
Carbohydrate Research p. 26 - 31 (1999)
Update date:2022-08-30
Topics:
 Tokuyasu, Ken
Tokuyasu, Ken
 Ono, Hiroshi
Ono, Hiroshi
 Hayashi, Kiyoshi
Hayashi, Kiyoshi
 Mori, Yutaka
Mori, Yutaka
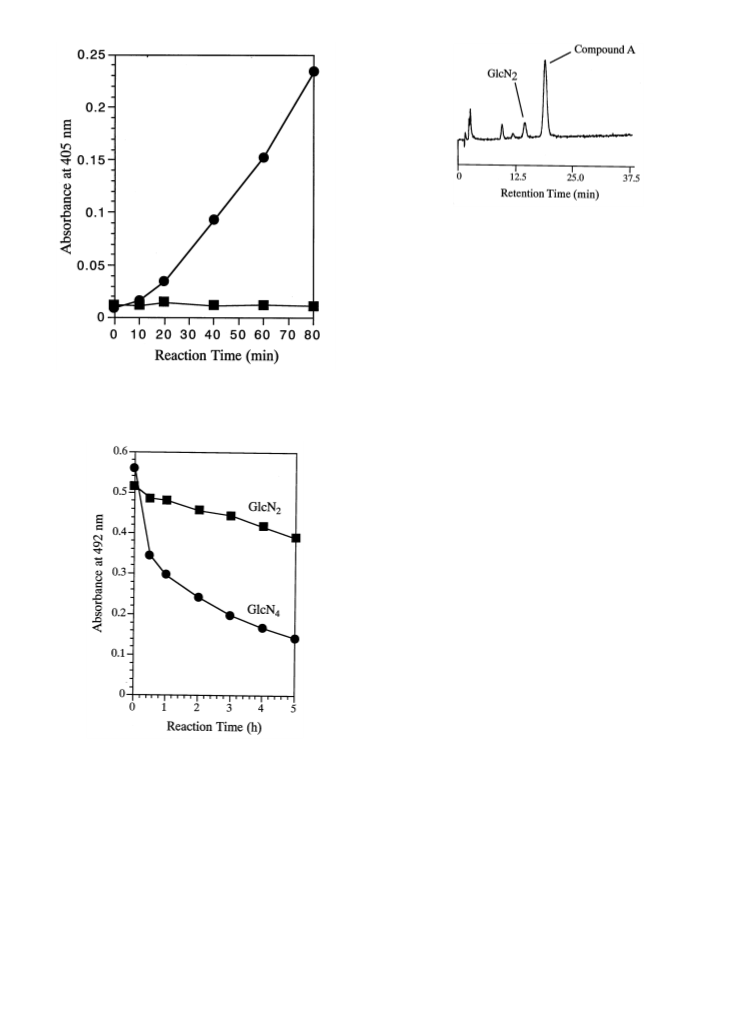
We found that a chitin deacetylase from Colletotrichum lindemuthianum could acetylate free amino sugar residues into N-acetylated forms in the presence of 3.0 M sodium acetate. The result was analyzed using a β-N-acetylhexosaminidase-coupled assay system with p-nitrophenyl 2-amino-2-deoxy-β-D-glucopyranosyl-(1→4)-2-acetamido-2-deoxy-β-D-glucopyranoside as the substrate, and the liberation of p-nitrophenol was observed as a consequence of enzymatic N-acetylation of the glucosamine residue at the nonreducing end of the substrate. The chitin deacetylase also acetylated chitobiose and chitotetraose as substrates, which was evidenced by the decrease in the amount of free amino sugar residues in the chitooligosaccharides. The reaction product of chitobiose after the acetylation reaction was exclusively 2-acetamido-2-deoxy-β-D-glucopyranosyl-(1→4)-2-amino-2-deoxy-D-glucose [GlcNAcGlcN], the structure of which was determined by FABMS and NMR analyses. This study offers a novel method for enzymatic N-acetylation of amino sugars, and especially with chitobiose as substrate, a selectively N-acetylated product, GlcNAcGlcN, can be synthesized. Copyright (C) 1999 Elsevier Science Ltd.
View More

Shanghai Chemchallenger Biotech Co., Ltd.
Contact:0086-21-68110815
Address:Room 2056, Building 1, No. 1065, Jiaxin Road Shanghai China (Mainland) 201800
FOSHAN NANHAI ZHONGNAN PHARMACEUTICAL FACTORY
Contact:0086-0757-85609331
Address:XIAHENGTIAN INDUSTRIAL ZONE,SHAYONG VILLAGE,LISHUI TOWN
SEA BRGIHT INDUSTRY CO.,LIMITED
Contact:0086 755 8622 3990
Address:Rm 17B3,GuangCaiXinTianDi Bldg,GuiMiao Rd,NanShan District,Shenzhen,China
HANGZHOU ZHONGCHANG SCIENTIFIC CO.,LTD
Contact:+86-571-88932183
Address:Hangzhou
website:http://www.finetechnology-ind.com
Contact:86-27-87465837
Address:wuhan
Doi:10.1016/S0022-328X(00)99534-2
(1986)Doi:10.1002/aoc.5741
(2020)Doi:10.1007/BF00864173
(1992)Doi:10.1524/zpch.2013.0377
(2013)Doi:10.1016/j.bmcl.2016.10.079
(2016)Doi:10.1021/om501287k
(2015)