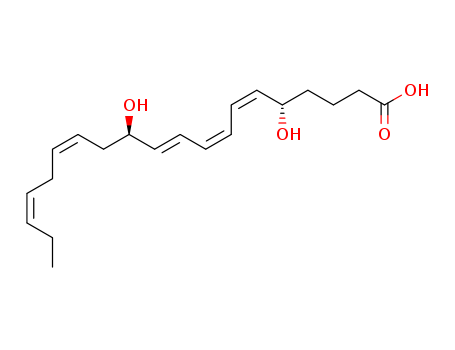
Multi-step reaction with 13 steps
1: triphenyl phosphite, I2, imidazol / diethyl ether; acetonitrile / 0.67 h / 25 °C
2: Cu(I)
3: 90 percent / tetrahydrofuran / 12 h / 65 °C
4: 91 percent / LiAlH4 / diethyl ether / 5 h / 25 °C
5: 76 percent / dimethyl(S,S)(-)-tartarate
6: 90 percent / H2 / Lindlar / tetrahydrofuran / 760 Torr
7: 84 percent / O2 / oxidation with Collins reagent
8: 94 percent / tetrahydrofuran / -78 deg C, 10 min,; and then from -78 deg Cto 0 deg C over 1 h.
9: 84 percent / acetic acid; acetonitrile; H2O / 6 h / 25 °C
10: triphenylphosphine, imidazole, I2, diisopropylamine / diethyl ether; acetonitrile / 0.67 h / 25 °C
11: acetonitrile / 16 h / 60 °C
13: 1.) potassium isopropoxide; 2.) base / 1.) 0 deg C, 1 h; 2.) hydrolysis
With
1H-imidazole; triphenyl phosphite; lithium aluminium tetrahydride; Dimethyl D-tartrate; hydrogen; iodine; oxygen; potassium isopropoxide; diisopropylamine; triphenylphosphine;
Lindlar's catalyst; copper;
In
tetrahydrofuran; diethyl ether; water; acetic acid; acetonitrile;
DOI:10.1016/S0040-4039(01)99801-6



 F,
F, Xi
Xi

