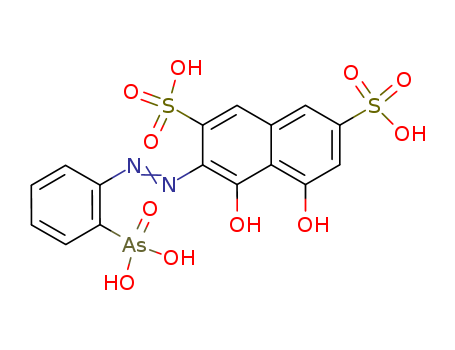
Chemical Property of Arsenazo I
Edit
Chemical Property:
- Melting Point:≥300 °C(lit.)
- Boiling Point:°Cat760mmHg
- PKA:-0.71±0.40(Predicted)
- Flash Point:°C
- PSA:256.70000
- Density:g/cm3
- LogP:2.63540
- Solubility.:methanol: 1 mg/mL, clear, red
- Hydrogen Bond Donor Count:6
- Hydrogen Bond Acceptor Count:13
- Rotatable Bond Count:5
- Exact Mass:547.917671
- Heavy Atom Count:32
- Complexity:971
- Purity/Quality:
-
99%, *data from raw suppliers
ARSENAZO (I) 95.00% *data from reagent suppliers
Safty Information:
- Pictogram(s):
 T
T N
N
- Hazard Codes:T,N
- Statements:
23/25-50/53
- Safety Statements:
20/21-28-45-60-61
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Dyes -> Azo Dyes
- Canonical SMILES:C1=CC=C(C(=C1)N=NC2=C(C3=C(C=C(C=C3C=C2S(=O)(=O)O)S(=O)(=O)O)O)O)[As](=O)(O)O
-
Uses
The reagent reacts with many metal ions, such as uranium(IV), thorium(IV),
zirconium(IV), scandium(III), lanthanum(III), cerium(III), aluminium(III), beryllium(
III), titanium(III), niobium(lll), tantalum(III), vanadium(IV), tin(IV), bismuth(
III), gallium(III), copper(III), palladium(II), magnesium(II) and calcium(II)
to give coloured complexes. It has been used for the photometric determination
of the majority of the above-listed metal ions. Though the reagent reacts
with such a variety of metal ions, the complex formation reaction may be made
selective for individual metal ions by appropriate choice of the reaction conditions,
primarily of the pH of the solution. For instance, thorium can be determined
with Arsenazo I in the presence of almost all rare earth metals, and
similarly zirconium and thorium in the presence of uranium, etc.
The stabilities of the metal complexes of Arsenazo I are lower than those of the
corresponding complexes of EDTA. It can also therefore be used advantageously
as indicator in complexometric determination of some metals (e.g. plutonium,
thorium).
The analytical sensitivity of the formation of Arsenazo I complexes is comparatively
high (0.05-0.1 γ/ml). However, the importance of the reagent is decreasing
in analytical practice, in parallel with an increase in significance of the
related reagent, Arsenazo III, which gives more stable complexes with higher
absorbances, and which is therefore of greater analytical sensitivity.