Please do not adjust margins
Catalysis Science & Technology
Page 10 of 12
DOI: 10.1039/C8CY01688G
ARTICLE
Journal Name
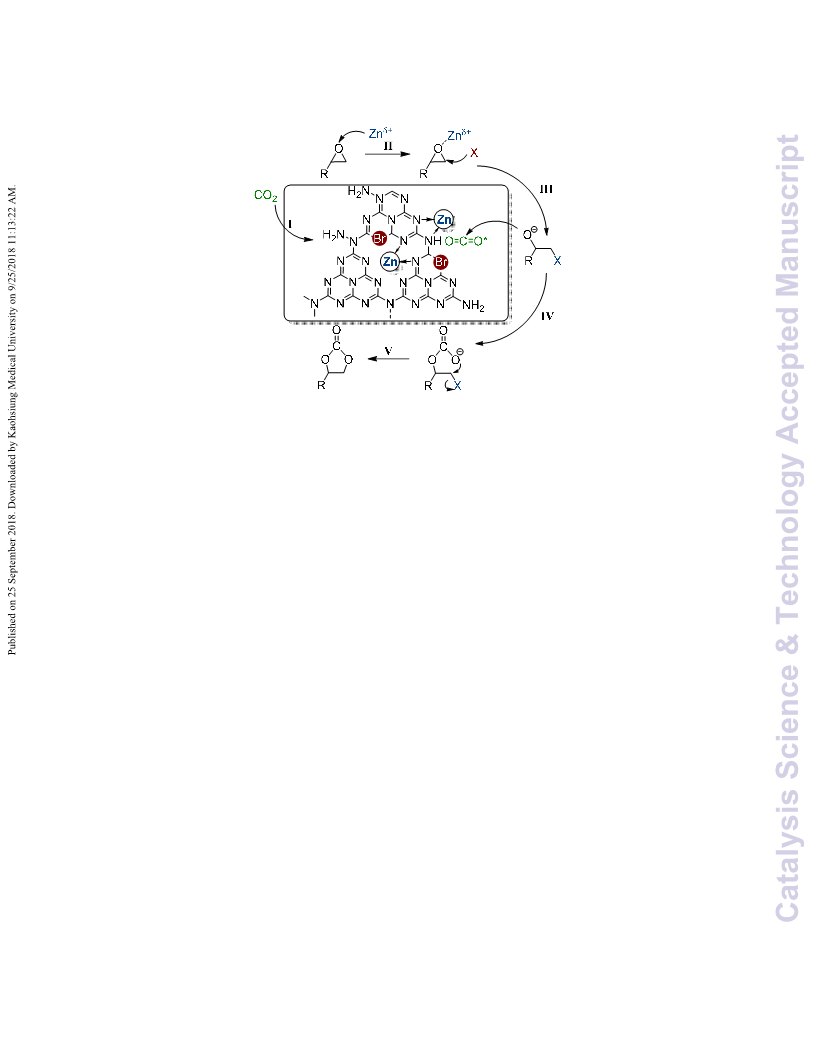
epoxides lies on both activation of CO2 and ringꢀopening of The work was financially supported by PetroChina Innovation
epoxides. Owing to its abundant basic sites in forms of amino Foundation (2018Dꢀ5007ꢀ0508), Advanced Catalysis and
group, CO2 molecules were initially adsorbed and then Green Manufacturing Collaborative Innovation Centre
activated by gꢀC3N4. In addition, the basic solvent of DMF also (ACGM2016ꢀ06ꢀ28), Foundation of State Key Laboratory of
contributes to the activation step. As for epoxide, zinc cations Highꢀefficiency Utilization of Coal and Green Chemical
coordinate with the oxygen of epoxide, leading to polarization Engineering (2017ꢀK28), Foundation of State Key Laboratory
of C‒O bonds thereof and meanwhile, the halide anions of Chemical Resource Engineering (CREꢀ2017ꢀCꢀ109), and the
nucleophilically attack the less bulky carbon atoms of epoxide. Topꢀnotch Academic Programs Project of Jiangsu Higher
Among the three halide anions (Cl−/Br−/I−), Cl− and I− own the Education Institutions (PPZY2015B145). J. Xu also thanks Junꢀ
highest nucleophilic and leaving abilities 21,64, respectively. In Jing Ding of Shiyanjia Lab for his help in XPS characterization.
comparison, Br− has more suitable capability in
nucleophilically attacking and leaving, reasonably explaining
Notes and references
its superior catalytic activity in the cycloaddition reaction to its
counterparts (Table 1). The dual steps initiated by ZnX2
accelerate the ringꢀopening of epoxide, yielding haloalkoxy
anion. Subsequently, the haloalkoxy intermediate attacks the
active CO2 molecule attached on the surface of gꢀC3N4 and thus
1. L. Wang, G. Zhang, K. Kodama and T. Hirose, Green Chem., 2016,
18, 1229-1233.
2. J. Roeser, K. Kailasam and A. Thomas, ChemSusChem, 2012, 5,
1793–1799.
3. M. North, R. Pasquale and C. Young, Green Chem., 2010, 12,
1514-1539.
4. J. Peng, H.-J. Yang, N. Song and C.-Y. Guo, J. CO2 Util., 2015, 9,
16-22.
5. D. Ma, H. Zheng, H.-M. Wan, Y. Chen, J. Xu and B. Xue, Micropor.
Mesopor. Mater., 2018, 258, 244-250.
