FULL PAPER
(MeCN)2][ClO4]2, an equimolar mixture of triethylamine and for-
mic acid was then added (c = 3 mm in an NMR tube). H NMR
Acknowledgments
1
(500 MHz, CD3CN, 25 °C): δ = 0.93 (m, 12 H, CH3), 1.26–1.47 [m,
24 H, (CH2)3], 2.34 (m, 8 H, CH2CH), 3.73 (s, 3 H, ImCH3), 3.74
(s, 6 H, ImCH3), 4.17 (HCOOin), 4.30–4.43 (m, 8 H, OCH2,inO,
ArCH2O), 4.53–4.59 (m, 8 H, OCH2,inO, ImCH2O), 4.62 and 4.69
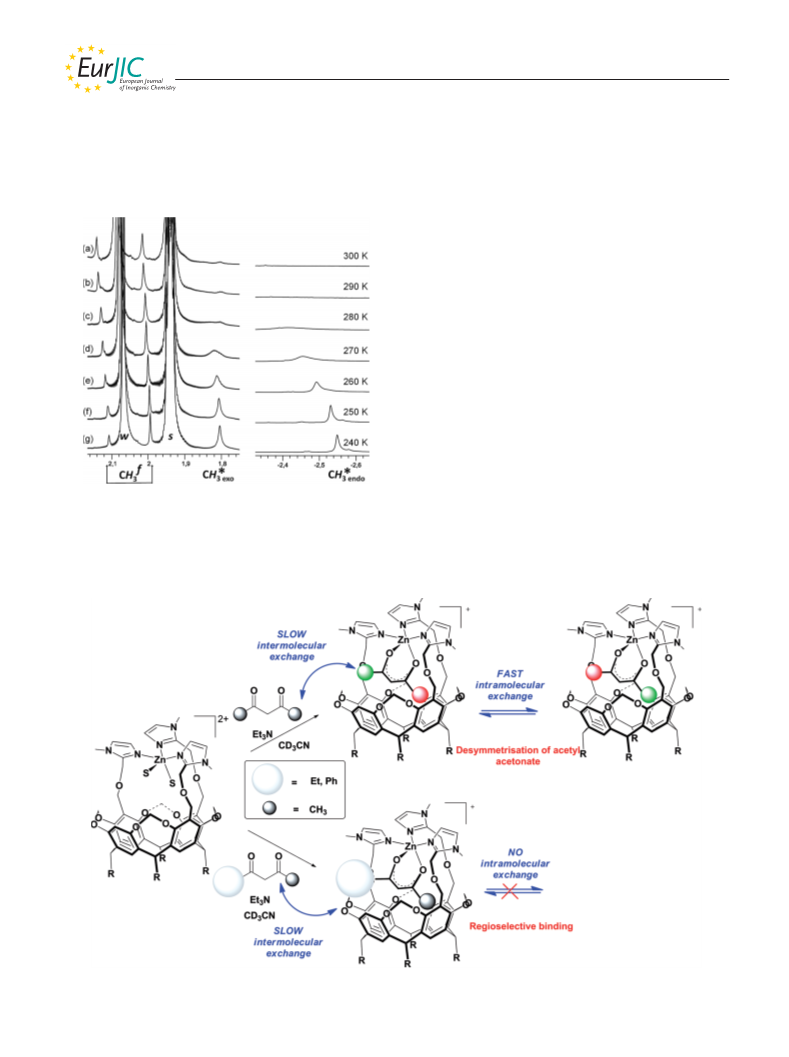
This project was supported by the Centre National de la Recherche
Scientifique (CNRS) (Institut de Chimie), the Ministère de l’En-
seignement Supérieur et de la Recherche, and the Agence Nationale
pour la Recherche [Cavity-zyme(Cu) Project No. ANR-2010-
BLAN-7141].
3
3
(t, JH,H = 8.0 Hz, 4 H, CH2CH), 5.47 and 5.48 [2 d, JH,H
=
7.6 Hz, 4 H, OCH2,outO], 6.26 (s, 2 H, ImH), 6.27 (s, 1 H, ArH
up), 6.34 (s, 1 H, ImH), 7.09 (s, 2 H, ImH), 7.11 (s, 1 H, ImH),
7.45 (s, 1 H, ArH low), 7.49 (s, 1 H, ArH low), 7.51 (s, 2 H, ArH
low) ppm.
