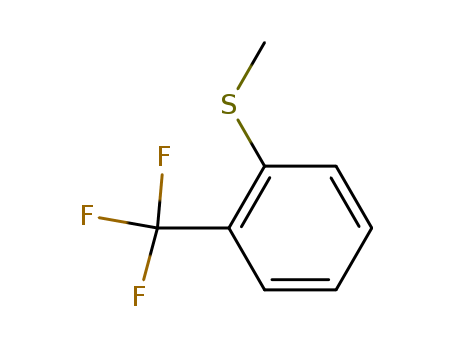
- Chemical Name:2-Trifluoromethylthioanisole
- CAS No.:322-58-7
- Molecular Formula:C8H7F3S
- Molecular Weight:192.205
- Hs Code.:
- DSSTox Substance ID:DTXSID60375347
- Nikkaji Number:J2.928.796A
- Wikidata:Q82163875
- Mol file:322-58-7.mol
Synonyms:2-Trifluoromethylthioanisole;322-58-7;Methyl(2-(trifluoromethyl)phenyl)sulfane;1-(METHYLSULFANYL)-2-(TRIFLUOROMETHYL)BENZENE;1-methylsulfanyl-2-(trifluoromethyl)benzene;1-methylsulfanyl-2-trifluoromethyl-benzene;SCHEMBL514678;DTXSID60375347;MFCD01882714;AKOS006277705;2-(Trifluoromethyl)thiophenol, S-methyl-;AS-84230;1-(Methylthio)-2-(trifluoromethyl)benzene;CS-0102147;D76277