6591-63-5 Usage
Description
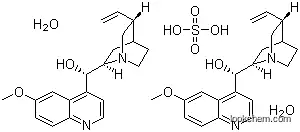
Quinidine sulfate dihydrate is a dextrorotatory diastereoisomer of quinine, derived from various species of Cinchona and their hybrids. It is an alkaloid with a hydroxymethyl group that serves as a link between a quinoline ring and a quinuclidine moiety. The structure contains two basic nitrogens, with the quinuclidine nitrogen being the stronger base (pKa 10). Quinidine sulfate dihydrate is bitter, light-sensitive, and nearly neutral or slightly alkaline in nature. It is soluble to the extent of 1% in water and more highly soluble in alcohol or chloroform.
Uses
Used in Pharmaceutical Industry:
Quinidine sulfate dihydrate is used as an antiarrhythmic agent (class IA) for the treatment of various cardiac arrhythmias. It helps regulate the heart's rhythm and is particularly effective in managing atrial and ventricular arrhythmias.
Used in Antimalarial Applications:
Quinidine sulfate dihydrate is used as an antimalarial drug for the treatment of malaria. It is effective against the Plasmodium parasite, which causes the disease, and helps alleviate the symptoms and prevent the progression of the infection.
Used in Chemical Research:
Quinidine sulfate dihydrate is used as a research compound in the field of chemistry, particularly for studying its chemical properties, interactions with other compounds, and potential applications in drug development.
Clinical Use
Quinidine sulfate is the prototype of antiarrhythmicdrugs and a class IA antiarrhythmic agent according to theVaughan Williams classification. It reduces Na+ current bybinding the open ion channels (i.e., state A). The decreasedNa+entry into the myocardial cell depresses phase 4 diastolicdepolarization and shifts the intracellular threshold potentialtoward zero. These combined actions diminish thespontaneous frequency of pacemaker tissues, depress theautomaticity of ectopic foci, and, to a lesser extent, reduceimpulse formation in the SA node. This last action results inbradycardia. During the spike action potential, quinidinesulfate decreases transmembrane permeability to passiveinflux of Na+, thus slowing the process of phase 0 depolarization,which decreases conduction velocity. This is shownas a prolongation of the QRS complex of electrocardiograms.Quinidine sulfate also prolongs action potential duration,which results in a proportionate increase in the QTinterval. It is used to treat supraventricular and ventricularectopic arrhythmias, such as atrial and ventricular prematurebeats, atrial and ventricular tachycardia, atrial flutter, andatrial fibrillation.
Check Digit Verification of cas no
The CAS Registry Mumber 6591-63-5 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 6,5,9 and 1 respectively; the second part has 2 digits, 6 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 6591-63:
(6*6)+(5*5)+(4*9)+(3*1)+(2*6)+(1*3)=115
115 % 10 = 5
So 6591-63-5 is a valid CAS Registry Number.
InChI:InChI=1/2C20H24N2O2.H2O4S.2H2O/c2*1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18;1-5(2,3)4;;/h2*3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3;(H2,1,2,3,4);2*1H2/t2*13-,14-,19+,20-;;;/m00.../s1