[1] R. Gramage-Doria, D. Armspach, D. Matt, Coord. Chem. Rev.
2013, 257, 776–816.
[2] For cavities formed upon metal complexation by non-cavity-
shaped macrocyclic ligands, see: a) B. Kersting, Z. Anorg. Allg.
Chem. 2004, 630, 765–780; b) B. Kersting, U. Lehmann,
“Chemistry of Metalated Container Molecules” in Advances in
Inorganic Chemistry, vol. 61 (Eds.: R. van Eldik, C. D. Hub-
bard), Elsevier, The Netherlands, 2009, p. 407.
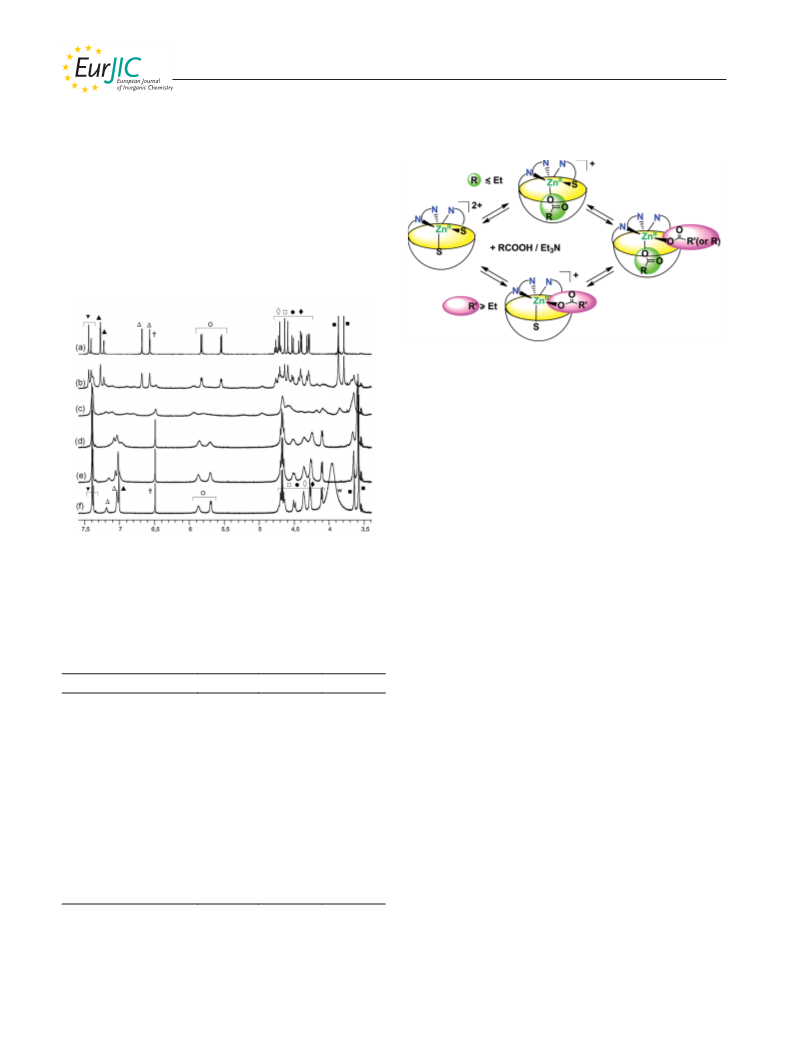
[Zn(Rim)(CH3COCHCOCH3)]+: An equimolar mixture containing
triethylamine and acetylacetone (2 equiv.) was added to a solution
of [Zn(Rim)(MeCN)2][ClO4]2 in CD3CN (c = 3.0 mm in an NMR
1
tube). H NMR (500 MHz, CD3CN, 25 °C): δ = –2.23 (very br. s,
3
3 H, CH3,inCOCHCOCH3), 0.91 and 0.92 (2 t, JH,H = 7.1 Hz,
[3] For cavities covalently linked to metals other than transition
elements, see: a) J. L. Atwood, L. J. Barbour, M. J. Hardie,
C. L. Raston, Coord. Chem. Rev. 2001, 222, 3–32; b) J. T. Len-
thall, J. W. Steed, Coord. Chem. Rev. 2007, 251, 1747–1760.
