Products Categories
| CAS No.: | 14708-14-6 |
|---|---|
| Name: | NICKEL TETRAFLUOROBORATE |
| Molecular Structure: | |
|
|
|
| Formula: | B2F8Ni |
| Molecular Weight: | 232.30 |
| Synonyms: | Borate(1-),tetrafluoro-, nickel(2+) (8CI);Nickel fluoborate (Ni(BF4)2) (6CI);Nickeltetrafluoroborate (7CI);Nickel bis(tetrafluoroborate);Nickel borofluoride;Nickel fluoroborate;Nickel tetrafluoroborate (Ni(BF4)2);Nickel(II)tetrafluoroborate;Nickelous tetrafluoroborate; |
| EINECS: | 238-753-4 |
| Density: | 1.47 |
| Solubility: | water soluble |
| Appearance: | Green odorless aqueous solution |
| Hazard Symbols: |
 T, T,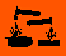 C C
|
| Risk Codes: | 45-34-42/43-31 |
| Safety: | 53-45-36/37/39-26-23 |
| PSA: | 0.00000 |
| LogP: | 2.60000 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride
- Total:33 Page 1 of 1 1

What can I do for you?
Get Best Price
Chemistry
Chemistry informtion about Nickel(II) Fluoborate (CAS NO.14708-14-6) is:
IUPAC Name: Nickel(2+) Ditetrafluoroborate
Synonyms: Borate(1-),Tetrafluoro-,Nickel(2+)(2:1) ; Nickel(Ii)Fluoborate ; Nickelborofluoride ; Nickelfluoroborate ; Nickeloustetrafluoroborate ; Nickeltetrafluoroborate(Solution40%) ; Tetrafluoro-Borate(1-Nickel(2+)(2:1) ; Tetrafluoro-Borate(1-Nickel(2++)(2:1)
MF: B2F8Ni
MW: 232.3
EINECS: 238-753-4
Density: 1.47
Following is the molecular structure of Nickel(II) Fluoborate (CAS NO.14708-14-6) is:

Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LCLo | inhalation | 530mg/m3/10M (530mg/m3) | National Defense Research Committee, Office of Scientific Research and Development, Progress Report.Vol. No.9-4-1-19, Pg. 1943, | |
| rat | LDLo | oral | 500mg/kg (500mg/kg) | National Academy of Sciences, National Research Council, Chemical-Biological Coordination Center, Review. Vol. 5, Pg. 28, 1953. |
Consensus Reports
NTP 10th Report on Carcinogens. Nickel and its compounds are on the Community Right-To-Know List. Reported in EPA TSCA Inventory.
Safety Profile
Confirmed carcinogen. Moderately toxic by ingestion and inhalation. When heated to decomposition it emits very toxic fumes of F−. See also FLUORIDES, BORON, and NICKEL COMPOUNDS.
Hazard Codes:
 T
T
 C
C
Risk Statements:
R45:May cause cancer.
R34:Causes burns.
R42/43:May cause sensitization by inhalation and skin contact.
R31 :Contact with acids liberates toxic gas.
Safety Statements:
S53:Avoid exposure - obtain special instructions before use.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S23:Do not breathe vapour.
RIDADR: 1759
RTECS: QR6650000
Hazard Note: Corrosive
HazardClass: 6.1(b)
PackingGroup: III
Standards and Recommendations
OSHA PEL: TWA 0.1 mg (Ni)/m3; TWA 2.5 mg(F)/m3; BEI: 3 mg/g creatinine of fluorides in urine prior to shift; 10 mg/g creatinine of fluorides in urine at end of shift.
ACGIH TLV: TWA 0.2 mg(Ni)/m3; Human Carcinogen
NIOSH REL: (Inorganic Nickel) TWA 0.015 mg(Ni)/m3
Specification
General description about Nickel(II) Fluoborate (CAS NO.14708-14-6) ,it is a green odorless aqueous solution. It can sinks in and mixes with water. Acidic inorganic salts, such as Nickel Tetrafluoroborate , are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions. Ingestion causes irritation of mouth and stomach. Liquid irritates eyes and skin and may cause dermatitis.
