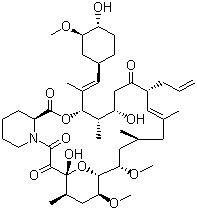
- Chemical Name:1,14-Dihydroxy-12-[1-(4-hydroxy-3-methoxycyclohexyl)prop-1-en-2-yl]-23,25-dimethoxy-13,19,21,27-tetramethyl-17-prop-2-enyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetrone
- CAS No.:104987-11-3
- Molecular Formula:C44H69NO12
- Molecular Weight:804.031
- Hs Code.:29349990
- European Community (EC) Number:627-021-3,658-056-2
- Wikipedia:Tacrolimus
- Wikidata:Q105222713
- Mol file:104987-11-3.mol
Synonyms:104987-11-3;1,14-dihydroxy-12-[1-(4-hydroxy-3-methoxycyclohexyl)prop-1-en-2-yl]-23,25-dimethoxy-13,19,21,27-tetramethyl-17-prop-2-enyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetrone;144490-63-1;MFCD00869853;129212-35-7;QJJXYPPXXYFBGM-UHFFFAOYSA-N;SY069910;FT-0626431;17-allyl- 1,14-dihydroxy-12-[2'-(4"-hydroxy-3"-methoxycyclohexyl)1-methylvinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9 ]octacos-18-ene-2,3,10,16-tetraone;17-allyl-1,14-di-hydroxy-12-[2-(4-hydroxy-3-methoxy-cyclohexyl)-1-methyl-vinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-aza-tricyclo[22.3.1.04,9] octacos-18-ene-2,3,10,16-tetraone;17-Allyl-1,14-di-hydroxy-12-[2-(4-hydroxy-3-methoxy-cyclohexyl)-1-methyl-vinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-aza-tricyclo[22.3.1.04,9]octacos-18-ene-2,3,10,16-tetraone;17-allyl-1,14-dihydroxy-12-[2'-(4"-hydroxy-3"-methoxy -cyclohexyl)-1'-methylvinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-azatricyclo-[22.3.1.04,9 ]octacos-18-ene-2,3,10,16-tetraone;17-allyl-1,14-dihydroxy-12-[2'-(4"-hydroxy-3"-methoxycyclohexyl)-1'-methylvinyl]-23,25-dimethoxy-13,19,21,-27-tetramethyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9 ]octacos-18-ene-2,3,10,16-tetraone;17-allyl-1,14-dihydroxy-12-[2'-(4"-hydroxy-3"-methoxycyclohexyl)-1'-methylvinyl]-23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9 ]octacos-18-ene-2,3,10,16-tetraone;17-allyl-1,14-dihydroxy-12-[2'-(4"-hydroxy-3"-methoxycyclohexyl)-1'-methylvinyl]23,25-dimethoxy-13,19,21,27-tetramethyl-11,28-dioxa-4-azatricyclo[22.3.1.04,9 ]-octacos-18-ene-2,3,10,16-tetraone


 T,
T, Xi
Xi

