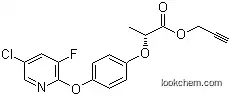
105512-06-9 Usage
Description
Clodinafop-propargyl is a foliar-absorbed aryloxyphenoxypropionate herbicide, belonging to the Oxyphenoxy acid ester chemical class. It is a carboxylic ester resulting from the formal condensation of the carboxy group of clodinafop with the hydroxy group of prop-2-yn-1-ol. This herbicide has the capacity to inhibit the lipid biosynthesizing enzyme, acetyl coenzyme-A-carboxylase, which interferes with the production of fatty acids required for plant growth. Pure Clodinafop-propargyl is a colorless crystal with specific chemical properties, such as vapor pressure, solubility in various solvents, and a distribution coefficient.
Uses
1. Used in Agriculture:
Clodinafop-propargyl is used as a systemic herbicide for the control of annual grass weeds in cereal crops. It is particularly effective against post-emergent weeds such as selected grasses and does not act on broad-leaved weeds. The herbicide is applied to the foliar parts of the weeds, where it is absorbed through the leaves and translocated to the meristematic growing points of the plant.
2. Used in Wheat Crop Protection:
Clodinafop-propargyl is used as a herbicide for weed control in wheat, providing moderate control of Italian rye-grass and controlling grass weeds such as wild oats, rough meadow-grass, green foxtail, barnyard grass, Persian darnel, and volunteer canary seed.
3. Used in Crop Compatibility:
Clodinafop-propargyl is suitable for use on various crops, including all varieties of wheat, autumn-sown spring wheat, rye, triticale, and durum wheat.
4. Used in Chemical Synthesis:
Clodinafop-propargyl can inhibit the synthesis of ester-like biology, making it useful for controlling various types of weeds, such as amur foxtail, oat grass, avena sterilis, and green bristlegrass.
Mode of action
Clodinafop-propargyl is to inhibit the activity of acetyl-CoA carboxylase in plants. It is a systemic conductive herbicide, absorbed by the leaves and sheaths of plants, transmitted by phloem, and accumulated in meristems of plants. In this case, acetyl-CoA carboxylase is inhibited, and fatty acid synthesis is stopped. So cell growth and division cannot proceed normally, and lipid-containing structures such as membrane systems are destroyed, leading to plant death.
Toxicity evaluation
a) Mammalian toxicity : WHO Classification : Class III slightly hazardous”b) Environmental toxicity : It is non-toxic to fish, birds and bees.Shelf life :Two years under normal storage conditions (Tech grade and Formulation)
Check Digit Verification of cas no
The CAS Registry Mumber 105512-06-9 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,0,5,5,1 and 2 respectively; the second part has 2 digits, 0 and 6 respectively.
Calculate Digit Verification of CAS Registry Number 105512-06:
(8*1)+(7*0)+(6*5)+(5*5)+(4*1)+(3*2)+(2*0)+(1*6)=79
79 % 10 = 9
So 105512-06-9 is a valid CAS Registry Number.
InChI:InChI=1/C17H13ClFNO4/c1-3-8-22-17(21)11(2)23-13-4-6-14(7-5-13)24-16-15(19)9-12(18)10-20-16/h1,4-7,9-11H,8H2,2H3/t11-/m1/s1
105512-06-9Relevant articles and documents
Synthesis method of clodinafop-propargyl
-
Paragraph 0015-0021, (2021/01/29)
The invention relates to the field of chemical engineering, and discloses a synthesis method of clodinafop-propargyl. The method comprises the following steps of: dissolving DHPPA in an organic solvent, heating to a temperature of 50 DEG C, adding an anti
Preparation method of clodinafop propargyl
-
Paragraph 0038; 0044; 0045; 0046; 0047, (2016/10/17)
The invention relates to a preparation method of clodinafop propargyl used for controlling grassy weeds in wheat fields. R-2-(p-hydroxyphenoxy)propionic acid is taken as a raw material and has a reaction with caustic alkali in water and an aprotic polar s