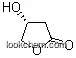58081-05-3Relevant articles and documents
Inversion of configuration of (S)-β-hydroxy-γ-butyrolactone with total retention of the enantiomeric purity
De Angelis, Francesco,De Fusco, Enrico,Desiderio, Paola,Giannessi, Fabio,Piccirilli, Fabrizio,Tinti, Maria Ornella
, p. 2705 - 2707 (1999)
In this paper we report the inversion of configuration of (S)β-hydroxy- γ-butyrolactone [(S)-1] to its (R) enantiomer (R)-1, with total retention of the enantiomeric purity, by a four-step procedure. The (R)-β-hydroxy-γ- butyrolactone [(R)-1] was thus synthetized with an overall chemical yield of 47% and > 97% ee. This transformation opens an economic route to the production of (R)-GABOB and (R)-carnitine, among other biologically active compounds, from a D-hexose source, or, alternatively, from the industrial waste compound (S)carnitine. During the reaction sequence, the intermediate β-lactone 4 is also prepared, which is now under investigation as a chiral synthon for new synthetic applications.
A Concise Stereoselective Total Synthesis of Methoxyl Citreochlorols and Their Structural Revisions
Sunnapu, Ranganayakulu,Rajendar, Goreti
, p. 1637 - 1642 (2021/03/15)
A concise, stereoselective and protecting group free approaches for the total synthesis of (?)-(2S,4R)- and (+)-(2R,4S)-3′-methoxyl citreochlorols and their stereoisomers are demonstrated. All four stereoisomers were synthesized to establish the absolute stereochemistry of the reported structures and the structures were revised accordingly. The approach involves chelation controlled regioselective reduction of a diester, silyl iodide promoted ring-opening iodo esterification of lactones, highly chemo- and regioselective ring-opening of an epoxy ester, dichloromethylation of a carboxyl group, and syn- and anti-selective reduction of the resulted β-hydroxy ketone as key steps.
Synthesis method of R-3-propyl-gamma-butyrolactone
-
Paragraph 0042; 0045-0048, (2020/07/12)
The invention discloses a synthetic method of R-3-propyl-gamma-butyrolactone, and belongs to the technical field of organic synthesis. The method comprises the following steps: by taking D-malic acidas a raw material, performing monomethyl esterification, reduction, halogenation or sulfonic acid esterification, and finally coupling with a Grignard reagent under the catalysis of zinc chloride to obtain a brivaracetam intermediate that is the R-3-propyl-gamma-butyrolactone. The method has the advantages of cheap and easily available starting raw materials, good stereoselectivity, no need of chiral resolution, mild condition, short route and the like, and provides a feasible scheme for brivaracetam process research.