874338-91-7 Usage
General Description
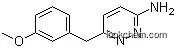
3-Pyridazinamine, 6-[(3-methoxyphenyl)methyl]- is a chemical compound that belongs to the class of pyridazines, which are organic heterocyclic compounds containing a pyridazine ring, characterized by a six-membered heterocyclic ring containing four carbon atoms, two nitrogen atoms, and two double bonds. This particular compound consists of a pyridazinamine core with a 6-[(3-methoxyphenyl)methyl]- moiety attached to it. It is commonly used in pharmaceutical research and development as a building block for the synthesis of various drugs and biologically active molecules. Additionally, it may also have potential applications in chemical and material sciences due to its unique structural and physicochemical properties.
Check Digit Verification of cas no
The CAS Registry Mumber 874338-91-7 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 8,7,4,3,3 and 8 respectively; the second part has 2 digits, 9 and 1 respectively.
Calculate Digit Verification of CAS Registry Number 874338-91:
(8*8)+(7*7)+(6*4)+(5*3)+(4*3)+(3*8)+(2*9)+(1*1)=207
207 % 10 = 7
So 874338-91-7 is a valid CAS Registry Number.
InChI:InChI=1/C12H13N3O/c1-16-11-4-2-3-9(8-11)7-10-5-6-12(13)15-14-10/h2-6,8H,7H2,1H3,(H2,13,15)
874338-91-7Relevant articles and documents
COMPOUNDS AND USES THEREOF
-
, (2020/10/20)
The present invention features compounds useful in the treatment of neurological disorders and primary brain cancer. The compounds of the invention, alone or in combination with other pharmaceutically active agents, can be used for treating or preventing neurological disorders and primary brain cancer.
A convenient one-pot Negishi coupling of amino-heteroaryl chlorides and alkyl bromides
Walters, Iain A.S.
, p. 341 - 344 (2007/10/03)
A simple Ni-catalysed cross-coupling protocol for amino-heteroaryl chlorides with alkylzinc reagents has been developed. The alkylzinc reagents can be commercially available dialkylzincs or alkylzinc halides, or can be conveniently generated in situ from diethylzinc and primary alkyl bromides in the presence of the same inexpensive Ni catalyst used to effect the subsequent coupling reaction.