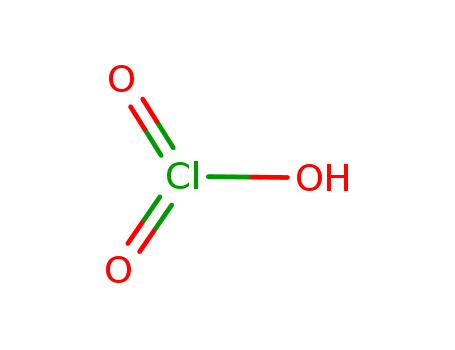
Chemical Property of Chloric acid
Edit
Chemical Property:
- Melting Point:<-20℃ [CRC10]
- Boiling Point:>100oC
- PSA:54.37000
- Density:1.2 g/mL at 25oC
- LogP:0.27510
- Water Solubility.:very soluble H2O [CRC10]
- XLogP3:2.1
- Hydrogen Bond Donor Count:1
- Hydrogen Bond Acceptor Count:3
- Rotatable Bond Count:0
- Exact Mass:83.9614216
- Heavy Atom Count:4
- Complexity:46.2
- Transport DOT Label:Oxidizer
- Purity/Quality:
-
99% *data from raw suppliers
Safty Information:
- Pictogram(s):
Toxic by ingestion and inhalation. Strong oxidizer, ignites organic materials on contact.
- Hazard Codes:O,C
- Statements:
8-34
- Safety Statements:
17-26-36/37/39-45
- MSDS Files:
-
SDS file from LookChem
Useful:
- Chemical Classes:Toxic Gases & Vapors -> Acids, Inorganic
- Canonical SMILES:OCl(=O)=O
-
Description
Chloric acid, HClO3, is an oxoacid of chloride, and
the formal precursor of chlorate salts. It is a strong
acid (pKa=-1) and a strong oxidizing agent. It is stable
in cold aqueous solution up to a concentration of
approximately 30%, and solution of up to 40% can be
prepared by careful evaporation under reduced pressure.
Above these concentrations, and on warming,
chloric acid solutions decompose to give a variety of
products, for example:
8HClO3→4HClO4+2H2O+2Cl2+3O2
Note that it disproportionates into perchloric acid
in which the chlorine atom has a+7 oxidation state
and also the zero oxidation state of chlorine gas (an
oxidation–reduction reaction). Thus, under the proper
conditions, it can also be used to make perchloric acid.
3HClO3→HClO4+H2O+2ClO2
-
Uses
Oxidizing agent; with H2SO3 as catalyst in acrylonitrile polymerization.