6. D.-H. Lan, N. Fan, Y. Wang, X. Gao, P. Zhang, L. Chen, C.-T. Au and
S.-F. Yin, Chin. J. Catal., 2016, 37, 826-845.
7. R. A. Watile, K. M. Deshmukh, K. P. Dhake and B. M. Bhanage,
Catal. Sci. Technol., 2012, 2, 1051-1055.
8. J. N. Appaturi and F. Adam, Appl. Catal. B, 2013, 136-137, 150-
159.
9. L. Han, H. Li, S.-J. Choi, M.-S. Park, S.-M. Lee, Y.-J. Kim and D.-W.
Park, Appl. Catal. A, 2012, 429, 67-72.
10. J. K. Lee, Y. J. Kim, Y. Choi, H. Lee, J. S. Lee, J. Hong, E. Jeong, H. S.
Kim and M. Cheong, Appl. Catal. B, 2012, 111-112, 621-627.
11. Y. Xie, T. T. Wang, X. H. Liu, K. Zou and W. Q. Deng, Nat.
Commun., 2013, 4, 1960.
transforms into
a linear halocarbonate. Afterwards, the
halocarbonate closes its ring by the leaving of halogen anion,
and eventually turns into cyclic carbonate. At this juncture, it
can be further clarified that the activation processes of CO2 and
epoxide are primarily achieved by gꢀC3N4 and ZnX2. However,
it should be stressed that, far more than a solid base, gꢀC3N4
acts a significant role in loading the catalytic active species of
ZnX2. As illuminated above, through the formation of Zn‒N
complex together with the insertion of halogen into gꢀC3N4
matrix, the gꢀC3N4 support steadily anchors and immobilizes
ZnX2 species on its surface, and thus restrains the loss of ZnX2
into liquid phase, consequently realizing heterogeneous
catalysts.
5. Conclusion
In summary, a series of ZnX2/gꢀC3N4 composites have been
fabricated by a simple preparation approach. The temperature
of heating treatment has a significant effect on the bonding
states of Zn and halogen species. Under optimized conditions,
zinc can be coordinated with nitrogen atoms of gꢀC3N4 while
halogen substitutes the nitrogen atoms in the framework of gꢀ
C3N4. Among various catalysts, ZnBr2/gꢀC3N4 demonstrates the
highest activity in cycloaddition of CO2 with PO. The catalysts
could be reused for at least five times and show wide
applicability for other cycloaddition reactions with various
epoxides. Zinc halide are confirmed as a key component in
activating epoxide while gꢀC3N4 not only serves as a solid base
to activate CO2 but also plays a vital role in anchoring and
immobilizing zinc halide on the surface of gꢀC3N4. We think
that the findings could provide a comprehensive insight of the
support effect of gꢀC3N4 for zinc halides and other wide metal
halides, and constitute a new strategy for designing more novel
heterogeneous metalꢀhalideꢀbased catalysts.
12. J. Peng, H.-J. Yang, S. Wang, B. Ban, Z. Wei, B. Lei and C.-Y. Guo,
J. CO2 Util., 2018, 24, 1-9.
13. J. He, T. Wu, Z. Zhang, K. Ding, B. Han, Y. Xie, T. Jiang and Z. Liu,
Chem. -Eur. J., 2007, 13, 6992-6997.
14. J. Langanke, L. Greiner and W. Leitner, Green Chem., 2013, 15,
1173-1182.
15. W.-L. Dai, S.-L. Luo, S.-F. Yin and C.-T. Au, Appl. Catal. A, 2009,
366, 2-12.
16. J. Sun, L. Wang, S. Zhang, Z. Li, X. Zhang, W. Dai and R. Mori, J.
Mol. Catal. A, 2006, 256, 295-300.
17. Z. Huang, F. Li, B. Chen, T. Lu, Y. Yuan and G. Yuan, Appl. Catal. B,
2013, 136, 269-277.
18. J. Zhu, T. Diao, W. Wang, X. Xu, X. Sun, S. A. C. Carabineiro and Z.
Zhao, Appl. Catal. B, 2017, 219, 92-100.
19. R. R. Kuruppathparambil, T. Jose, R. Babu, G. Hwang, A. C.
Kathalikkattil, D. Kim and D. Park, Appl. Catal. B, 2016, 182, 562-
569.
20. L. Wang, K. Kodama and T. Hirose, Catal. Sci. Technol., 2016, 6,
3872-3877.
21. M. Liu, B. Liu, L. Shi, F. Wang, L. Liang and J. Sun, RSC Adv., 2015,
5, 960-966.
22. M. Liu, K. Gao, L. Liang, J. Sun, L. Sheng and M. Arai, Catal. Sci.
Technol., 2016, 6, 6406-6416.
Conflicts of interest
There are no conflicts to declare.
23. S.-S. Wu, X.-W. Zhang, W.-L. Dai, S.-F. Yin, W.-S. Li, Y.-Q. Ren and
C.-T. Au, Appl. Catal. A, 2008, 341, 106-111.
24. Y. Wang, X. Wang and M. Antonietti, Angew. Chem. Int. Ed.,
Acknowledgements
10 | J. Name., 2012, 00, 1-3
This journal is © The Royal Society of Chemistry 20xx
Please do not adjust margins

 Xu, Jie
Xu, Jie
 Gan, Yu-Lin
Gan, Yu-Lin
 Hu, Peng
Hu, Peng
 Zheng, Huan
Zheng, Huan
 Xue, Bing
Xue, Bing