R. Burgada et al. / Tetrahedron Letters 48 (2007) 2315–2319
2319
3
1
1
2
132–2139; (b) Neuman, A.; Safsaf, A.; Gillier, H.;
Leroux, Y.; El Manouni, D. Phosphorus Sulfur Silicon
992, 70, 273–285; (c) Silvestre, J-P.; Qui Dao, N.; Leroux,
(ester): P NMR: 23.7 (CDCl
t, JHH 7 (36H); 2.13–2.45 m (9H); 2.57 t, JHH 7.03 (6H);
2.92 d, J 6.25 (6H); 4.18 p, JHH 7.03 (24H) (CDCl3).
3
). H NMR: 0.8 s (3H); 1.34
1
HH
1
3
Y. Heteroatom Chem. 2001, 12, 73–89; (d) Jurisson, S. S.;
Benedict, J.-J.; Elder, R. C.; Whittle, I. R.; Deutsch, E.
Inorg. Chem. 1983, 22, 1332–1338; (e) Song, H. H.; Yin,
P.; Zheng, L. M.; Korp, J. D.; Jacobson, A. J.; Gao, S.;
Xin, X. Q. J. Chem. Soc., Dalton Trans. 2002, 2752–
C NMR: 16.3; 19; 21.7; 34.7; 35.7 t, JCP 133; 42.5; 50.3;
31
3
62.9; 172.9. (CDCl ). Compound 2d (acid): P NMR: 22.4
1
3
1
(D
2 2
O, pH 1). P NMR: 20.1 (D O, pH 14) H NMR: 0.68
s (3H); 2.02–2.29 m (9H); 2.45 s broad (6H); 2.91 s (6H)
1
(D
2
O, pH 1) H NMR: 0.54 s (3H); 1.28 tt, JHCP 22.27
2
759.
. (a) Hancock, R. D.; Martell, A. E. Chem. Rev. 1989, 89,
875–1914; (b) Martell, A. E.; Hancock, R. D.; Moteka-
J
HH 5.24 (3H); 1.6–1.8 m (6H); 2.1–2.29 m (6H); 2.79 s
1
3
8
9
(6H) (D O, pH 14). C NMR: 20.7; 24; 37.4; 39.3 t, JCP
2
1
127.7; 42.8; 45.6; 178. (D O, pH 1). Compound 2e (ester):
2
31 1
itis, R. J. Coord. Chem. Rev. 1994, 133, 39–65.
P NMR: 23.8 (CDCl
7.06 J 3.07 (36H); 2.74 dt, JHP 16.24 JHH 6.14 (6H); 3.1 d,
J 6.4 (6H); 3.25 tt, JHH 6.14 JHP 23.77 (3H); 4–4.19 m
3 1
). H NMR: 0.8 s (3H); 1.3 dt, J
. (a) Clark, S.; Elliott, J. M.; Chipperfield, J. R.; Styring, P.;
Sinn, E. Inorg. Chem. Commun. 2002, 5, 249–251; (b)
Franczyk, T. S.; Czerwinski, X. R.; Raymond, K. N. J.
Am. Chem. Soc. 1992, 114, 8138–8146, and References
cited there in; (c) Graziani, R.; Bombieri, G.; Forsellini, E.
J. Chem. Soc., Dalton 1972, 2059–2061; (d) Kannan, S.;
Rajalakshmi, N.; Chetty, K. V.; Venugopal, V.; Drew, M.
G. B. Polyhedron 2004, 23, 1527–1533; (e) Masci, B.;
Thu e´ ry, P. Polyhedron 2005, 24, 229–237; (f) Templeton,
D. H.; Zalkin, A.; Ruben, H.; Templeton, L. K. Acta
Crystallogr., Sect. C 1985, 1439–1441; (g) ValIet, V.; Moll,
H.; Wahlgren, U.; Szabo, Z.; Grenthe, I. Inorg. Chem.
2
1
3
(24H); 7.5 t, J 6.14 (3H) (CDCl
32; 32.3 t, JCP 135; 37; 41; 44.2; 62.9 dd, J
3
). C NMR: 16.5; 20.3;
39 J 5.8; 170.8.
). Compound 2e (acid): P NMR: 21 (D O) pH 1;
P NMR: 19.56 (D O) pH 14. H NMR: 0.87 s (3H); 2.7–
1
2
3
1
(CDCl
3
2
3
1
1
2
1
2
2.9 m (9H); 3.13 s (6H) (D O) pH 1. H NMR: 0.89 s (3H);
2.11 tt, JHH 5.7 JHP 21.6 (3H); 2.70–2.77 m (6H); 3.12 s
(6H) (D O) pH 14.
