CDCl3) of our com-
pound B was identical with the data published for A;1
however the elemental analysis clearly supported the
structure B: C12H12N2O2 (MW: 216.24) requires C, 66.65;
H, 5.59; N, 12.95; O, 14.80; found: C, 66.74; H, 5.73; N,
12.91; O, 14.83.
Introduction
Recently Njar has reported a high-yielding synthesis of
imidazoles and triazoles from alcohols and phenols.1 Ac-
cording to the procedures described N-alkyl- and N-
arylimidazoles were prepared directly from alcohols or
phenols with N,N’-carbonyldiimidazole (CDI) under very
mild conditions. Our interest in N-arylimidazoles as pre-
cursors for catalyst ligands prompted us to study these
striking and unexpected reactions.
The structure B was further supported by a strong IR-ab-
sorption (KBr) at 1769 cm–1 (C=O stretch vibration).
Additional chemical evidence for structure B was found
when the product was hydrolysed in ethanolic aqueous
NaOH solution on standing or brief heating to reflux:
1
Only 2,6-dimethylphenol, identified by H NMR, was
formed besides some imidazole.
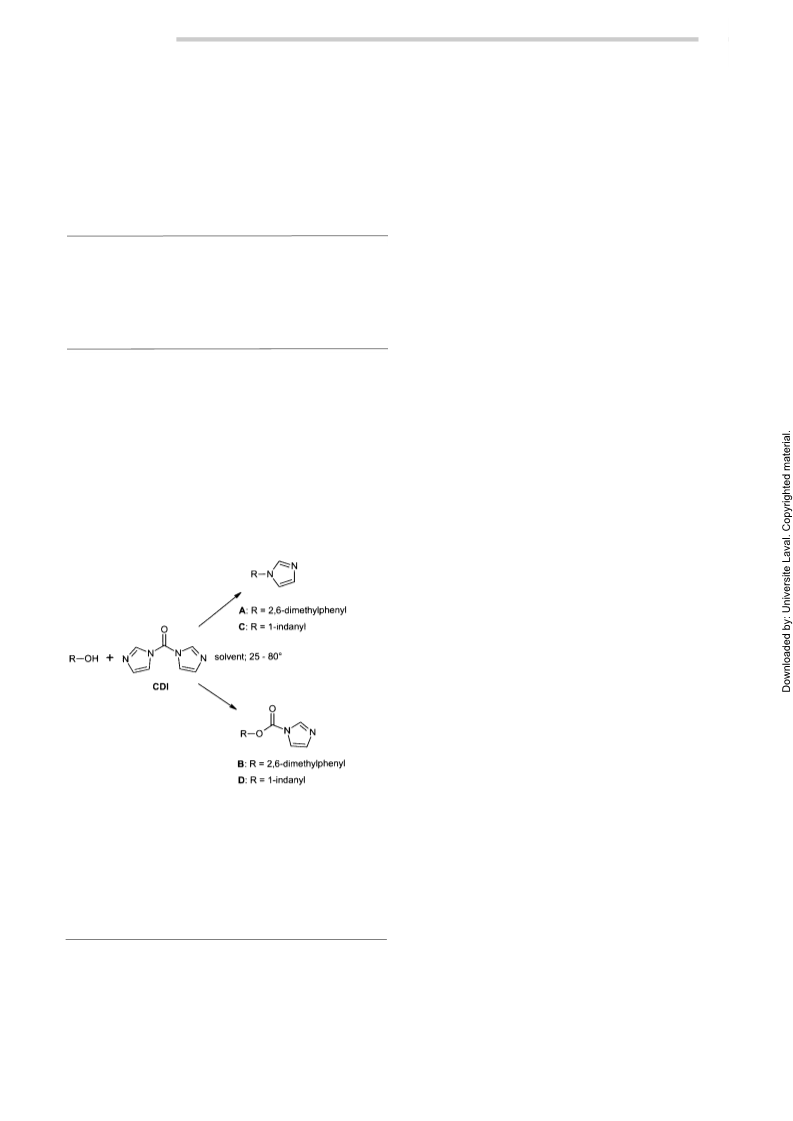
Reaction of CDI with 1-Indanol
We next repeated the reported synthesis of 1-(1H-imida-
zol-1-yl)indane (C) (Lit.1: Table, 25; Entry 17) by react-
ing 1-indanol with 1.3 equivalents of CDI in acetonitrile
at reflux for 1 hour. We isolated (1-indanyloxy)carbonyl-
1-imidazole (D) in 42% yield, mp 89–90 °C. Indene was
formed as the only identifiable side product. This instabil-
ity of D was also mentioned in the literaure.1
1
Again the H NMR-spectrum (300 MHz, CDCl3) was
identical with the published data for C;1 however the ele-
mental analysis clearly supported the structure D:
C13H12N2O2 (MW 228.25) requires C, 68.41; H, 5.30; N,
12.27; O, 14.02; found: C, 68.42, H, 5.43; N, 12.23; O,
13.81.
Scheme
Reaction of CDI with 2,6-Dimethylphenol
The structure D was further supported by a strong IR-ab-
sorption (KBr) at 1743 cm–1 (C=O stretch vibration) typi-
cal for a N-alkoxycarbonyl-imidazole.3
We repeated exactly the reported procedure of synthesis
of 2,6-dimethylphenyl-1-imidazole (A) (Lit.1: Table, 38,
Entry 31) by reacting 2,6-dimethylphenol with 1.3 equiv-
Additional chemical evidence for structure D was found
when the product was hydrolysed in ethanolic aqueous
NaOH solution on standing, only 1-indanol and indene,
identified by 1H NMR, were formed besides some imida-
zole.
Synthesis 2002, No. 1, 28 12 2001. Article Identifier:
1437-210X,E;2002,0,01,0029,0030,ftx,en;Z09901SS.pdf.
© Georg Thieme Verlag Stuttgart · New York
ISSN 0039-7881

 Fischer
Fischer