[4] For a very recent example, see: M. Guitet, P. Zhang, F. Mar-
celo, C. Tugny, J. Jiménez-Barbero, O. Buriez, C. Amatore, V.
Mouriès-Mansuy, J.-P. Goddard, L. Fensterbank, Y. Zhang, S.
Roland, M. Ménand, M. Sollogoub, Angew. Chem. Int. Ed.
2013, 52, 7213–7218; Angew. Chem. 2013, 125, 7354.
[5] S. Blanchard, L. Le Clainche, M.-N. Rager, B. Chansou, J. P.
Tuchagues, A. Duprat, Y. Le Mest, O. Reinaud, Angew. Chem.
Int. Ed. 1998, 37, 2732–2735; Angew. Chem. 1998, 110, 2861.
[6] C. Wieser-Jeunesse, D. Matt, A. De Cian, Angew. Chem. Int.
Ed. 1998, 37, 2861–2864; Angew. Chem. 1998, 110, 3027.
[7] O. Sénèque, M.-N. Rager, M. Giorgi, O. Reinaud, J. Am. Chem.
Soc. 2000, 122, 6183–6189.
12 H, CH3), 1.26–1.48 [m, 24 H, (CH2)3], 1.80 (very br. s, 3 H,
CH3,inCOCHCOCH3), 2.01 [s, free CH3COCHC(OH)CH3], 2.14 (s,
free CH3COCH2COCH3), 2.33 (m, 8 H, CH2CH), 3.58 (s, 6 H,
ImCH3), 3.60 (s, free CH3COCH2COCH3), 3.75 (s, 3 H, ImCH3),
4.15–4.77 (m, 20 H, ImCH2O, CH2CH, ArCH2O, OCH2,inO), 5.12
(br. s, 2 H, OCH2,outO), 5.61 [s, free CH3COCHC(OH)CH3], 5.73
3
(d, JH,H = 7.4 Hz, 2 H, OCH2,outO), 6.59 (s, 1 H, ArH up), 6.79
(s, 2 H, ImH), 6.83 (s, 1 H, ImH), 7.15 (s, 1 H, ImH), 7.05 (s, 2 H,
ImH), 7.40 (s, 3 H, ArH low), 7.42 (s, 1 H, ArH low) ppm. ESI-MS
(CH3CN): m/z = 676.3 [Zn(Rim)(CH3COCH2COCH3)]2+, 1351.5
[Zn(Rim)(CH3COCHCOCH3)]+.
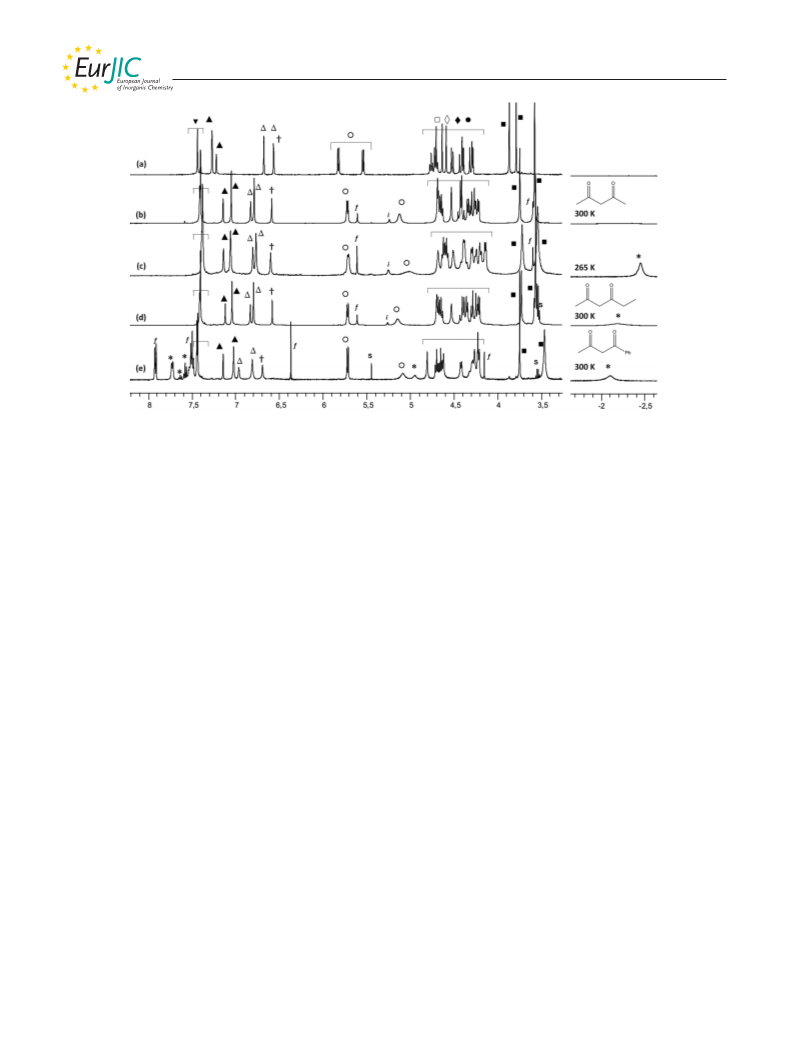
[Zn(Rim)(CH3COCHCOCH2CH3)]+: An equimolar mixture con-
taining triethylamine and hexane-2,4-dione (2 equiv.) was added to
a solution of [Zn(Rim)(MeCN)2][ClO4]2 in CD3CN (c = 3.0 mm in