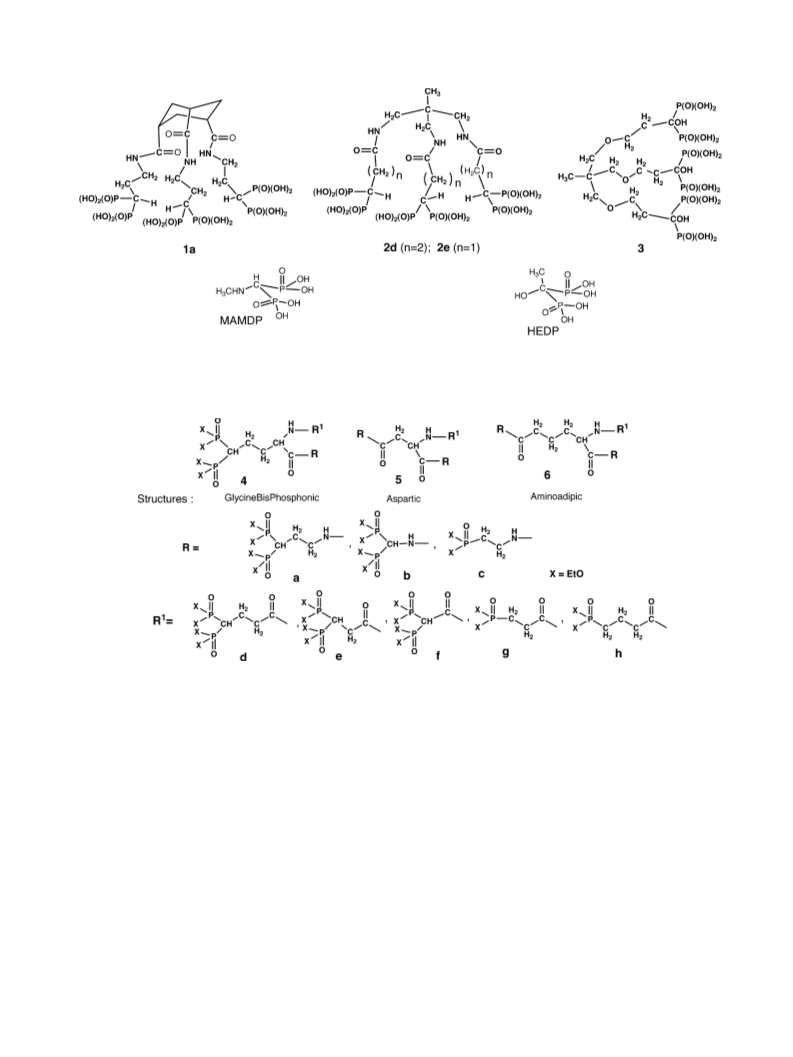
Ligands obtained from amino acid derivatives as supports:
Compound 4ad (ester): P NMR: 23.74; 23.79; 23.97
(CDCl
2
3
1
1
3
) H NMR: 1.33 t, J 7.3 (36H); 2–2.52 m (9H); 2.7–
2
003, 42, 1982–1993; (h) V aˆ zquez, J.; Bo, C.; Poblet, J. M.;
de Pablo, J.; Bruno, J. lnorg. Chem. 2003, 42, 6136–6141;
i) Zhang, Y.; Livens, F. R.; Collison, D.; Helliwell, M.;
2.9 m (2H); 3.2–3.4 m (2H); 4.1 m (24H); 4.6 m (1H)
3
1
(CDCl
(CDCl
3
). Compound 5ad (ester): P NMR: 23.7; 24.
1
(
3
). H NMR: 1.3 t, J 7 (36H); 2–2.1 and 2.1–2.37 m
Heatley, F.; Powell, A. K.; Wocadlo, S.; Eccles, H.
Polyhedron 2002, 21, 69–79.
(6H); 2.42 tt, JHH 5.5; JHP 24.7 (3H); 2.55 q, J 7.3 (4H); 2.6
d, J 9.16 (HMPT); 2.7 m (2H); 3.38 m (4H); 4.13 m (24H)
(CDCl ). C NMR: 16.3; 21; 25; 33.9; 34.9; 35.4; 36.7
3
1
3
1
0. (a) Matsumoto, K.; Ozawa, T.; Jitsukawa, K.; Einaga, H.;
Masuda, H. Inorg. Chem. 2001, 40, 190–191; (b) Matsu-
moto, K.; Suzuki, N.; Ozawa, T.; Jitsukawa, K.; Masuda,
H. Eur. J. Inorg. Chem. 2001, 2481–2484.
1. Budimir, N.; Fournier, F.; Bailly, T.; Burgada, R.; Tabet,
J.-C. Rapid Commun. Mass Spectrom. 2005, 19, 1822–
(HMPT); 37.7; 38.5; 48.9; 50; 62.9; 171; 172; 172.5.
(CDCl ) dept: 34.8 t, J 133.6; 35 t, J 133.6; 35.2 t, 133.6;
3
3
1
35.9 t, J 131.14; 36 t, J 131.5. Compound 5ad (acid)
NMR: 24.4 (D O, pH 1). Compound 5a (ester): P NMR:
23.9 (CDCl
P
3
1
1
1
1
2
1
3
). H NMR: 1.34 t, J 7.03 (24H); 2.08 s (3H);
1
828.
2.16–2.22 m (4H); 2.39–2.88 m (2+2H); 3.46 m (4H); 4.2 p
1
3
2. Sawicki, M.; Siaugue, J.-M.; Jacopin, C.; Moulin, C.;
Bailly, T.; Burgada, R.; Meunier, S.; Baret, P.; Pierre,
J.-L.; Taran, F. Chem. Eur. J. 2005, 11, 3689–3697.
J 6.64 (16H); (CDCl
3
). C NMR: 16.3; 24.9; 31.5 t J 116;
). Com-
33.8; 37.4; 38.5; 50.2; 62.8; 170.4; 171.2; (CDCl
3
3
1
1
pound 5a (acid): P NMR: 22 (D
1.87 s (3H); 1.9–2 m (4H); 2.15 tt, JHP 23.44, JHH 7 (2H);
2.42–2.64 m (3H); 3.26 m (4H); 4.47 m (1H) (D O, pH 1).
O, pH 14): 1.64 tt, JHP 21.5 JHH 7.4 (2H);
2
O, pH 1). H NMR:
3. Castano, A. M.; Echavarren, A. M. Tetrahedron 1992, 48,
3
377–3384.
2
1
1
1
4. Sturtz, G.; Guervenou, J. Synthesis 1991, 661–662.
5. P; H; C NMR: (Varian Inova 500: H 500.6;
H NMR: (D
2
3
1
1
13
1
31
31
P
P
1.7–1.83 m (4H); 1.9 s (3H); 2.41–2.76 m (3H); 3.2 m (4H);
1
3
1
13
2
8
00.7;1 C 125.9 MHz and Varian Gemini 200: H 200;
0.9; C 50.3 MHz).
4.5 m (1H). C NMR: 25.5; 27.8; 39.7 t, J 113; 40; 42.5;
3
31
53.6; 190.9; 208.7; (D O, pH 14). Compound 8: P NMR:
23.4 (D
2
1
Ligands obtained from triacid or polyamine carriers:
Compound 1a (ester): P NMR: 23.6 (CDCl3).
NMR: 1.27 t, J 6.95 (36H); 1.57 m (3H); 2.04 m (9H);
2
O). H NMR: 1.31 t, J 7 (12H); 2.25 m (3H); 2.52
3
1
1
H
m (3H); 2.94 m (2H); 4.15 m (8H); 4.6 m (1H) (D O).
2
3
1
1
Compound 9: P NMR: 34.6 (D O). H NMR: 1.3 t, J
2
2
.2 m (3H); 2.97 tt, JHP 24, JHH 5.85 (3H); 3.3 m (6H); 4.09
7.03 (6H); 2.09–2.25 m (2H); 2.49–2.68 m (2H); 2.88 m
1
3
0
*
m (24H) (CDCl
3
). C NMR: 16.4; 25.2; 31.9 ; 34.6 t,
(2H); 4.1 p, J 7.4 (4H); 4.6 m (1H) (D
2
O). Compound 9 :
**
*
**
31
1
1
33.6; 38.7; 44 ; 62.9; 174.5 (CDCl ) CH cycloh.; CH
P NMR: 32.7 (CDCl ). H NMR: 1.3 t, J 7.3 (6H); 1.78–
3
2
3
3
1
13
Cycloh. Compound 1a (acid): P NMR 21.7 (D
NMR: 38.2 t, J 128 (P–CH–P) (D
2
O)
C
1.99 m (4H); 2.6 m (2H); 2.9–3 m (2H); 4 p, J 7.3 (4H); 4.8
m (1H) (CDCl ).
2
O). Compound 2d
3

 Burgada, Ramon
Burgada, Ramon
 Bailly, Théodorine
Bailly, Théodorine
 Prangé, Thierry
Prangé, Thierry
 Lecouvey, Marc
Lecouvey, Marc