an NMR tube). 1H NMR (500 MHz, CD3CN, 25 °C): δ = –2.19
(br. s, 3 H, CH3,inCOCHCOEt), 0.92 and 0.93 (2 t, 3JH,H = 7.1 Hz,
12 H, CH3), 0.97 and 1.08 [s, free MeCOCHC(OH)CH2CH3], 1.24–
1.47 [m, 24 H, (CH2)3], 2.08 [s, free EtCOCHC(OH)CH3], 2.14 (s,
free EtCOCH2COCH3), 2.36 (m, 8 H, CH2CH), 2.47 [s, free
MeCOCHC(OH)CH2CH3], 3.56 (s, 6 H, ImCH3), 3.58 (s, free
CH3COCH2COEt3), 3.73 (s, 3 H, ImCH3), 4.14–4.74 (m, 20 H,
[8] D. Coquière, S. Gac, U. Darbost, O. Sénèque, I. Jabin, O. Rein-
aud, Org. Biomol. Chem. 2009, 7, 2485–2500.
[9] The term “exogenous” is used here to designate ligands with
which the cavity complex interacts, in contrast to the cavity
ligand itself, which can be considered as “endogenous” to the
complex.
[10] a) L. Le Clainche, M. Giorgi, O. Reinaud, Inorg. Chem. 2000,
39, 3436–3437; b) L. Le Clainche, Y. Rondelez, O. Sénèque, S.
Blanchard, M. Campion, M. Giorgi, A. F. Duprat, Y. Le Mest,
O. Reinaud, C. R. Acad. Sci., Ser. IIc: Chim. 2000, 3, 811–819.
[11] G. Izzet, Y. M. Frapart, T. Prangé, K. Provost, A. Michalowicz,
O. Reinaud, Inorg. Chem. 2005, 44, 9743–9751.
[12] a) A. Zanotti-Gerosa, E. Solari, L. Giannini, C. Floriani, N.
Re, A. Chiesi-Villa, C. Rizzoli, Inorg. Chim. Acta 1998, 270,
298–311; b) F. Corazza, C. Floriani, A. Chiesi-Villa, C. Guas-
tini, J. Chem. Soc., Chem. Commun. 1990, 640–641; c) F. Co-
razza, C. Floriani, A. Chiesi-Villa, C. Rizzoli, Inorg. Chem.
1991, 30, 4465–4468; d) A. Arduini, C. Massera, A. Pochini,
A. Secchi, F. Ugozzoli, New J. Chem. 2006, 30, 952–958; e) P.
Mongrain, J. Douville, J. Gagnon, M. Drouin, A. Decken, D.
Fortin, P. D. Harvey, Can. J. Chem. 2004, 82, 1452–1461; f)
V. C. Gibson, C. Redshaw, W. Clegg, M. R. J. Elsegood, J.
Chem. Soc., Chem. Commun. 1995, 2371–2372.
[13] a) E. Engeldinger, D. Armspach, D. Matt, Angew. Chem. Int.
Ed. 2001, 40, 2526–2529; b) E. Engeldinger, D. Armspach, D.
Matt, P. G. Jones, Chem. Eur. J. 2003, 9, 3091–3105.
[14] D. Armspach, D. Matt, F. Peruc, P. Lutz, Eur. J. Inorg. Chem.
2003, 805–809.
[15] a) I. Bertini, H. B. Gray, E. I. Stiefel, J. S. Valentine in Bio-
logical Inorganic Chemistry, Structure and Reactivity, Univer-
sity Science Books, Sausalito, 2007; b) S. J. Lippard, J. M. Berg
in Principles of Bioinorganic Chemistry, University Science
Books, Mill Valley, 1994; c) R. H. Holm, P. Kennepohl, E. I.
Solomon, Chem. Rev. 1996, 96, 2239–2314.
ImCH2O, CH2CH, m, ArCH2O, OCH2,inO), 5.15 (br. s, 2 H,
3
OCH2,outO), 5.62 [s, free CH3COCHC(OH)Et], 5.72 (d, JH,H
=
7.3 Hz, 2 H, OCH2,outO), 6.58 (s, 1 H, ArH up), 6.84 (s, 1 H, ImH),
6.81 (s, 2 H, ImH), 7.05 (s, 2 H, ImH), 7.12 (s, 1 H, ImH), 7.41 (s,
3 H, ArH low), 7.42 (s, 1 H, ArH low) ppm.
[Zn(Rim)(CH3COCHCOPh)]+: 1-Benzoylacetone (2 equiv.) in
CD3CN was added to a solution of [Zn(Rim)(MeCN)2][ClO4]2 in
CD3CN (c = 4.3 mm) in an NMR tube, followed by the addition
of triethylamine (1 equiv.). 1H NMR (500 MHz, CD3CN, 25 °C): δ
= –2.10 [br. s, 3 H, bound CH3COCHC(O)Ph], 0.92 and 0.93 (2 t,
3JH,H = 7.1 Hz, 12 H, CH3), 1.48–1.26 [m, 24 H, (CH2)3], 2.18 [s,
free PhCOCHC(OH)CH3], 2.23 (s, free PhCOCH2COCH3), 2.36
(m, 8 H, CH2CH), 3.46 (s, 6 H,ImCH3), 3.75 (s, 3 H, ImCH3), 4.15
(s, free CH3COCH2COPh), 4.18–4.84 (m, 20 H, ImCH2O, CH2CH,
ArCH2O, OCH2,inO), 4.95 [s, 1 H, bound CH3COCHC(O)Ph], 5.09
3
(m, 2 H, OCH2,outO), 5.72 (d, JH,H = 7.4 Hz, 2 H, OCH2,outO),
6.37 [free CH3COCHC(OH)Ph], 6.70 (s, 1 H, ArH up, s), 6.81 (s,
2 H, ImH), 6.97 (s, 1 H, ImH), 7.03 (s, 2 H, ImH), 7.15 (s, 1 H,
ImH), 7.38–7.67 (m, free and bound CHmetaPhArH, free and
3
bound CHorthoPh, ArH low), 7.72 (d, JH,H = 7.7 Hz, 2 H, coord.
3
CHorthoPh), 7.92 (d, JH,H = 7.7 Hz, free CHorthoPh) ppm.
Supporting Information (see footnote on the first page of this arti-
cle): NMR and ESI mass spectra.
[16] a) W. N. Lipscomb, N. Sträter, Chem. Rev. 1996, 96, 2375–2433;
b) G. Parkin, Chem. Rev. 2004, 104, 699–767.
Eur. J. Inorg. Chem. 2014, 2819–2828
2827
© 2014 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim

 Gout, Jér?me
Gout, Jér?me
 Rat, Stéphanie
Rat, Stéphanie
 Bistri, Olivia
Bistri, Olivia
 Reinaud, Olivia
Reinaud, Olivia